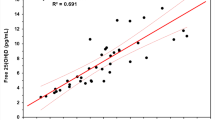
Abstract
Vitamin D3 and its metabolites are lipophilic molecules with low aqueous solubility and must be transported bound to plasma carrier proteins, primarily to vitamin D binding protein (DBP). The biological functions of vitamin D3 metabolites are intimately dependent on the protein, hence the importance of determining their affinity for DBP. In this study, we developed a novel procedure for measuring the kinetic and equilibrium constants of human-DBP with vitamin D3 and three metabolites: 1,25-dihydroxyvitamin D3 [1,25(OH)2D3], 25-hydroxyvitamin D3 (25OHD3) and 24,25-dihydroxyvitamin D3 [24,25(OH)2D3] by surface plasmon resonance (SPR). At the same time, five different analogues, synthetized in our laboratory, were evaluated in order to compare the affinity values with the metabolites. Real-time SPR measurements showed that 25OHD3 and 24,25(OH)2D3 had higher affinity (0.3 μM) than 1,25(OH)2D3 (5 μM), with the higher affinity of 25OHD3 and 24,25(OH)2D3 due to dissociation constants 1 order of magnitude slower. In the case of the analogues, the affinity values were lower, with 1-hydroxy-25-nitro-vitamin D3 (NO2-446), structurally closer to 1,25(OH)2D3, showing the highest value with a K D of 50 μM. (24R)-1,25-dihydroxyvitamin-24-buthyl-28-norvitamin D2 (Bu-471) and (24R)-1,25-dihydroxyvitamin-24-phenyl-28-norvitamin D2 (Ph-491), structurally similar, had affinities of 180 and 170 μM, respectively. (22R,23E)-1-hydroxy-22-ethenyl-25-methoxy-23-dehydrovitamin D3 (MeO-455) and 1-hydroxy-20(R)-[5(S)-(2,2-dimethyltetrahydropyran-5-yl)]-22,23-dinor vitamin D3 (Oxan-429) had affinities of 310 and 100 μM, respectively. The binding of the metabolites and analogues was reversible allowing the rapid capture of data for replicates. The kinetic and equilibrium data for both the metabolites and the analogues fitted to the Langmuir model describing a 1:1 interaction.

Label-free, real time binding study between vitamin D binding protein immmobilized on the surface of a SPR sensor chip and the vitamin D, metabolites and analogues passed over it as analytes




Similar content being viewed by others
References
Bikle DD, Gee E, Halloran B, Kowalski MA, Ryzen E, Haddad JG. Assessment of the free fraction of 25-hydroxyvitamin D in serum and its regulation by albumin and the vitamin D-binding protein. J Clin Endocrinol Metab. 1986;63:954–9.
Bikle DD, Siiteri PK, Ryzen E, Haddad JG. Serum protein binding of 1,25-dihydroxyvitamin D: a reevaluation by direct measurement of free metabolite levels. J Clin Endocrinol Metab. 1985;61:969–75.
Gomme PT, Bertolini J. Therapeutic potential of vitamin D-binding protein. Trends Biotechnol. 2004;22:340–5.
Schmid A, Walther B. Natural vitamin D content in animal products. Adv Nutr. 2013;4:453–6.
Sintov AC, Yarmolinsky L, Dahan A, Ben-Shabat S. Pharmacological effects of vitamin D and its analogs: recent developments. Drug Discov Today. 2014;19:1769–74.
Swamy N, Head JF, Weitz D, Ray R. Biochemical and preliminary crystallographic characterization of the vitamin D sterol-and actin-binding by human vitamin D-binding protein. Arch Biochem Biophys. 2002;402:14–23.
Verboven C, Rabijns A, De Maeyer M, Van Baelen H, Bouillon R, De Ranter C. A structural basis for the unique binding features of the human vitamin D-binding protein. Nat Struct Biol. 2002;9:131–6.
Yamamoto K. Vitamin D, and related proteins, CYP, DBP and VDR; from the viewpoint of structural life science. Vitamins. 2013;87:669–77.
Swamy N, Dutta A, Ray R. Roles of the structure and orientation of ligands and ligand mimics inside the ligand-binding pocket of the vitamin D-binding protein. Biochemistry. 1997;36:7432–6.
White P, Cooke N. The multifunctional properties and characteristics of vitamin D-binding protein. Trends Endocrinol Metab. 2000;11:320–7.
Safadi FF, Thornton P, Magiera H, Hollis BW, Gentile M, Haddad JG, et al. Osteopathy and resistance to vitamin D toxicity in mice null for vitamin D binding protein. J Clin Invest. 1999;103:239–51.
Ray R. Molecular recognition in vitamin D-binding protein. Proc Soc Exp Biol Med. 1996;212:305–12.
Verboven CC, De Bondt HL, De Ranter C, Bouillon R, Van Baelen H. Crystallization and X-ray investigation of vitamin D-binding protein from human serum. Identification of the crystal content. J Steroid Biochem Mol Biol. 1995;54:11–4.
Youssef DA, Miller CWT, El-Abbassi AM, Cutchins DC, Cutchins C, Grant WB, et al. Antimicrobial implications of vitamin D. Derm Endocrinol. 2011;3:220–9.
Bouillon R, Okamura WH, Norman AW. Structure-function relationships in the vitamin D endocrine system. Endocr Rev. 1995;16:200–57.
Leyssens C, Verlinden L, Verstuyf A. The future of vitamin D analogs. Genome-wide view physiol vitamin D. 2014;5:122.
Jones G, Vitamin D. Analogs. Rheum Dis Clin N Am. 2012;38:207–32.
Sherman M, Yu R, Engle D, Ding N, Atkins A, Tiriac H, et al. Vitamin D receptor-mediated stromal reprogramming suppresses pancreatitis and enhances pancreatic cancer therapy. Cell. 2014;159:80–93.
Gopinath SCB. Biosensing applications of surface plasmon resonance-based Biacore technology. Sensors Actuators B Chem. 2010;50:722–33.
Liu X, Sun Y, Song D, Zhang Q, Tian Y, Zhang H. Enhanced optical immuosensor based on surface plasmon resonance for determination of transferrin. Talanta. 2006;68:1026–31.
Olaru A, Bala C, Jaffrezic-Renault N, Aboul-Enein HY. Surface plasmon resonance (SPR) biosensors in pharmaceutical analysis. Crit Rev Anal Chem. 2015;45:97–105.
Xia X, Zun-Zhong Y, Jian W, Yi-Bin Y. Application and research development of surface plasmon resonance-based immunosensors for protein detection. Fenxi Huaxue Chin J Anal Chem. 2010;38:1052–9.
Huber W, Mueller F. Biomolecular interaction analysis in drug discovery using surface plasmon resonance technology. Curr Pharm Des. 2006;12:3999–4021.
Karlsson R. SPR for molecular interaction analysis: a review of emerging application areas. J Mol Recogn. 2004;17:151–161
Homola J. Surface plasmon resonance sensors for detection of chemical and biological species. Chem Rev. 2008;108:462–93.
Nguyen HH, Park J, Kang S, Kim M. Surface plasmon resonance: a versatile technique for biosensor applications. Sensors. 2015;15:10481–510.
Esaki K. Methods for detecting binding of low-molecular-weight compound and its binding partner molecule. 2004.
Pritchard D, Preston L, Lawlor, M. Binding molecule for DPB and method for measuring vitamin D. 2012.
Carlucci L, Favero G, Tortolini C, Di Fusco M, Romagnoli E, Minisola S, et al. Several approaches for vitamin D determination by surface plasmon resonance and electrochemical affinity biosensors. Biosens Bioelectron. 2015;40:350–5.
Pasquali M, Tartaglione L, Rotondi S, Leonangeli C, Mazzaferro S. Further vitamin D analogs. Curr Vasc Pharmacol. 2014;12:329–38.
Fall Y, Vitale C, Mouriño A. An efficient synthesis of the 25-hydroxy Windaus–Grundmann ketone. Tetrahedron Lett. 2000;41:7337–40.
Torneiro M, Fall Y, Castedo L, Mouriño A. A short, efficient copper-mediated synthesis of 1α, 25-dihydroxyvitamin D2 (1α, 25-dihydroxyergocalciferol) and C-24 analogs1, 2. J Org Chem. 1997;62:6344–52.
Rivadulla ML, Pérez-García X, Pérez M, Gómez G, Fall Y. Pd-allylic substitution mediated synthesis of 25-amino vitamin D3 derivatives. Tetrahedron Lett. 2013;54:3164–6.
Fall Y, Fernandez C, Vitale C, Mouriño A. Stereoselective synthesis of vitamin D3 analogues with cyclic side chains. Tetrahedron Lett. 2000;41:7323–6.
Cohavi O, Tobi D, Schreiber G. Docking of antizyme to ornithine decarboxylase and antizyme inhibitor using experimental mutant and double-mutant cycle data. J Mol Biol. 2009;390:503–15.
Bravman T, Bronner V, Lavie K, Notcovich A, Papalia GA, Myszka DG. Exploring “one-shot” kinetics and small molecule analysis using the ProteOn XPR36 array biosensor. Anal Biochem. 2006;358:281–8.
Jones G. Pharmacokinetics of vitamin D toxicity. Am J Clin Nutr. 2008;88:582S–6S.
Chun RF, Peercy BE, Orwoll ES, Nielson CM, Adams JS, Hewison M. Vitamin D and DBP: the free hormone hypothesis revisited. J Steroid Biochem Mol Biol. 2014;144, Part A:132-137.
Teegarden D, Meredith SC, Sitrin MD. Determination of the affinity of vitamin D metabolites to serum vitamin D binding protein using assay employing lipid-coated polystyrene beads. Anal Biochem. 1991;199:293–7.
Shankaran DR, Gobi KV, Miura N. Recent advancements in surface plasmon resonance immunosensors for detection of small molecules of biomedical, food and environmental interest. Sensors Actuators B Chem. 2007;121:158–77.
Nilsson SF, Östberg L, Peterson PA. Binding of vitamin D to its human carrier plasma protein. Biochem Biophys Res Commun. 1972;46:1380–7.
Kawakami M, Imawari M, Goodman DS. Quantative studies of the interactions of cholecalciferol (vitamin D3) and its metabolites with different genetic variants of the serum binding protein for these sterols. Biochem J. 1979;179:413–23.
Haddad Jr JG, Walgate J. 25 Hydroxyvitamin D transport in human plasma. Isolation and partial characterization of calcifidiol binding protein. J Biol Chem. 1976;251:4803–9.
Vieth R. Simple method for determining specific binding capacity of vitamin D-binding protein and its use to calculate the concentration of “free” 1,25-dihydroxyvitamin D. Clin Chem. 1994;40:435–41.
van Etten E, Decallonne B, Verlinden L, Verstuyf A, Bouillon R, Mathieu C. Analogs of 1α, 25‐dihydroxyvitamin D3 as pluripotent immunomodulators. J Cell Biochem. 2003;88:223–6.
Acknowledgements
We are grateful to Dishman Netherlands BV for the gift of some starting materials. Pilar Canoa acknowledge the financial support of Celta Ingenieros, S.L. We would also like to thank the following organizations and individuals: the proteomic service of CACTI, University of Vigo, for SPR facilities and Carlos Eugenio Pérez Lara for his help with the preparation of Fig. 4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding source
This work was financially supported by the Xunta de Galicia (CN 2012/184).
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 458 kb)
Rights and permissions
About this article
Cite this article
Canoa, P., Rivadulla, M.L., Popplewell, J. et al. Use of surface plasmon resonance in the binding study of vitamin D, metabolites and analogues with vitamin D binding protein. Anal Bioanal Chem 409, 2547–2558 (2017). https://doi.org/10.1007/s00216-017-0200-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-017-0200-6