Abstract
Background
Whole-body magnetic resonance imaging (WB-MRI) is an increasingly used guideline-based imaging modality for oncological and non-oncological pathologies during childhood and adolescence. While diffusion-weighted imaging (DWI), a part of WB-MRI, enhances image interpretation and improves sensitivity, it also requires the longest acquisition time during a typical WB-MRI scan protocol. Interleaved short tau inversion recovery (STIR) DWI with simultaneous multi-slice (SMS) acquisition is an effective way to speed up examinations.
Objective
In this study of children and adolescents, we compared the acquisition time, image quality, signal-to-noise ratio (SNR) and apparent diffusion coefficient (ADC) values of an interleaved STIR SMS-DWI sequence with a standard non-accelerated DWI sequence for WB-MRI.
Materials and methods
Twenty children and adolescents (mean age: 13.9 years) who received two WB-MRI scans at a maximum interval of 18 months, consisting of either standard DWI or SMS-DWI MRI, respectively, were included. For quantitative evaluation, the signal-to-noise ratio (SNR) was determined for b800 images and ADC maps of seven anatomical regions. Image quality evaluation was independently performed by two experienced paediatric radiologists using a 5-point Likert scale. The measurement time per slice stack, pause between measurements including shim and total measurement time of DWI for standard DWI and SMS-DWI were extracted directly from the scan data.
Results
When including the shim duration, the acquisition time for SMS-DWI was 43% faster than for standard DWI. Qualitatively, the scores of SMS-DWI were higher in six locations in the b800 images and four locations in the ADC maps. There was substantial agreement between both readers, with a Cohen’s kappa of 0.75. Quantitatively, the SNR in the b800 images and the ADC maps did not differ significantly from one another.
Conclusion
Whole body-MRI with SMS-DWI provided equivalent image quality and reduced the acquisition time almost by half compared to the standard WB-DWI protocol.
Similar content being viewed by others
Introduction
Whole-body magnetic resonance imaging (WB-MRI) is an imaging technique for visualization of the entire body and is used primarily in childhood and adolescence to avoid other imaging modalities that use ionizing radiation [1]. The national German S1 guideline “Whole Body Magnetic Resonance Imaging in Childhood and Adolescence,” which was most recently modified in 2021, highlights the diagnostic value of WB-MRI in this age range [2]. The Taskforce Oncology Guidelines of the European Society for Paediatric Radiology confirm their significance on a global scale [3]. WB-MRI is highly valuable in the diagnosis of oncological conditions such as Hodgkin’s and non-Hodgkin’s lymphomas [4], the diagnosis of distant disease in osteo-, Ewing and soft tissue sarcomas [5] and in Langerhans cell histiocytosis [6] and for the monitoring of tumour predisposition syndromes [7]. Additionally, WB-MRI is performed for radiation-free diagnosis in infancy and adolescence of non-oncological disorders such as chronic non-bacterial osteomyelitis and chronic recurrent multifocal osteomyelitis [8, 9]. Other important indications include fever of unknown origin [10] and complications of sickle cell disease [11].
Currently, there is no standard protocol for whole-body imaging [12]. However, WB-MRI with diffusion-weighted imaging (DWI) has been shown to increase diagnostic accuracy in cases of residual lymphoma in adults [13]. Diffustion-weighted imaging in WB-MRI can provide additional information on diffusivity, highlighting changes in the cell density of different tissues, in addition to features seen on standard sequences such as short tau inversion recovery (STIR) and T1 contrast. [14]. This can be helpful in oncological diagnostics for initial staging, treatment response and follow-up monitoring [15]. A particular problem with diffusion-weighted sequences in WB-MRI is the long acquisition time. Younger children have difficulty lying motionless for long periods and older (larger) children may require more slice stacks to achieve whole-body coverage. Diffusion-weighted imaging alone can require more than 20 min to cover the entire body depending on the size of the patient and the chosen slice thickness. Diffusion-weighted imaging with a quicker acquisition time would, therefore, be a clear benefit in paediatric WB-MRI.
One possibility of sequence acceleration is the simultaneous excitation and readout of multiple slices (SMS) by exploiting the different spatial sensitivity profiles of multi-coil arrays [16, 17]. This technique has already been established for MRI [18], and its advantages have been outlined in various studies on different organ systems, such as the liver [19], breast [20] and skeletal system [21], but mainly in adult patients. Kenkel et al. showed both a drastic reduction in the duration of whole-body MRI using SMS-DWI and its clinical applicability in adults [22]. Tabari et al. and, more recently, Glutig et al. demonstrated the benefit of simultaneous multi-slice abdominal MRI during childhood and adolescence using the kidney as an example in patients with cystic fibrosis and tuberous sclerosis complex [23, 24]. The use of an efficient interleaving scheme for WB STIR DWI imaging [2, 25] represents another method of reducing the acquisition time of WB-MRI.
This study aimed to investigate the feasibility and clinical utility of an interleaved WB-STIR DWI-SMS sequence in childhood and adolescence.
Materials and methods
Study design
All children with a clinical indication for WB-MRI who underwent at least two whole-body examinations between January 2020 and July 2021 were included in the study. The indications for recurrent WB-MRI included oncologic and non-oncologic follow-up and screening for tumour predisposition syndromes. The interval between the current WB-MRI and the earlier examination was 18 months or less. The follow-up whole-body examination used a vendor-supplied “works in progress” (WIP) simultaneous multi-slice diffusion-weighted sequence (SMS-DWI). For reasons of compliance, only one WB-DWI investigation was performed on a single date. This study was approved by the local ethics committee (Reg. No. 2022–2600). All patients or their legal guardians provided written informed consent for the examination.
Patient measurements
All MRI examinations were performed on a clinical 1.5-tesla (T) whole body MRI system (MAGNETOM Sola, Siemens Healthineers, Erlangen, Germany) using the following vendor-supplied clinical coils for WB-MRI: 20-channel cranial-neck coil, 12-channel BioMatrix body coil, 18-channel body coil, 36-channel peripheral angio coil, and 32-channel spine coil. The patients underwent a WB-MRI examination consisting of a coronal short tau inversion recovery turbo spin echo (STIR TSE), a coronal T1 SPACE (sampling perfection with application optimized contrast using different flip angle evolution) and an axial whole-body diffusion-weighted sequence. During the first WB-MRI, a standard DWI (sDWI) was used; in the follow-up scan, DWI was performed using the prototype SMS technique with enabled interleaved STIR module (SMS-DWI). All DWI sequences used an echo-planar imaging (EPI) readout. The acquisition parameters for both sequences are listed in Table 1. Except for repetition duration (TR), which could be shortened by the SMS approach, the parameters of the diffusion-weighted sequences were the same. All sequences were acquired while breathing freely. Depending on patient size, only the number of slice stacks of DWI was adjusted as necessary, while keeping the number of slice stacks constant between the two scans performed for each patient.
Assessment of acquisition time
To compare the acquisition times of sDWI and SMS-DWI, the digital imaging and communications in medicine (DICOM) tags of all the acquired imaging series were analyzed retrospectively. For this purpose, the start time and sequence duration of each layer stack were extracted, whereas the start time of the subsequent layer stack was subtracted from those of its predecessor. From this information, the total time for the acquisition of a one-layer stack, including all scan adjustments and shimming, as well as the duration of only the sDWI and SMS-DWI sequences, were extracted.
Assessment of image quality
Qualitative evaluation was performed using Mint-Lesion (MINT Medical GmbH, Heidelberg, Germany) by two paediatric radiologists, one with 10 years (P.C.K.) and the other with 15 years (K.G.) of paediatric imaging experience. Using a 5-point Likert scale (1 = very good, 2 = good, 3 = adequate, 4 = poor, 5 = insufficient), the image quality assessment of b800 and the ADC parameter maps of the sDWI and SMS-DWI were performed independently. The readers were blinded to the patient data and sequences used.
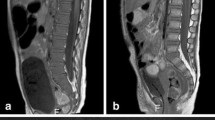
Image quality was assessed for each of the following locationst: brain, chest, trunk and extremities for each patient (Figs. 1 and 2). Particular attention to distortion-free imaging and artifacts was noted as well as general image quality.
Whole-body-magnetic resonance scan in a 16-year-old girl with fever of unknown origin. Comparison of axial orientated diffusion-weighted examinations at b800 for brain (a,b), chest (c,d), upper abdomen (e,f), kidneys (g,h), pelvis (i,j) and thigh muscles (k,l). a,c,e,g,i,k Standard diffusion-weighted imaging (DWI). b,d,f,h,j,l Simultaneous multi-slice DWI. Note small field inhomogeneities of the subcutaneous fat of the limb, probably due to limited compatibility of the used coils with the simultaneous multi-slice technique (arrows in l)
Comparison of axial orientated ADC (apparent diffusion coefficient) parameter map for brain (a,b), chest (c,d), upper abdomen (e,f), kidneys (g,h), pelvis (i,j) and thigh muscles (k,l) for the same 16-year-old girl shown in Fig. 1 with fever of unknown origin. a,c,e,g,i,k Standard diffusion-weighted imaging (DWI), b,d,f,h,j,l Simultaneous multi-slice-DWI. Overall comparable image quality
Quantitative assessment of signal-to-noise ratio and apparent diffusion coefficient
For quantitative evaluation, regions of interest (ROI) were placed in seven different areas, both at b = 800 s/mm2 and on the ADC parameter map. The areas were chosen according to the respective regions and included the white matter, cerebrospinal fluid (CSF), mediastinum, liver, spleen, kidneys and thigh muscles. The sizes of the ROI were adjusted for the target localization. The Mint-Lesion software ensured the same size and location of ROI for different b800 and ADC images. For each ROI, the mean value, standard deviation (SD) and maximum value were automatically acquired via the Mint Lesion, and the SNR was calculated for b800.
Statistical analysis
Statistical analysis was performed using Python programming language (Python Software Foundation, https://python.org/) and statsmodels [26, 27].
Ratings, ADC and SNR were recorded as mean and SD. Descriptive statistics were used to summarize the population characteristics and image findings. Scores for the image quality of sDWI and SMS-DWI were compared using the Wilcoxon signed-rank test. Interrater agreement was determined using Cohen’s kappa (correlation: < 0.2 poor; 0.2–0.4 fair; 0.4–0.6 moderate; 0.6–0.8 substantial; > 0.8 almost perfect).
Results
Clinical characteristics
A total of twenty children and adolescents (9 male) were included in the main study. The mean age was 13.9 years ± 5.4 years (range 5–18 years). Table 2 shows the demographic data of the patients divided into different diagnostic groups.
Measuring times
The sDWI took an average of 3 min 56 s ± 12 s per stack, of which 1 min 31 s ± 12 s was used for the shim and 2 min 25 s was used for acquisition. The average time for SMS-DWI was 2 min 15 s ± 6 s for each stack, which was broken down into 1 min 28 s for acquisition and 47 s ± 6 s for the shim. As a result, the overall measurement time for a patient undergoing the acquisition of six-layer stacks was 13 min 29 s ± 0:35 min for SMS-DWI and 23 min 38s ± 01:15 min for sDWI.
Using the SMS-DWI resulted in a 43% reduction in examination time and a reduction of 39% in sequence acquisition time. Table 3 provides an overview of the examination times for patients depending on the two different diffusion-weighted sequences used for WB-MRI.
Qualitative analysis–subjective image quality
Interobserver agreement–interrater variability–Cohen’s kappa
On average the Cohen’s kappa of both readers was substantial (0.75). There was substantial agreement with respect to the image quality (IQ) of the DWI b800 images and almost perfect agreement with respect to the ADC. Further details are provided in Table 4.
Mean ratings
Both sequences showed subjectively good and comparable image quality for diffusion weightings as well as for the ADC parameter maps. The mean ratings of the sDWI were slightly lower than those of the SMS-DWI. Overall, the SMS-DWI was preferred in six out of seven ratings for the b800 image and in four out of seven ratings for the ADC, with significantly better ratings for the white matter in the brain (b800 and ADC), liver (b800), CSF (ADC) and kidney (b800 and ADC). Significantly lower ratings of SMS-DWI were observed only for the limb muscles in the b800 images. Table 5 provides an overview of the scores for the DWI with b800 and Table 6 shows the scores for the ADC parameter values.
For a better overview, the combined DWI stacks were reconstructed as a composite 3-dimensional (D) dataset and displayed using an inverted grayscale colormap. Qualitative evaluation showed that the SMS-DWI data exhibited noticeable step artifacts in the reconstruction along the slice direction. Figure 3 shows a comparison of the 3-D composite reconstruction of a colormap inverted b800 image from a 15-year-old girl with tumour predisposition syndrome.
Comparison of the coronal 3-dimensional reconstructions for standard diffusion-weighted imaging (sDWI) (a) and simultaneous multi-slice diffusion-weighted imaging (SMS-DWI) (b) in a 15-year-old girl with cancer preposition syndrome. Both measurements with 6 stacks of DWI inline composed on the scanner. There are visible step formations in the SMS-DWI due to the different shimming algorithm without slice adjustments (black arrow). Overall, however, there is comparable quality with a homogeneous signal
Quantitative analysis
The SNR values of sDWI and SMS-DWI at b800 showed no significant differences for all the assessed regions (white matter, CSF, chest, liver, kidney, and thigh muscle). Figure 4 demonstrates the SNR values for b800 for the assessed regions as a boxplot.
Boxplot of signal to noise ratio (SNR) values measured in b800 in 20 patients comparing sDWI und SMS-DWI. There is no significant difference in the measured b800 values in the regions studied. CSF cerebrospinal fluid; sDWI standard diffusion weighted image; SMS-DWI simultaneous multi-slice DWI; WM white matter
There were no significant differences in the recorded ADC in the investigated regions, except for the right kidney, which showed significantly higher values on the SMS-DWI. Figure 5 presents the ADC of the assessed regions, comparing sDWI and SMS-DWI.
Boxplot of ADC values measured in 20 patients comparing sDWI und SMS-DWI. There is no significant difference in the measured ADC values in the regions studied. ADC apparent diffusion coefficient; CSF cerebrospinal fluid; sDWI standard diffusion weighted image; SMS-DWI simultaneous multi-slice DWI; WM white matter
An overview of the measured ADC for sDWI and SMS-DWI and a comparison with published values is given in Table 7.
Discussion
Analysis of the results demonstrates that for WB-MRI, the STIR-SMS diffusion sequence (SMS-DWI) reduces the examination time by an average of 43% compared with our previously used standard diffusion sequence without SMS acceleration. A faster examination time was achieved by combining a modern interleaved scheme in the SMS technique with an SMS factor of 2, in addition to parallel imaging with a GRAPPA acceleration factor of 2, which was used by both sequences. Image quality, SNR and ADC values were comparable to those of standard DWI.
The acceleration of the examination time achieved was remarkable compared to other studies in adults. Kenkel et al. showed a 24.2–25.9% reduction in examination time using a slice-acceleration factor of 3 with the same image quality and ADC values [22]. Currently, we are not aware of any work that has systematically investigated the duration of WB-DWI measurements during childhood and adolescence.
The SMS technique allows the simultaneous excitation of slices and thus a significant acceleration in data acquisition time depending on the number of excited slices [16]. Compared to conventional parallel imaging, there is only a slight potential SNR penalty and minor effects on TE when using SMS. Setsompop et al. demonstrated the application of the SMS technique as one of the first for diffusion-weighted imaging techniques [28]. Taron et al. investigated the SMS-DWI for whole-body positron-emission tomography/MRI with a reduction in scan time of 40% [29] and similarly for abdominal MRI [19, 30]. They were able to show a substantial reduction in liver DWI scan time (70%), with comparable image quality. Recently, regarding abdominal MRI in children and adolescents with cystic fibrosis, Glutig et al. showed a 32% reduction in examination time with no decrease in SNR using SMS-DWI and improved image quality by additional reconstruction with motion-correction [24]. In this work, the combination of STIR-DWI and the SMS technique for whole-body imaging was used for the first time.
In addition to the intrinsic sequence acquisition time, other factors such as the number of layer stacks and the time spent shimming before each layer stack must be considered when analyzing the total examination time of the DWI sequence. In this study, the total measurement time was significantly reduced by decreasing the sequence-only measurement time by 39% and the shim time between layer stacks by 46%. The shorter shim time results from the fact that the SMS-DWI sequence is based on a simple volume shim and is not compatible with the whole-body shim “SliceAdjust” [31] of the MRI device manufacturer, as is the case with the standard DWI sequence. As a result of the more simplified shim algorithm, typical inhomogeneities and image artefacts, such as “broken spine artefacts”, may occur more frequently [31].
The degree of interobserver agreement in image quality evaluations showed that the ratings in our study were quite trustworthy. Our quantitative analysis did not reveal any appreciable variations in the SNR of b800 images. In this study, the ADC values showed no significant differences in six of seven localizations analyzed. This is in agreement with the results of Glutig et al. [24] and Xu et al. [32, 33], but contrasts with the data of Taron et al. [19], who measured lower ADC values.
This study has some limitations. First, only a small number of patients were identified. Second, it was not a homogeneous group of patients with the same disease, and we did not examine focal or specific pathologies, but only compared the examination time, general image quality, SNR and ADC values. Furthermore, this was a retrospective study from a single centre, which may have introduced some bias. All patients included in the study had already received a WB-MRI examination with the standard DWI. Because of the standardization of the examinations, we could use these data for the study and no patient had to receive an MRI examination solely for the study. The maximum interval between the current SMS-DWI and the sDWI was 18 months; a lot can happen in those 18 months, especially in oncology patients.
Large tumours or lymphadenopathy on initial or follow-up MRI may affect image quality. However, this was not the case in the included patients; disease-related findings were only detected in one examination.
This study was performed as part of routine clinical surveillance imaging. In future research, we aim to take practical steps, such as applying artificial intelligence and deep learning, to address deficiencies and to broaden the results of this study.
Conclusion
Whole body-MRI with interleaved STIR SMS-DWI provided equivalent image quality and significantly reduced acquisition time, which is important in paediatric patients and should therefore replace the previous sDWI sequence.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Darge K, Jaramillo D, Siegel MJ (2008) Whole-body MRI in children: current status and future applications. Eur J Radiol 68:289–298
Schaefer JF, Berthold LD, Hahn G et al (2019) Whole-body MRI in children and adolescents - S1 guideline. Rofo 191:618–625
Schafer JF, Granata C, von Kalle T et al (2020) Whole-body magnetic resonance imaging in pediatric oncology - recommendations by the Oncology Task Force of the ESPR. Pediatr Radiol 50:1162–1174
Kwee TC, Takahara T, Vermoolen MA et al (2010) Whole-body diffusion-weighted imaging for staging malignant lymphoma in children. Pediatr Radiol 40:1592–1602
Schaefer JF, Schlemmer HP (2006) Total-body MR-imaging in oncology. Eur Radiol 16:2000–2015
Steinborn M, Wortler K, Nathrath M et al (2008) Whole-body MRI in children with Langerhans cell histiocytosis for the evaluation of the skeletal system. Rofo 180:646–653
Glutig K, Pfeil A, Renz DM (2021) Imaging of tumor predisposition syndromes. Radiologe 61:658–666
Zadig P, von Brandis E, Lein RK et al (2021) Whole-body magnetic resonance imaging in children - how and why? A systematic review. Pediatr Radiol 51:14–24
Menashe SJ, Iyer RS, Ngo AV et al (2022) Whole-body MRI at initial presentation of pediatric chronic recurrent multifocal osteomyelitis and correlation with clinical assessment. Pediatr Radiol 52:2377–2387
Tavakoli AA, Reichert M, Blank T et al (2020) Findings in whole body MRI and conventional imaging in patients with fever of unknown origin-a retrospective study. BMC Med Imaging 20:94
Khedr SA, Hassaan MA, Shabana AA et al (2012) Musculoskeletal manifestations of sickle cell disease, diagnosis with whole body MRI. Egypt J Radiol Nucl Med 43:77–84
Eutsler EP, Khanna G (2016) Whole-body magnetic resonance imaging in children: technique and clinical applications. Pediatr Radiol 46:858–872
Littooij AS, Kwee TC, de Keizer B et al (2015) Whole-body MRI-DWI for assessment of residual disease after completion of therapy in lymphoma: a prospective multicenter study. J Magn Reson Imaging 42:1646–1655
Taouli B, Beer AJ, Chenevert T et al (2016) Diffusion-weighted imaging outside the brain: consensus statement from an ISMRM-sponsored workshop. J Magn Reson Imaging 44:521–540
Padhani AR, Koh DM (2011) Diffusion MR imaging for monitoring of treatment response. Magn Reson Imaging Clin N Am 19:181–209
Barth M, Breuer F, Koopmans PJ et al (2016) Simultaneous multislice (SMS) imaging techniques. Magn Reson Med 75:63–81
Muller S (1989) Simultaneous multislice imaging (SIMUSIM) for improved cardiac imaging. Magn Reson Med 10:145–155
Auerbach EJ, Xu J, Yacoub E et al (2013) Multiband accelerated spin-echo echo planar imaging with reduced peak RF power using time-shifted RF pulses. Magn Reson Med 69:1261–1667
Taron J, Martirosian P, Erb M et al (2016) Simultaneous multislice diffusion-weighted MRI of the liver: analysis of different breathing schemes in comparison to standard sequences. J Magn Reson Imaging 44:865–879
Ohlmeyer S, Laun FB, Palm T et al (2019) Simultaneous multislice echo planar imaging for accelerated diffusion-weighted imaging of malignant and benign breast lesions. Invest Radiol 54:524–530
Buus TW, Sivesgaard K, Jensen AB et al (2020) Simultaneous multislice diffusion-weighted imaging with short tau inversion recovery fat suppression in bone-metastasizing breast cancer. Eur J Radiol 130:109142
Kenkel D, Wurnig MC, Filli L et al (2016) Whole-body diffusion imaging applying simultaneous multi-slice excitation. Rofo 188:381–388
Tabari A, Machado-Rivas F, Kirsch JE et al (2021) Performance of simultaneous multi-slice accelerated diffusion-weighted imaging for assessing focal renal lesions in pediatric patients with tuberous sclerosis complex. Pediatr Radiol 51:77–85
Glutig K, Kruger PC, Oberreuther T et al (2022) Preliminary results of abdominal simultaneous multi-slice accelerated diffusion-weighted imaging with motion-correction in patients with cystic fibrosis and impaired compliance. Abdom Radiol (NY) 47:2783–2794
Stemmer A, Horger W, Kiefer B (2013) Whole-body STIR diffusion-weighted MRI in one third of the time. Proc Intl Soc Mag Reson Med 21. https://cds.ismrm.org/protected/13MProceedings/PDFfiles/2059.PDF. Accessed 01.09.2022
Skipper S PJ (2010) Statsmodel: econometric and statistical modeling with python. Proc of the 9th python in science conf. https://conference.scipy.org/proceedings/scipy2010/pdfs/seabold.pdf. Accessed 01.09.2022
Vallat R (2018) Pingouin: statistics in Python. Journal of Open Source Software 3:1026
Setsompop K, Cohen-Adad J, Gagoski BA et al (2012) Improving diffusion MRI using simultaneous multi-slice echo planar imaging. Neuroimage 63:569–580
Taron J, Schraml C, Pfannenberg C et al (2018) Simultaneous multislice diffusion-weighted imaging in whole-body positron emission tomography/magnetic resonance imaging for multiparametric examination in oncological patients. Eur Radiol 28:3372–3383
Taron J, Martirosian P, Schwenzer NF et al (2016) Scan time minimization in hepatic diffusion-weighted imaging: evaluation of the simultaneous multislice acceleration technique with different acceleration factors and gradient preparation schemes. MAGMA 29:739–749
Blasche M, Fischer D (2017) Magnet homogeneity and shimming (white paper). Journal https://cdn0.scrvt.com/39b415fb07de4d9656c7b516d8e2d907/1800000003946047/b36c69893983/mreadings_mr-in-rt_3rd-edition_magnet-homogeneity-and-shimming_blasche_v2_1800000003946047.pdf. Accessed 01.09.2022
Xu H, Zhang N, Yang DW et al (2021) Scan time reduction in intravoxel incoherent motion diffusion-weighted imaging and diffusion kurtosis imaging of the abdominal organs: using a simultaneous multislice technique with different acceleration factors. J Comput Assist Tomogr 45:507–515
Xu H, Zhang N, Yang DW et al (2021) Feasibility study of simultaneous multislice diffusion kurtosis imaging with different acceleration factors in the liver. BMC Med Imaging 21:132
Filli L, Wurnig M, Nanz D, Luechinger R et al (2014) Whole-body diffusion kurtosis imaging: initial experience on non-Gaussian diffusion in various organs. Invest Radiol 49:773–778
Garcia Santos JM, Ordonez C, Torres del Rio S (2008) ADC measurements at low and high b values: insight into normal brain structure with clinical DWI. Magn Reson Imaging 26:35–44
Rosenkrantz AB, Geppert C, Kiritsy M et al (2015) Diffusion-weighted imaging of the liver: comparison of image quality between monopolar and bipolar acquisition schemes at 3T. Abdom Imaging 40:289–298
Gurses B, Kilickesmez O, Tasdelen N et al (2011) Diffusion tensor imaging of the kidney at 3 Tesla MRI: normative values and repeatability of measurements in healthy volunteers. Diagn Interv Radiol 17:317–322
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization: Paul-Christian Krueger, Katja Glutig; methodology: Paul-Christian Krueger, Katja Glutig, Sophia Ertel; formal analysis and investigation: Paul-Christian Krueger, Sophia Ertel, Martin Krämer; writing—original draft preparation: Paul-Christian Krüger; writing—review and editing: Paul-Christian Krueger, Katja Glutig, Matthias Waginger, Ulf Teichgräber, Hans-Joachim Mentzel, Thomas Benkert; sequence development and time analysis: Thomas Benkert; resources: Paul-Christian Krueger, Matthias Waginger; supervision: Katja Glutig, Ulf Teichgräber, Hans-Joachim Mentzel.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the local ethics committee (Reg. Nr. 2022–2600).
Conflicts of interest
None
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Krueger, PC., Krämer, M., Benkert, T. et al. Whole-body diffusion magnetic resonance imaging with simultaneous multi-slice excitation in children and adolescents. Pediatr Radiol 53, 1485–1496 (2023). https://doi.org/10.1007/s00247-023-05622-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-023-05622-9