Abstract
Background
Longitudinal measures of structural brain changes using MRI in relation to clinical features and progression patterns in PD have been assessed in previous studies, but few were conducted in well-defined and large cohorts, including prospective clinical assessments of both motor and non-motor symptoms.
Objective
We aimed to identify brain volumetric changes characterizing PD patients, and determine whether regional brain volumetric characteristics at baseline can predict motor, psycho-behavioral and cognitive evolution at one year in a prospective cohort of PD patients.
Methods
In this multicentric 1 year longitudinal study, PD patients and healthy controls from the MPI-R2* cohort were assessed for demographical, clinical and brain volumetric characteristics. Distinct subgroups of PD patients according to motor, cognitive and psycho-behavioral evolution were identified at the end of follow-up.
Results
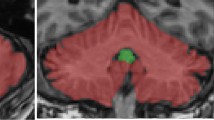
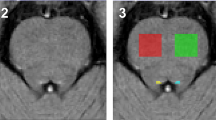
One hundred and fifty PD patients and 73 control subjects were included in our analysis. Over one year, there was no significant difference in volume variations between PD and control subjects, regardless of the brain region considered. However, we observed a reduction in posterior cingulate cortex volume at baseline in PD patients with motor deterioration at one year (p = 0.017). We also observed a bilateral reduction of the volume of the amygdala (p = 0.015 and p = 0.041) and hippocampus (p = 0.015 and p = 0.053) at baseline in patients with psycho-behavioral deterioration, regardless of age, dopaminergic treatment and center.
Conclusion
Brain volumetric characteristics at baseline may predict clinical trajectories at 1 year in PD as posterior cingulate cortex atrophy was associated with motor decline, while amygdala and hippocampus atrophy were associated with psycho-behavioral decline.


Similar content being viewed by others
Data availability
Not applicable.
References
Greenland JC, Williams-Gray CH, Barker RA (2019) The clinical heterogeneity of Parkinson’s disease and its therapeutic implications. Eur J Neurosci 49:328–338. https://doi.org/10.1111/ejn.14094
Schrag A, Schott JM (2006) Epidemiological, clinical, and genetic characteristics of early-onset parkinsonism. Lancet Neurol 5:355–363. https://doi.org/10.1016/S1474-4422(06)70411-2
Yang J, Burciu RG, Vaillancourt DE (2018) Longitudinal Progression Markers of Parkinson’s Disease: Current View on Structural Imaging. Curr Neurol Neurosci Rep 18:83. https://doi.org/10.1007/s11910-018-0894-7
McGhee DJ, Royle PL, Thompson PA, Wright DE, Zajicek JP, Counsell CE (2013) A systematic review of biomarkers for disease progression in Parkinson’s disease. BMC Neurol 13:35. https://doi.org/10.1186/1471-2377-13-35
Sarasso E, Agosta F, Piramide N, Filippi M (2021) Progression of grey and white matter brain damage in Parkinson’s disease: a critical review of structural MRI literature. J Neurol 268:3144–3179. https://doi.org/10.1007/s00415-020-09863-8
Filippi M, Sarasso E, Piramide N, Stojkovic T, Stankovic I, Basaia S, Fontana A, Tomic A, Markovic V, Stefanova E, Kostic VS, Agosta F (2020) Progressive brain atrophy and clinical evolution in Parkinson’s disease. Neuroimage Clin 28:102374. https://doi.org/10.1016/j.nicl.2020.102374
Mak E, Su L, Williams GB, Firbank MJ, Lawson RA, Yarnall AJ, Duncan GW, Owen AM, Khoo TK, Brooks DJ, Rowe JB, Barker RA, Burn DJ, O’Brien JT (2015) Baseline and longitudinal grey matter changes in newly diagnosed Parkinson’s disease: ICICLE-PD study. Brain 138:2974–2986. https://doi.org/10.1093/brain/awv211
Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatr 55:181–184
Goetz CG, Fahn S, Martinez-Martin P, Poewe W, Sampaio C, Stebbins GT, Stern MB, Tilley BC, Dodel R, Dubois B, Holloway R, Jankovic J, Kulisevsky J, Lang AE, Lees A, Leurgans S, LeWitt PA, Nyenhuis D, Olanow CW, Rascol O, Schrag A, Teresi JA, Van Hilten JJ, LaPelle N (2007) Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Process, format, and clinimetric testing plan. Mov Disord 22:41–47. https://doi.org/10.1002/mds.21198
Hoehn MM, Yahr MD (1967) Parkinsonism: onset, progression and mortality. Neurology 17:427–442
Schwab RS, England AC (1969) Projection techniques for evaluating surgery in Parkinson’s Disease. E. & S. Livingstone Ltd., Edinburgh
Giladi N, Tal J, Azulay T, Rascol O, Brooks DJ, Melamed E, Oertel W, Poewe WH, Stocchi F, Tolosa E (2009) Validation of the freezing of gait questionnaire in patients with Parkinson’s disease. Mov Disord 24:655–661. https://doi.org/10.1002/mds.21745
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiatry 23:56–62. https://doi.org/10.1136/jnnp.23.1.56
Thompson E (2015) Hamilton Rating Scale for Anxiety (HAM-A). Occup Med (Lond) 65:601. https://doi.org/10.1093/occmed/kqv054
Sockeel P, Dujardin K, Devos D, Denève C, Destée A, Defebvre L (2006) The Lille apathy rating scale (LARS), a new instrument for detecting and quantifying apathy: validation in Parkinson’s disease. J Neurol Neurosurg Psychiatry 77:579–584. https://doi.org/10.1136/jnnp.2005.075929
Rieu I, Martinez-Martin P, Pereira B, De Chazeron I, Verhagen Metman L, Jahanshahi M, Ardouin C, Chéreau I, Brefel-Courbon C, Ory-Magne F, Klinger H, Peyrol F, Schupbach M, Dujardin K, Tison F, Houeto JL, Krack P, Durif F (2015) International validation of a behavioral scale in Parkinson’s disease without dementia. Mov Disord 30:705–713. https://doi.org/10.1002/mds.26223
Shulman LM, Gruber-Baldini AL, Anderson KE, Fishman PS, Reich SG, Weiner WJ (2010) The clinically important difference on the unified Parkinson’s disease rating scale. Arch Neurol 67:64–70. https://doi.org/10.1001/archneurol.2009.295
Lhommée E, Wojtecki L, Czernecki V, Witt K, Maier F, Tonder L, Timmermann L, Hälbig TD, Pineau F, Durif F, Witjas T, Pinsker M, Mehdorn M, Sixel-Döring F, Kupsch A, Krüger R, Elben S, Chabardès S, Thobois S, Brefel-Courbon C, Ory-Magne F, Regis J-M, Maltête D, Sauvaget A, Rau J, Schnitzler A, Schüpbach M, Schade-Brittinger C, Deuschl G, Houeto J-L, Krack P, Negovanska V, Welter M-L, Corvol J-C, Agid Y, Navarro S, Meier N, Hartmann A, Hesekamp H, Cornu P, Möller B, Nebel A, Raethjen J, Knudsen K, Volkmann J, Falk D, Paschen S, Meister I, Kuhn J, Donner K, Kessler J, Barbe M, Fink G, Maarouf M, Kühn A, Müller B, Faust K, Gruber D, Schneider G-H, Seigneuret E, Pollak P, Fraix V, Kistner A, Rascol O, Arbus C, Danet L, Chaynes P, Groiss SJ, Hartmann C, Südmeyer M, Partowinia-Peters M, Vesper J, Ledily S, Damier P, Raoul S, Trenkwalder C, Richter-Dreske W, Wächter T, Weiss D, Eusebio A, Azulay JP, Polo G, Pinto S, Levin J, Dornier S, Pene F, Hourton D, Quintin M, Hoffart-Jourdain C, Brocvielle H, Balthasar K, Stein M, Harnisch S, Reuss A, Aminossadati B, Nasemann C, Oertel W, Bataille B, Hellwig D, Gharabaghi A, Amtage F, Mertens P, Kloss M, Post B, Speelman H (2018) Behavioural outcomes of subthalamic stimulation and medical therapy versus medical therapy alone for Parkinson’s disease with early motor complications (EARLYSTIM trial): secondary analysis of an open-label randomised trial. Lancet Neurol 17:223–231. https://doi.org/10.1016/S1474-4422(18)30035-8
Li A, Yue L, Xiao S, Liu M (2022) Cognitive Function Assessment and Prediction for Subjective Cognitive Decline and Mild Cognitive Impairment. Brain Imaging Behav 16:645–658. https://doi.org/10.1007/s11682-021-00545-1
Bender R, Lange S (2001) Adjusting for multiple testing–when and how? J Clin Epidemiol 54:343–349. https://doi.org/10.1016/s0895-4356(00)00314-0
Feise RJ (2002) Do multiple outcome measures require p-value adjustment? BMC Med Res Methodol 2:8
Ibarretxe-Bilbao N, Junque C, Segura B, Baggio HC, Marti MJ, Valldeoriola F, Bargallo N, Tolosa E (2012) Progression of cortical thinning in early Parkinson’s disease: Cortical Thinning in Early PD. Mov Disord 27:1746–1753. https://doi.org/10.1002/mds.25240
Mollenhauer B, Zimmermann J, Sixel-Döring F, Focke NK, Wicke T, Ebentheuer J, Schaumburg M, Lang E, Trautmann E, Zetterberg H, Taylor P, Friede T, Trenkwalder C (2016) Monitoring of 30 marker candidates in early Parkinson disease as progression markers. Neurology 87:168–177. https://doi.org/10.1212/WNL.0000000000002651
Jia X, Liang P, Li Y, Shi L, Wang D, Li K (2015) Longitudinal Study of Gray Matter Changes in Parkinson Disease. AJNR Am J Neuroradiol 36:2219–2226. https://doi.org/10.3174/ajnr.A4447
Melzer TR, Myall DJ, MacAskill MR, Pitcher TL, Livingston L, Watts R, Keenan RJ, Dalrymple-Alford JC, Anderson TJ (2015) Tracking Parkinson’s Disease over One Year with Multimodal Magnetic Resonance Imaging in a Group of Older Patients with Moderate Disease. PLoS ONE. 10:e0143923. https://doi.org/10.1371/journal.pone.0143923
Blair JC, Barrett MJ, Patrie J, Flanigan JL, Sperling SA, Elias WJ, Druzgal TJ (2019) Brain MRI Reveals Ascending Atrophy in Parkinson’s Disease Across Severity. Front Neurol 10:1329. https://doi.org/10.3389/fneur.2019.01329
Oh BH, Moon HC, Kim A, Kim HJ, Cheong CJ, Park YS (2021) Prefrontal and hippocampal atrophy using 7-tesla magnetic resonance imaging in patients with Parkinson’s disease. Acta Radiol Open 10:2058460120988097. https://doi.org/10.1177/2058460120988097
Ramírez-Ruiz B, Martí MJ, Tolosa E, Bartrés-Faz D, Summerfield Ch, Salgado-Pineda P, Gómez-Ansón B, Junqué C (2005) Longitudinal evaluation of cerebral morphological changes in Parkinson’s disease with and without dementia. J Neurol 252:1345–1352. https://doi.org/10.1007/s00415-005-0864-2
Benarroch EE (2015) The amygdala: Functional organization and involvement in neurologic disorders. Neurology 84:313–324. https://doi.org/10.1212/WNL.0000000000001171
Ibarretxe-Bilbao N, Ramírez-Ruiz B, Tolosa E, Martí MJ, Valldeoriola F, Bargalló N, Junqué C (2008) Hippocampal head atrophy predominance in Parkinson’s disease with hallucinations and with dementia. J Neurol 255:1324–1331. https://doi.org/10.1007/s00415-008-0885-8
Carey G, Görmezoğlu M, de Jong JJA, Hofman PAM, Backes WH, Dujardin K, Leentjens AFG (2021) Neuroimaging of Anxiety in Parkinson’s Disease: A Systematic Review. Mov Disord 36:327–339. https://doi.org/10.1002/mds.28404
Thobois S, Prange S, Sgambato-Faure V, Tremblay L, Broussolle E (2017) Imaging the Etiology of Apathy, Anxiety, and Depression in Parkinson’s Disease: Implication for Treatment. Curr Neurol Neurosci Rep 17:76. https://doi.org/10.1007/s11910-017-0788-0
Melzer TR, Watts R, MacAskill MR, Pitcher TL, Livingston L, Keenan RJ, Dalrymple-Alford JC, Anderson TJ (2012) Grey matter atrophy in cognitively impaired Parkinson’s disease. J Neurol Neurosurg Psychiatry 83:188–194. https://doi.org/10.1136/jnnp-2011-300828
Kamagata K, Motoi Y, Hori M, Suzuki M, Nakanishi A, Shimoji K, Kyougoku S, Kuwatsuru R, Sasai K, Abe O, Mizuno Y, Aoki S, Hattori N (2011) Posterior hypoperfusion in Parkinson’s disease with and without dementia measured with arterial spin labeling MRI. J Magn Reson Imaging 33:803–807. https://doi.org/10.1002/jmri.22515
Hosokai Y, Nishio Y, Hirayama K, Takeda A, Ishioka T, Sawada Y, Suzuki K, Itoyama Y, Takahashi S, Fukuda H, Mori E (2009) Distinct patterns of regional cerebral glucose metabolism in Parkinson’s disease with and without mild cognitive impairment. Mov Disord 24:854–862. https://doi.org/10.1002/mds.22444
Vogt BA (2019) Cingulate cortex in Parkinson’s disease. Handb Clin Neurol 166:253–266. https://doi.org/10.1016/B978-0-444-64196-0.00013-3
de Schipper LJ, van der Grond J, Marinus J, Henselmans JML, van Hilten JJ (2017) Loss of integrity and atrophy in cingulate structural covariance networks in Parkinson’s disease. NeuroImage 15:587–593. https://doi.org/10.1016/j.nicl.2017.05.012
Goldman JG, Stebbins GT, Dinh V, Bernard B, Merkitch D, deToledo-Morrell L, Goetz CG (2014) Visuoperceptive region atrophy independent of cognitive status in patients with Parkinson’s disease with hallucinations. Brain. 137:849–859. https://doi.org/10.1093/brain/awt360
Vercruysse S, Leunissen I, Vervoort G, Vandenberghe W, Swinnen S, Nieuwboer A (2015) Microstructural changes in white matter associated with freezing of gait in Parkinson’s disease: FOg-Related White Matter Changes. Mov Disord 30:567–576. https://doi.org/10.1002/mds.26130
Evers LJW, Krijthe JH, Meinders MJ, Bloem BR, Heskes TM (2019) Measuring Parkinson’s disease over time: the real-world within-subject reliability of the MDS-UPDRS. Mov Disord 34:1480–1487. https://doi.org/10.1002/mds.27790
Aleksovski D, Miljkovic D, Bravi D, Antonini A (2018) Disease progression in Parkinson subtypes: the PPMI dataset. Neurol Sci 39:1971–1976. https://doi.org/10.1007/s10072-018-3522-z
Lessig S, Nie D, Xu R, Corey-Bloom J (2012) Changes on brief cognitive instruments over time in Parkinson’s disease. Mov Disord 27:1125–1128. https://doi.org/10.1002/mds.25070
Makarious MB, Leonard HL, Vitale D, Iwaki H, Sargent L, Dadu A, Violich I, Hutchins E, Saffo D, Bandres-Ciga S, Kim JJ, Song Y, Maleknia M, Bookman M, Nojopranoto W, Campbell RH, Hashemi SH, Botia JA, Carter JF, Craig DW, Van Keuren-Jensen K, Morris HR, Hardy JA, Blauwendraat C, Singleton AB, Faghri F, Nalls MA (2022) Multi-modality machine learning predicting Parkinson’s disease. NPJ Parkinsons Dis 8:35. https://doi.org/10.1038/s41531-022-00288-w
Funding
France Parkinson Association, Federation for Brain Research, Neuroscience-Parkinson network, call for tenders for the interregional clinical research hospital program.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no relevant financial or non-financial interests to disclose. The authors have no competing interests to declare that are relevant to the content of this article.
Supplementary Information
Below is the link to the electronic supplementary material.
415_2023_11947_MOESM2_ESM.pdf
Supplementary file2 Box plots representation of posterior cingulate cortex volumes in parkinsonian patients with motor impairment vs parkinsonian patients without motor impairment. Considering univariate analysis, effect-size for PCC volumes in PD with motor impairment vs PD without motor impairment using box plots was moderate (− 0.41 [− 0.77; − 0.06]) according to Cohen who has defined effect size bounds as small (ES = 0.2), medium (ES = 0.5), and large (ES = 0.8) (PDF 216 KB)
415_2023_11947_MOESM3_ESM.pdf
Supplementary file3 Univariate results concerning relations between clinical and volumetric changes between V1 and V2, for PD patients: the heatmap represents correlation coefficients intensity (Pearson or Spearman according to statistical distribution. LARS = Lille Apathy Rating Scale; HAM-D = Hamilton Depression scale; HAM-A = Hamilton Anxiety scale; MoCA = Montreal Cognitive Assessment; FOG-Q = Freezing Of Gate Questionnaire; ASBPD = Ardouin Scale of Behavior in Parkinson’s Disease; MDS-UPDRS = Movement Disorder’s Society Unified Parkinson's Disease Rating Scale; LEDD = Levodopa Equivalent Daily Dose; H&Y = Hoehn & Yahr scale. L = left; R = Right. White = No correlation (− 0.2 < r < 0.2); Blue = Low to moderate inverse correlation (− 0.5 < r < − 0.2); Red = Low to moderate correlation (0.2 < r < 0.5) (PDF 55 KB)
415_2023_11947_MOESM4_ESM.pdf
Supplementary file4 Scatter plots representing correlations between brain volume changes and clinical changes in PD patients over 1 year. ASBPD = Ardouin Scale of Behavior in Parkinson’s Disease; MDS-UPDRS = Movement Disorder’s Society Unified Parkinson's Disease Rating Scale (PDF 115 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marques, A., Macias, E., Pereira, B. et al. Volumetric changes and clinical trajectories in Parkinson’s disease: a prospective multicentric study. J Neurol 270, 6033–6043 (2023). https://doi.org/10.1007/s00415-023-11947-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11947-0