Abstract
Rationale
2-([2-(4-cyano-2,5-dimethoxyphenyl)ethylamino]methyl)phenol (25CN-NBOH) is structurally similar to N-benzyl substituted phenethylamine hallucinogens currently emerging as drugs of abuse. 25CN-NBOH exhibits dramatic selectivity for 5-HT2A receptors in vitro, but has not been behaviorally characterized.
Objective
25CN-NBOH was compared to the traditional phenethylamine hallucinogen R(-)-2,5-dimethoxy-4-iodoamphetamine (DOI) using mouse models of drug-elicited head twitch behavior and drug discrimination.
Methods
Drug-elicited head twitches were quantified for 10 min following administration of various doses of either DOI or 25CN-NBOH, with and without pretreatments of 0.01 mg/kg 5-HT2A antagonist M100907 or 3.0 mg/kg 5-HT2C antagonist RS102221. The capacity of 25CN-NBOH to attenuate DOI-elicited head twitch was also investigated. Mice were trained to discriminate DOI or M100907 from saline, and 25CN-NBOH was tested for generalization.
Results
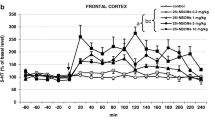
25CN-NBOH induced a head twitch response in the mouse that was lower in magnitude than that of DOI, blocked by M100907, but not altered by RS102221. DOI-elicited head twitch was dose-dependently attenuated by 25CN-NBOH pretreatment. 25CN-NBOH produced an intermediate degree of generalization (55 %) for the DOI training dose, and these interoceptive effects were attenuated by M100907. Finally, 25CN-NBOH did not generalize to M100907 at any dose, but ketanserin fully substituted in these animals.
Conclusions
25CN-NBOH was behaviorally active, but less effective than DOI in two mouse models of hallucinogenic effects. The effectiveness with which M100907 antagonized the behavioral actions of 25CN-NBOH strongly suggests that the 5-HT2A receptor is an important site of agonist action for this compound in vivo.





Similar content being viewed by others
References
Advisory Council on the Misuse of Drugs (ACMD) (2013) “NBOMe” compounds: A review of the evidence of use and harm. Available at: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/20480 8/J_TCDO_report_on_5-6APB_and_NBOMe_compounds.pdf. Accessed 03 July 2014
Blaazer AR, Smid P, Kruse CG (2008) Structure-activity relationships of phenylalkylamines as agonist ligands for 5-HT(2A) receptors. Chem Med Chem 3(9):1299–1309
Braden MR, Parrish JC, Naylor JC, Nichols DE (2006) Molecular interaction of serotonin 5-HT2A receptor residues Phe339(6.51) and Phe340(6.52) with superpotent N-benzyl phenethylamine agonists. Mol Pharmacol 70(6):1956–1964
Canal CE, Morgan D (2012) Head-twitch response in rodents induced by the hallucinogen 2,5-dimethoxy-4-iodoamphetamine: a comprehensive history, a re-evaluation of mechanisms, and its utility as a model. Drug Test Anal 4(7–8):556–576
Dekeyne A, Iob L, Hautefaye P and Millan MJ (2002) The selective serotonin(2A) receptor antagonist, MDL100,907, elicits a specific interoceptive cue in rats. Neuropsychopharmacology 26(4):552–6
Fantegrossi WE, Murnane KS, Reissig CJ (2008) The behavioral pharmacology of hallucinogens. Biochem Pharmacol 75(1):17–33
Fantegrossi WE, Simoneau J, Cohen MS, Zimmerman SM, Henson CM, Rice KC, Woods JH (2010) Interaction of 5-HT2A and 5-HT2C receptors in R(-)-2,5-dimethoxy-4-iodoamphetamine-elicited head twitch behavior in mice. J Pharmacol Exp Ther 335(3):728–734
Glennon RA, Raghupathi RK, Bartyzel P, Teitler M, Leonhardt S (1992) Binding of phenylalkylamine derivatives at 5-HT1C and 5-HT2 serotonin receptors: evidence for a lack of selectivity. J Med Chem 35:734–740
Halberstadt AL, Geyer MA (2014) Effects of the hallucinogen 2,5-dimethoxy-4- iodophenethylamine (2C-I) and superpotent N-benzyl derivatives on the head twitch response. Neuropharmacol 77:200–207
Hansen M (2011) Design and Synthesis of Selective Serotonin Receptor Agonists for Positron Emission Tomography Imaging of the Brain. Ph.D. thesis, University of Copenhagen. Accessed at https://docs.google.com/file/d/0BwXelgjm5BeEaEJJU0lPa1NnaGM, 01 July 2014
Hansen M, Phonekeo K, Paine JS, Leth-Petersen S, Begtrup M, Bräuner-Osborne H, Kristensen JL (2014) Synthesis and structure-activity relationships of N-benzyl phenethylamines as 5-HT2A/2C agonists. ACS Chem Neurosci 5(3):243–249
Juncosa JI Jr, Hansen M, Bonner LA, Cueva JP, Maglathlin R, McCorvy JD, Marona-Lewicka D, Lill MA, Nichols DE (2013) Extensive rigid analogue design maps the binding conformation of potent N-benzylphenethylamine 5-HT2A serotonin receptor agonist ligands. ACS Chem Neurosci 4(1):96–109
Knight AR, Misra A, Quirk K, Benwell K, Revell D, Kennett G, Bickerdike M (2004) Pharmacological characterisation of the agonist radioligand binding site of 5-HT(2A), 5-HT(2B) and 5-HT(2C) receptors. Naunyn Schmiedebergs Arch Pharmacol 370(2):114–123
Lawn W, Barratt M, Williams M, Horne A and Winstock A (2014) The NBOMe hallucinogenic drug series: Patterns of use, characteristics of users and self-reported effects in a large international sample. J Psychopharmacol, in press.
Leth-Petersen S, Bundgaard C, Hansen M, Carnerup MA, Kehler J and Kristensen JL (2014) Correlating the Metabolic Stability of Psychedelic 5-HT2A Agonists with Anecdotal Reports of Human Oral Bioavailability. Neurochem Res, in press.
Li JX, Unzeitig A, Javors MA, Rice KC, Koek W, France CP (2009) Discriminative stimulus effects of 1-(2,5-dimethoxy-4-methylphenyl)-2-aminopropane (DOM), ketanserin, and (R)-(+)-{alpha}-(2,3-dimethoxyphenyl)-1-[2-(4-fluorophenyl)ethyl]-4-pipidinemethanol (MDL100907) in rats. J Pharmacol Exp Ther 331(2):671–679
McLean TH, Parrish JC, Braden MR, Marona-Lewicka D, Gallardo-Godoy A, Nichols DE (2006) 1-Aminomethylbenzocycloalkanes: conformationally restricted hallucinogenic phenethylamine analogues as functionally selective 5-HT2A receptor agonists. J Med Chem 49(19):5794–5803
Nichols DE (2004) Hallucinogens. Pharmacol Ther 101(2):131–181
Nichols DE, Frescas SP, Chemel BR, Rehder KS, Zhong D, Lewin AH (2008) High specific activity tritium-labeled N-(2-methoxybenzyl)-2,5-dimethoxy-4 -iodophenethylamine (INBMeO): a high-affinity 5-HT2A receptor-selective agonist radioligand. Bioorg Med Chem 16:6116–6123
Nikolaou P, Papoutsis I, Stefanidou M, Spiliopoulou C and Athanaselis S (2014) 2C-I-NBOMe, an "N-bomb" that kills with "Smiles". Toxicological and legislative aspects. Drug Chem Toxicol, in press.
Parker MA, Marona-Lewicka D, Lucaites VL, Nelson DL, Nichols DE (1998) A novel (benzodifuranyl)aminoalkane with extremely potent activity at the 5-HT2A receptor. J Med Chem 41(26):5148–5149
Roth BL, Willins DL, Kristiansen K, Kroeze WK (1998) 5-Hydroxytryptamine2-family receptors (5-hydroxytryptamine2A, 5-hydroxytryptamine2B, 5-hydroxytryptamine2C): where structure meets function. Pharmacol Ther 79(3):231–257
Tang MH, Ching CK, Tsui MS, Chu FK, Mak TW (2014) Two cases of severe intoxication associated with analytically confirmed use of the novel psychoactive substances 25B-NBOMe and 25C-NBOMe. Clin Toxicol 52(5):561–565
Walterscheid JP, Phillips GT, Lopez AE, Gonsoulin ML, Chen HH, Sanchez LA (2014) Pathological findings in 2 cases of fatal 25I-NBOMe toxicity. Am J Forensic Med Pathol 35(1):20–25
Acknowledgements
We thank the UAMS Division of Laboratory Animal Medicine for expert husbandry services. This research was generously supported, in part, by an NIGMS IDeA Program award (GM110702), by the UAMS Translational Research Institute (RR029884), and by the Lundbeck Foundation. DS received a Summer Undergraduate Research Fellowship from the American Society of Pharmacology and Experimental Therapeutics which funded his time in the laboratory conducting the head twitch experiments described herein. The views expressed herein are those of the authors and do not necessarily represent the views of the University of Arkansas for Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fantegrossi, W.E., Gray, B.W., Bailey, J.M. et al. Hallucinogen-like effects of 2-([2-(4-cyano-2,5-dimethoxyphenyl) ethylamino]methyl)phenol (25CN-NBOH), a novel N-benzylphenethylamine with 100-fold selectivity for 5-HT2A receptors, in mice. Psychopharmacology 232, 1039–1047 (2015). https://doi.org/10.1007/s00213-014-3739-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3739-3