Abstract
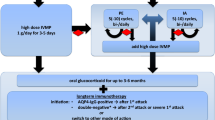
The objective of this study is to describe the treatment and outcome of children and adolescents with N-methyl-d-aspartate receptor (NMDA-R) encephalitis. A retrospective study of children and adolescents with NMDA-R encephalitis was performed by the French Paraneoplastic Neurological Syndrome Reference Center between January 1, 2007 and December 31, 2012. The modified Rankin scale (mRS) was used to assess outcome. Thirty-six children and adolescents with NMDA-R encephalitis were studied. All of the patients received first-line immunotherapy (corticosteroids, intravenous immunoglobulins or plasma exchange), and 81 % received second-line immunotherapy (rituximab or cyclophosphamide). Median time between first-line and second-line treatment was 26 days. During the first 24 months, 30 of 36 patients (83 %) achieved a good outcome (mRS ≤ 2) and 20 of 36 patients (56 %) achieved complete recovery (mRS = 0). Median time to good outcome and to complete recovery was 6 and 24 months, respectively. Three patients (8 %) relapsed, one patient died. In multivariate analysis, age >12 years was a predictor of good outcome and initial mRS ≤ 3 was a predictor of complete recovery. Despite a higher rate of patients who received second-line immunotherapy, the outcome of the patients in the present series was very similar to the outcome reported in previous series. The present study highlights the need for clinical trials to determine the optimal treatment of NMDA-R encephalitis.


Similar content being viewed by others
References
Dalmau J, Tu E, Rossi JE, Voloschin A, Baehring JM, Shimazaki H, Koide R, King D, Mason W, Sansing LH, Dichter MA, Rosenfeld MR, Lynch DR (2007) Paraneoplastic anti—N-methyl-d-aspartate receptor encephalitis associated with ovarian teratoma. Ann Neurol 61:25–36. doi:10.1002/ana.21050
Florance NR, Davis RL, Lam C, Szperka C, Zhou L, Ahmad S, Campen CJ, Moss H, Peter N, Gleichman AJ, Glaser CA, Lynch DR, Rosenfeld MR, Dalmau J (2009) Anti—N-methyl-d-aspartate receptor (NMDAR) encephalitis in children and adolescents. Ann Neurol 66:11–18. doi:10.1002/ana.21756
Irani SR, Bera K, Waters P, Zuliani L, Maxwell S, Zandi MS, Friese Ma, Galea I, Kullmann DM, Beeson D, Lang B, Bien CG, Vincent A (2010) N-methyl-d-aspartate antibody encephalitis: temporal progression of clinical and paraclinical observations in a predominantly non-paraneoplastic disorder of both sexes. Brain 133:1655–1667. doi:10.1093/brain/awq113
Dalmau J, Lancaster E, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R (2011) Clinical experience and laboratory investigations in patients with anti-NMDAR encephalitis. Lancet Neurol 10:63–74. doi:10.1016/S1474-4422(10)70253-2
Titulaer MJ, McCracken L, Gabilondo I, Armangué T, Glaser C, Iizuka T, Honig LS, Benseler SM, Kawachi I, Martinez-Hernandez E, Aguilar E, Gresa-Arribas N, Ryan-Florance N, Torrents A, Saiz A, Rosenfeld MR, Balice-Gordon R, Graus F, Dalmau J (2013) Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol 12:157–165. doi:10.1016/S1474-4422(12)70310-1
Armangue T, Titulaer MJ, Málaga I, Bataller L, Gabilondo I, Graus F, Dalmau J (2013) Pediatric anti-N-methyl-d-aspartate receptor encephalitis-clinical analysis and novel findings in a series of 20 patients. J Pediatr 162:850–856. doi:10.1016/j.jpeds.2012.10.011
Wright S, Hacohen Y, Jacobson L, Agrawal S, Gupta R, Philip S, Smith M, Lim M, Wassmer E, Vincent A (2015) N-methyl-d-aspartate receptor antibody-mediated neurological disease: results of a UK-based surveillance study in children. Arch Dis Child. doi:10.1136/archdischild-2014-306795
Mikasova L, De Rossi P, Bouchet D, Georges F, Rogemond V, Didelot A, Meissirel C, Honnorat J, Groc L (2012) Disrupted surface cross-talk between NMDA and Ephrin-B2 receptors in anti-NMDA encephalitis. Brain 135:1606–1621. doi:10.1093/brain/aws092
Dalmau J, Gleichman AJ, Hughes EG, Rossi JE, Peng X, Dessain SK, Rosenfeld MR, Balice-gordon R, Lynch DR (2008) Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol 7:1091–1098. doi:10.1016/S1474-4422(08)70224-2.Anti-NMDA-receptor
Hacohen Y, Absoud M, Hemingway C, Jacobson L, Lin J-P, Pike M, Pullaperuma S, Siddiqui A, Wassmer E, Waters P, Irani SR, Buckley C, Vincent A, Lim M (2014) NMDA receptor antibodies associated with distinct white matter syndromes. Neurol Neuroimmunol Neuroinflamm 1:e2. doi:10.1212/NXI.0000000000000002
Dale RC, Brilot F, Duffy LV, Twilt M, Waldman AT, Narula S, Muscal E, Deiva K, Andersen E, Eyre MR, Eleftheriou D, Brogan Pa, Kneen R, Alper G, Anlar B, Wassmer E, Heineman K, Hemingway C, Riney CJ, Kornberg A, Tardieu M, Stocco A, Banwell B, Gorman MP, Benseler SM, Lim M (2014) Utility and safety of rituximab in pediatric autoimmune and inflammatory CNS disease. Neurology 83:142–150. doi:10.1212/WNL.0000000000000570
Manto M, Dalmau J, Didelot A, Rogemond V, Honnorat J (2010) In vivo effects of antibodies from patients with anti-NMDA receptor encephalitis: further evidence of synaptic glutamatergic dysfunction. Orphanet J Rare Dis 5:31. doi:10.1186/1750-1172-5-31
Lancaster E, Dalmau J (2012) Neuronal autoantigens-pathogenesis, associated disorders and antibody testing. Nat Rev Neurol 8:380–390. doi:10.1038/nrneurol.2012.99
Camdessanché J-P, Streichenberger N, Cavillon G, Rogemond V, Jousserand G, Honnorat J, Convers P, Antoine J-C (2011) Brain immunohistopathological study in a patient with anti-NMDAR encephalitis. Eur J Neurol 18:929–931. doi:10.1111/j.1468-1331.2010.03180.x
Martinez-Hernandez E, Horvath J, Shiloh-Malawsky Y, Sangha N, Martinez-Lage M, Dalmau J (2011) Analysis of complement and plasma cells in the brain of patients with anti-NMDAR encephalitis. Neurology 77:589–593
Kosmidis ML, Dalakas MC (2010) Practical considerations on the use of rituximab in autoimmune neurological disorders. Ther Adv Neurol Disord 3:93–105. doi:10.1177/1756285609356135
Martin Mdel P, Cravens PD, Winger R, Kieseier BC, Cepok S, Eagar TN, Zamvil SS, Weber MS (2009) Depletion of B lymphocytes from cerebral perivascular spaces by rituximab. Arch Neurol 66:1016–1021
Binstadt B, Caldas A, Turvey S, Weinstein H, Jackson J, Fuhlbrigge R, Sundel R (2003) Rituximab therapy for multisystem autoimmune diseases in pediatric patients. J Pediatr 143:598–604
Nowak RJ, Dicapua DB, Zebardast N, Goldstein JM (2011) Response of patients with refractory myasthenia gravis to rituximab: a retrospective study. Ther Adv Neurol Disord 4:259–266. doi:10.1177/1756285611411503
Kim S-H, Huh S-Y, Lee SJ, Joung A, Kim HJ (2013) A 5-year follow-up of rituximab treatment in patients with neuromyelitis optica spectrum disorder. JAMA Neurol 70:1110–1117. doi:10.1001/jamaneurol.2013.3071
Fekete R, Jankovic J (2012) Childhood stiff-person syndrome improved with rituximab. Case Rep Neurol 4:92–96. doi:10.1159/000339446
Battaglia T, De Grandis E, Mirabelli-Badenier M, Boeri L, Morcaldi G, Barabino P, Intra C, Naselli F, Pistoia V, Veneselli E, Conte M (2012) Response to rituximab in 3 children with opsoclonus-myoclonus syndrome resistant to conventional treatments. Eur J Paediatr Neurol 16:192–195. doi:10.1016/j.ejpn.2011.05.013
Tatencloux S, Chretien P, Rogemond V, Honnorat J, Tardieu M, Deiva K (2015) Intrathecal treatment of anti-N-methyl-d-aspartate receptor encephalitis in children. Dev Med Child Neurol. 57(1):95–99. doi:10.1111/dmcn.12545
DeSena AD, Greenberg BM, Graves D (2014) Three phenotypes of anti-N-methyl-d-aspartate receptor antibody encephalitis in children: prevalence of symptoms and prognosis. Pediatr Neurol 51:542–549. doi:10.1016/j.pediatrneurol.2014.04.030
Acknowledgments
We would like to thank all of the physicians, patients and relatives who helped to collect the clinical data. CSF samples were collected with the help of Neurobiotec Bank and the Hospices Civils de Lyon.
Conflicts of interest
On behalf of all authors, the corresponding authors state that there is no conflict of interest.
Ethical standard
This study has been approved by the institutional review board and ethics committee of the University Claude Bernard Lyon 1/Hospices Civils de Lyon.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Anastasia Zekeridou and Evgenia Karantoni contributed equally to the manuscript.
Rights and permissions
About this article
Cite this article
Zekeridou, A., Karantoni, E., Viaccoz, A. et al. Treatment and outcome of children and adolescents with N-methyl-d-aspartate receptor encephalitis. J Neurol 262, 1859–1866 (2015). https://doi.org/10.1007/s00415-015-7781-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-015-7781-9