Abstract
Rationale
Baseline performance has been reported to predict dopamine (DA) effects on working memory, following an inverted-U pattern. This pattern may hold true for other executive functions that are DA-sensitive.
Objectives
The objective of this study is to investigate the effect of d-amphetamine, an indirect DA agonist, on two other putatively DA-sensitive executive functions, inhibition and motor planning, as a function of baseline performance.
Methods
Participants with no prior stimulant exposure participated in a double-blind crossover study of a single dose of 0.3 mg/kg, p.o. of d-amphetamine and placebo. Participants were divided into high and low groups, based on their performance on the antisaccade and predictive saccade tasks on the baseline day. Executive functions, mood states, heart rate and blood pressure were assessed before (T0) and after drug administration, at 1.5 (T1), 2.5 (T2) and 3.5 h (T3) post-drug.
Results
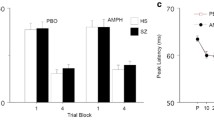
Antisaccade errors decreased with d-amphetamine irrespective of baseline performance (p = 0.025). For antisaccade latency, participants who generated short-latency antisaccades at baseline had longer latencies on d-amphetamine than placebo, while those with long-latency antisaccades at baseline had shorter latencies on d-amphetamine than placebo (drug × group, p = 0.04). d-amphetamine did not affect motor planning. Ratings of mood improved on d-amphetamine (p < 0.001). Magnitude of d-amphetamine-induced changes in elation was related to baseline reaction time variability.
Conclusions
d-amphetamine reduced antisaccade error rates in healthy controls, replicating and extending findings with DA agonists in clinical populations. d-amphetamine had baseline-dependent effects on antisaccade latency, consistent with an inverted-U relationship between performance and DA activity.



Similar content being viewed by others
References
Acheson A, de Wit H (2008) Buproprion improves attention but does not affect impulsive behavior in healthy volunteers. Exp Clin Psychopharm 16:113–123
Alexander GE, DeLong MR, Strick PL (1986) Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu Rev Neurosci 9(1):357–381
Amador N, Schlag-Rey M, Schlag J (2004) Primate antisaccade. II. Supplementary eye field neuronal activity predicts correct performance. J Neurophysiol 91(4):1672–1689
Amador SC, Hood AJ, Schiess MC, Izor R, Sereno AB (2006) Dissociating cognitive deficits involved in voluntary eye movement dysfunctions in Parkinson's disease patients. Neuropsychologia 44(8):1475–1482
Asghar SJ, Baker GB, Rauw GA, Silverstone PH (2001) A rapid method of determining amphetamine in plasma samples using pentafluorobenzenesulfonyl chloride and electron-capture gas chromatography. J Pharmacol Toxicol 46(2):111–115
Bäckman L, Lindenberger U, Li S, Nyberg L (2010) Linking cognitive aging to alterations in dopamine neurotransmitter functioning: recent data and future avenues. Neurosci Biobehav R 34(5):670–677
Baddeley A (1992) Working memory. Science 255(5044):556–559
Barch DM (2004) Pharmacological manipulation of human working memory. Psychopharmacology 174(1):126–135
Barch DM, Carter CS (2005) Amphetamine improves cognitive function in medicated individuals with schizophrenia and in healthy volunteers. Schizophr Res 77(1):43–58
Barkley RA (1997) Attention-deficit/hyperactivity disorder, self-regulation, and time: toward a more comprehensive theory. J Dev Behav Pediatr 18(4):271–279
Barkley RA, Grodzinsky G, DuPaul GJ (1992) Frontal lobe functions in attention deficit disorder with and without hyperactivity: a review and research report. J Abnorm Child Psych 20(2):163–188
Bedard A, Martinussen R, Ickowicz A, Tannock R (2004) Methylphenidate improves visual-spatial memory in children with attention-deficit/hyperactivity disorder. J Am Acad Child Psy 43(3):260
Bekker EM, Böcker KBE, Van Hunsel F, van den Berg MC, Kenemans JL (2005) Acute effects of nicotine on attention and response inhibition. Pharmacol Biochem Behav 82(3):539–548
Ben-Pazi H, Gross-Tsur V, Bergman H, Shalev RS (2003) Abnormal rhythmic motor response in children with attention-deficit-hyperactivity disorder. Dev Med Child Neurol 45(11):743–745
Bokura H, Yamaguchi S, Kobayashi S (2005) Event-related potentials for response inhibition in Parkinson's disease. Neuropsychologia 43(6):967–975
Briand KA, Strallow D, Hening W, Poizner H, Sereno AB (1999) Control of voluntary and reflexive saccades in Parkinson's disease. Exp Brain Res 129(1):38–48
Bronstein AM, Kennard C (1985) Predictive ocular motor control in Parkinson's disease. Brain 108(Pt 4):925–940
Burton C, Strauss E, Hultsch D, Moll A, Hunter M (2006) Intraindividual variability as a marker of neurological dysfunction: a comparison of Alzheimer's disease and Parkinson's disease. J Clin Exp Neuropsych 28(1):67–83
Camicioli RM, Wieler M, de Frias CM, Martin WW (2008) Early, untreated Parkinson's disease patients show reaction time variability. Neurosci Lett 441(1):77–80
Carr LA, Nigg JT, Henderson JM (2006) Attentional versus motor inhibition in adults with attention-deficit/hyperactivity disorder. Neuropsychology 20(4):430–441
Carter CS, Krener P, Chaderjian M, Northcutt C, Wolfe V (1995) Abnormal processing of irrelevant information in attention deficit hyperactivity disorder. Psychiat Res 56(1):59–70
Chan F, Armstrong IT, Pari G, Riopelle RJ, Munoz DP (2005) Deficits in saccadic eye-movement control in Parkinson's disease. Neuropsychologia 43(5):784–796
Chan C, Napolitano M, Foster G (2009) Assessment of general personality and psychopathology among persons with eating and weight-related concerns. In: Allison DB (ed) Handbook of assessment methods for eating behaviours and weight-related problems: measures, theory and research. SAGE Publications, Thousand Oaks, pp 1–33
Cools R (2006) Dopaminergic modulation of cognitive function-implications for l-DOPA treatment in Parkinson's disease. Neurosci Biobehav Rev 30(1):1–23
Cornelissen F, Kimmig H, Rutschmann RM, Maguire RP, Broerse A et al (2002) Event-related fMRI responses in the human frontal eye fields in a randomized pro- and antisaccade task. Exp Brain Res 145(2):270–274
Curran SL, Andrykowski MA, Studts JL (1995) Short form of the Profile of Mood States (POMS-SF): psychometric information. Psychol Assess 7(1):80–83
de Wit H (2009) Impulsivity as a determinant and consequence of drug use: a review of underlying processes. Addict Biol 14:22–31
de Wit H, Crean J, Richards JB (2000) Effects of d-amphetamine and ethanol on a measure of behavioral inhibition in humans. Behav Neurosci 114(4):830–837
de Wit H, Enggasser JL, Richards JB (2002) Acute administration of d-amphetamine decreases impulsivity in healthy volunteers. Neuropsychopharmacol 27(5):813–825
Dépatie L, O'Driscoll GA, Holohan AL, Atkinson V, Thavundayil JX, Kin NN et al (2002) Nicotine and behavioral markers of risk for schizophrenia: a double-blind, placebo-controlled, cross-over study. Neuropsychopharmacol 27(6):1056–1070
Devous MD, Trivedi MH, Rush AJ (2001) Regional cerebral blood flow response to oral amphetamine challenge in healthy volunteers. J Nucl Med 42(4):535–542
Dimoska A, Johnstone SJ, Barry RJ, Clarke AR (2003) Inhibitory motor control in children with attention-deficit/hyperactivity disorder: event-related potentials in the stop-signal paradigm. Biol Psychiat 54(12):1345–1354
Dommett EJ, Overton PG, Greenfield SA (2009) Drug therapies for attentional disorders alter the signal-to-noise ratio in the superior colliculus. Neuroscience 164(3):1369–1376
Duka T, Lupp A (1997) The effects of incentive on antisaccades: is a dopaminergic mechanism involved? Behav Pharmacol 8(5):373–382
Eagle D, Tufft M, Goodchild H, Robbins T (2007) Differential effects of modafinil and methylphenidate on stop-signal reaction time task performance in the rat, and interactions with the dopamine receptor antagonist cis-flupenthixol. Psychopharmacology 192(2):193–206
Elsinger CL, Rao SM et al (2003) Neural basis for impaired time reproduction in Parkinson's disease: an fMRI study. J Int Neuropsychol Soc 9(7):1088–1098
Engle RW (2002) Working memory capacity as executive attention. Curr Dir Psychol Sci 11(1):19–23
Epstein JN, Conners CK, Hervey AS, Tonev ST, Arnold LE, Abikoff HB et al (2005) Assessing medication effects in the MTA study using neuropsychological outcomes. J Child Psychol Psyc 47(5):446–456
Ernst M, Matochik JA, Heishman SJ, Van Horn JD, Jons PH, Henningfield JE et al (2001) Effect of nicotine on brain activation during performance of a working memory task. P Natl Acad Sci (USA) 98(8):4728–4733
Everling S, DeSouza JFX (2005) Rule-dependent activity for prosaccades and antisaccades in the primate prefrontal cortex. J Cogn Neurosci 17(9):1483–1496
Everling S, Fischer B (1998) The antisaccade: a review of basic research and clinical studies. Neuropsychologia 36(9):885–899
Everling S, Munoz DP (2000) Neuronal correlates for preparatory set associated with pro-saccades and anti-saccades in the primate frontal eye field. J Neurosci 20(1):387–400
Everling S, Dorris MC, Klein RM, Munoz DP (1999) Role of primate superior colliculus in preparation and execution of anti-saccades and pro-saccades. J Neurosci 19(7):2740–2754
Feifel D, Farber RH, Clementz BA, Perry W, Anllo-Vento L (2004) Inhibitory deficits in ocular motor behavior in adults with attention-deficit/hyperactivity disorder. Biol Psychiat 56(5):333–339
Fillmore MT, Rush CR, Hays L (2006) Acute effects of cocaine in two models of inhibitory control: implications of non-linear dose effects. Addiction 101(9):1323–1332
First M, McQueen L, Lincus H (1996) DSM-IV coding update (1996 edn). American Psychiatric Publishing Inc., Arlington
Fitzpatrick P, Klorman R, Brumaghim J, Borgstedt A (1992) Effects of sustained-release and standard preparations of methylphenidate on attention deficit disorder. J Am Acad Child Psy 31(2):226–234
Gagnon D, O'Driscoll GA, Petrides M, Pike GB (2002) The effect of spatial and temporal information on saccades and neural activity in oculomotor structures. Brain 125(1):123–139
Gauggel S, Rieger M, Feghoff T (2004) Inhibition of ongoing responses in patients with Parkinson's disease. J Neurol Neurosur Ps 75(4):539–544
Gaymard B, Ploner CJ, Rivaud S, Vermersch AI, Pierrot-Deseilligny C (1998) Cortical control of saccades. Exp Brain Res 123(1):159–163
Gibbs S, D'Esposito M (2005a) A functional MRI study of the effects of bromocriptine, a dopamine receptor agonist, on component processes of working memory. Psychopharmacology 180(4):1–10
Gibbs SEB, D'Esposito M (2005b) Individual capacity differences predict working memory performance and prefrontal activity following dopamine receptor stimulation. Cogn Affect Behav Neurosci 5(2):212–221
Goldman-Rakic PS, Muly EC, Williams GV (2000) D1 receptors in prefrontal cells and circuits. Brain Res Rev 31(2–3):295–301
Gowan JD, Coizet V, Devonshire IM, Overton PG (2008) d-amphetamine depresses visual responses in the rat superior colliculus: a possible mechanism for amphetamine-induced decreases in distractibility. J Neural Transm 115(3):377–387
Guitton D, Buchtel H, Douglas R (1985) Frontal lobe lesions in man cause difficulties in suppressing reflexive glances and in generating goal-directed saccades. Exp Brain Res 58(3):455–472
Hanes D, Patterson W II, Schall J (1998) Role of frontal eye fields in countermanding saccades: visual, movement, and fixation activity. J Neurophysiol 79(2):817–834
Hanisch C, Radach R, Holtkamp K, Herpertz-Dahlmann B, Konrad K (2006) Oculomotor inhibition in children with and without attention-deficit/hyperactivity disorder (ADHD). J Neural Transm 113(5):671–684
Heathcote A, Brown S, Mewhort DJ (2000) The power law repealed: the case for an exponential law of practice. Psychon B Rev 7(2):185–207
Hershey T, Black KJ, Hartlein J, Braver TS, Barch DM, Carl JL et al (2004) Dopaminergic modulation of response inhibition: an fMRI study. Cognitive Brain Res 20(3):438–448
Hervey AS, Epstein JN, Curry JF, Tonev S, Arnold LE, Conners CK et al (2006) Reaction time distribution analysis of neuropsychological performance in an ADHD sample. Child Neuropsychol 12(2):125–140
Hood AJ, Amador SC, Cain AE, Briand KA, Al-Refai AH, Schiess MC et al (2007) Levodopa slows prosaccades and improves antisaccades: an eye movement study in Parkinson's disease. J Neurol Neurosur Ps 78(6):565–570
Johnson KA, Barry E, Bellgrove MA, Cox M, Kelly SP, Daibhis A et al (2008) Dissociation in response to methylphenidate on response variability in a group of medication naïve children with ADHD. Neuropsychologia 46(5):1532–1541
Kahramanoglou I, Perantonis S, Smyrnis N, Evdokimidis I, Cutsuridis V (2008) Modeling the effects of dopamine on the antisaccade reaction times (aSRT) of schizophrenia patients. In Artificial Neural Networks—ICANN 2008, pp 290–299. Available at: http://dx.doi.org/10.1007/978-3-540-87559-8_30. Accessed 20 Mar 2010
Karatekin C, Bingham C, White T (2009) Oculomotor and pupillometric indices of pro- and antisaccade performance in youth-onset psychosis and attention deficit/hyperactivity disorder. Schizophr Bull:sbp035
Kimberg DY, Aguirre GK, Lease J, D'Esposito M (2001) Cortical effects of bromocriptine, a D-2 dopamine receptor agonist, in human subjects, revealed by fMRI. Hum Brain Mapp 12(4):246–257
Klein C, Ettinger U (2008) A hundred years of eye movement research in psychiatry. Brain Cogn 68(3):215–218
Konrad K, Gunther T, Hanisch C, Herpertz-Dahlmann B (2004) Differential effects of methylphenidate on attentional functions in children with attention-deficit/hyperactivity disorder. J Am Acad Child Psy 43(2):191–198
Kosslyn SM, Cacioppo JT, Davidson RJ, Hugdahl K, Lovallo WR, Spiegel D et al (2002) Bridging psychology and biology. The analysis of individuals in groups. Am Psychol 57(5):341–351
Larrison AL, Briand KA, Sereno AB (2004) Nicotine improves antisaccade task performance without affecting prosaccades. Human Psychopharm Clin 19(6):409–419
Leth-Steensen C, King Elbaz Z, Douglas VI (2000) Mean response times, variability, and skew in the responding of ADHD children: a response time distributional approach. Acta Psychol 104(2):167–190
LeVasseur AL, Flanagan JR, Riopelle RJ, Munoz DP (2001) Control of volitional and reflexive saccades in Tourette's syndrome. Brain 124(Pt 10):2045–2058
Levin ED, Conners CK, Silva D, Hinton SC, Meck WH, March J et al (1998) Transdermal nicotine effects on attention. Psychopharmacology 140(2):135–141
Leyton M, aan het Rot M, Booij L, Baker GB, Young SN, Benkelfat C (2007) Mood-elevating effects of d-amphetamine and incentive salience: the effect of acute dopamine precursor depletion. J Psychiatr Neurosci 32(2):129–136
Lichter DG, Cummings JL (2001) Frontal-subcortical circuits in psychiatric and neurological disorders. Guilford Press, New York
Loe IM, Feldman HM, Yasui E, Luna B (2009) Oculomotor performance identifies underlying cognitive deficits in attention-deficit/hyperactivity disorder. J Am Acad Child Psy 48(4):431–440
Logan GD (2004) Working memory, task switching, and executive control in the task span procedure. J Exp Psychol Gen 133(2):218–236
Luciana M, Collins P (1997) Dopaminergic modulation of working memory for spatial but not object cues in normal humans. J Cogn Neurosci 9(3):330–347
Luciana M, Collins P, Depue R (1998) Opposing roles for dopamine and serotonin in the modulation of human spatial working memory functions. Cereb Cortex 8(3):218–226
MacDonald SWS, Nyberg L, Bäckman L (2006) Intra-individual variability in behavior: links to brain structure, neurotransmission and neuronal activity. Trends Neurosci 29(8):474–480
MacDonald SW, Cervenka S, Farde L, Nyberg L, Bäckman L (2009) Extrastriatal dopamine D2 receptor binding modulates intraindividual variability in episodic recognition and executive functioning. Neuropsychologia 47(11):2299–2304
Manoach DS (2003) Prefrontal cortex dysfunction during working memory performance in schizophrenia: reconciling discrepant findings. Schizophr Res 60(2–3):285–298
Mattay VS, Callicott JH, Bertolino A, Heaton I, Frank JA, Coppola R et al (2000) Effects of dextroamphetamine on cognitive performance and cortical activation. Neuroimage 12(3):268–275
Mattay VS, Goldberg TE, Mattay VS, Kolochana BS, Callicott JH, Egan MF et al (2003) Catechol O-methyltransferase val158-met genotype and individual variation in the brain response to amphetamine. P Natl Acad Sci (USA) 100(10):6186–6191
McCloskey M, Palmer A, de Wit H (2010) Are attention lapses related to d-amphetamine liking? Psychopharmacology 208(2):201–209
McNair D, Lorr M, Droppleman L (1988) Manual for the profile of mood states. Educational and Industrial Testing Service, San Diego
Mostofsky SH, Lasker AG, Cutting LE, Denckla MB, Zee DS (2001) Oculomotor abnormalities in attention deficit hyperactivity disorder: a preliminary study. Neurology 57(3):423–430
Munoz DP, Everling S (2004) Look away: the anti-saccade task and the voluntary control of eye movement. Nat Rev Neurosci 5(3):218–228
Munoz DP, Le Vasseur AL, Flanagan JR (2002) Control of volitional and reflexive saccades in Tourette's syndrome. Prog Brain Res 140:467–481
Munoz DP, Armstrong IT, Hampton KA, Moore KD (2003) Altered control of visual fixation and saccadic eye movements in attention-deficit hyperactivity disorder. J Neurophysiol 90(1):503–514
Murtha S, Cismaru R, Waechter R, Chertkow H (2002) Increased variability accompanies frontal lobe damage in dementia. J Int Neuropsych Soc 8(3):360–372
Nigg JT, Butler KM, Huang-Pollock CL, Henderson JM (2002) Inhibitory processes in adults with persistent childhood onset ADHD. J Consult Clin Psych 70(1):153–157
O'Boyle DJ, Freeman JS, Cody FWJ (1996) The accuracy and precision of timing of self-paced, repetitive movements in subjects with Parkinson's disease. Brain 119(1):51–70
O'Driscoll GA, Depatie L, Holohan AL, Savion-Lemieux T, Barr RG, Jolicoeur C et al (2005) Executive functions and methylphenidate response in subtypes of attention-deficit/hyperactivity disorder. Biol Psychiat 57(11):1452–1460
O'Sullivan EP, Shaunak S, Henderson L, Hawken M, Crawford TJ, Kennard C (1997) Abnormalities of predictive saccades in Parkinson's disease. NeuroReport 8(5):1209–1213
Paré M, Hanes DP (2003) Controlled movement processing: superior colliculus activity associated with countermanded saccades. J Neurosci 23(16):6480–6489
Pierrot-Deseilligny C, Rivaud S, Gaymard B, Agid Y (1991) Cortical control of reflexive visually-guided saccades. Brain 114(Pt 3):1473–1485
Pierrot-Deseilligny C, Milea D, Mϋri RM (2004) Eye movement control by the cerebral cortex. Curr Opin Neurol 17(1):17–25
Powell J, Dawkins L, Davis RE (2002) Smoking, reward responsiveness, and response inhibition: tests of an incentive motivational model. Biol Psychiat 51(2):151–163
Rasch P, Pierson W, Brubaker M (1960) The effect of amphetamine sulfate and meprobamate on reaction time and movement time. Eur J Appl Physiol O 18(3):280–284
Riccardi P, Zald D, Li R, Park S, Ansari S, Dawant B et al (2006) Sex differences in amphetamine-induced displacement of [18F] fallypride in striatal and extrastriatal regions: a PET study. Am J Psychiatry 163(9):1639–1641
Riccio CA, Waldrop JJ, Reynolds CR, Lowe P (2001) Effects of stimulants on the continuous performance test (CPT): implications for CPT use and interpretation. J Neuropsychiatry Clin Neurosci 13(3):326–335
Rickard TC (1997) Bending the power law: a CMPL theory of strategy shifts and the automatization of cognitive skills. J Exp Psychol Gen 126(3):288–311
Rivaud S, Müri RM, Gaymard B, Vermersch AI, Pierrot-Deseilligny C (1994) Eye movement disorders after frontal eye field lesions in humans. Exp Brain Res 102(1):110–120
Robbins TW (2000) Chemical neuromodulation of frontal-executive functions in humans and other animals. Exp Brain Res 133(1):130–138
Robbins T, Arnsten A (2009) The neuropsychopharmacology of fronto-executive function: monoaminergic modulation. Ann Rev Neurosci 32(1):267–287
Roesch-Ely D, Scheddel H, Weiland S, Schwaniger M, Hundemer H-P, Kolter T et al (2005) Differential dopaminergic modulation of executive control in healthy subjects. Psychopharmacology 178(4):420–430
Rosa-Neto P, Lou HC, Cumming P, Pryds O, Karrebaek H, Lunding J et al (2005) Methylphenidate-evoked changes in striatal dopamine correlate with inattention and impulsivity in adolescents with attention deficit hyperactivity disorder. Neuroimage 25(3):868–876
Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FI et al (2001) Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin. Synapse 39(1):32–41
Rycroft N, Hutton SB, Rusted JM (2006) The antisaccade task as an index of sustained goal activation in working memory: modulation by nicotine. Psychopharmacology 188(4):521–529
Sato TR, Schall JD (2003) Effects of stimulus-response compatibility on neural selection in frontal eye field. Neuron 38(4):637–648
Scheres A, Oosterlaan J, Swanson J, Morein-Zamir S, Meiran N, Schut H et al (2003) The effect of methylphenidate on three forms of response inhibition in boys with AD/HD. J Abnorm Child Psych 31(1):105–120
Scheres A, Milham MP, Knutson B, Castellanos FX (2007) Ventral striatal hyporesponsiveness during reward anticipation in attention-deficit/hyperactivity disorder. Biol Psychiat 61(5):720–724
Schlag-Rey M, Amador N, Sanchez H, Schlag J (1997) Antisaccade performance predicted by neuronal activity in the supplementary eye field. Nature 390(6658):398–401
Schott BH et al (2007) Ageing and early-stage Parkinson's disease affect separable neural mechanisms of mesolimbic reward processing. Brain 130(Pt 9):2412–2424
Seamans JK, Yang CR (2004) The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol 74(1):1–58
Stelmach GE, Worringham CJ, Strand EA (1987) The programming and execution of movement sequences in Parkinson's disease. Int J Neurosci 36(1–2):55–65
Stuss DT, Murphy KJ, Binns MA, Alexander MP (2003) Staying on the job: the frontal lobes control individual performance variability. Brain 126(11):2363–2380
Tannock R, Schachar RJ, Carr RP, Chajczyk D, Logan GD (1989) Effects of methylphenidate on inhibitory control in hyperactive children. J Abnorm Child Psych 17(5):473–491
Toplak ME, Dockstader C, Tannock R (2006) Temporal information processing in ADHD: findings to date and new methods. J Neurosci Meth 151(1):15–29
Tucha O et al (2006) Effects of methylphenidate on multiple components of attention in children with attention deficit hyperactivity disorder. Psychopharmacology 185(3):315–326
Unsworth N, Schrock JC, Engle RW (2004) Working memory capacity and the antisaccade task: individual differences in voluntary saccade control. J Exp Psychol 30(6):1302–1321
Vaidya CJ, Austin G, Kirkorian G, Ridlehuber HW, Desmond JE, Glover GH et al (1998) Selective effects of methylphenidate in attention deficit hyperactivity disorder: a functional magnetic resonance study. P Natl Acad Sci (USA) 95(24):14494–14499
Vossel S, Thiel CM, Fink GR (2008) Behavioral and neural effects of nicotine on visuospatial attentional reorienting in non-smoking subjects. Neuropsychopharmacol 33(4):731–738
Wonodi I, Cassady SL, Adami H, Avila M, Thaker GK (2006) Effects of repeated amphetamine administration on antisaccade in schizophrenia spectrum personality. Psychiat Res 141(3):237–245
Acknowledgments
This research was supported by an NSERC Doctoral Fellowship (AA), a William Dawson Scholar Award (GO'D, ML), a James McGill Professorship (CB), a GW Stairs Memorial Fund grant (GO'D) and operating funds from the Canadian Institutes of Health Research (CB, AD, ML and G'OD). The expert technical assistance of Gail Rauw is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Allman, AA., Benkelfat, C., Durand, F. et al. Effect of d-amphetamine on inhibition and motor planning as a function of baseline performance. Psychopharmacology 211, 423–433 (2010). https://doi.org/10.1007/s00213-010-1912-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-010-1912-x