Abstract
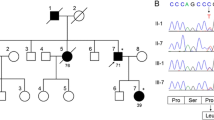
The detailed clinical, electrophysiological and imaging data of three German autosomal dominant cerebellar ataxia (ADCA) families are reported. Linkage to SCA2 was established using microsatellite markers D12S105, D12S1339(1328), D12S 1340(1329) yielding a lod score exceeding +3.0 for the combined data. Analysis of the pedigree data provided evidence of anticipation as observed in other neurodegenerative disorders due to polyglutamine expansion encoded by a CAG repeat. This hypothesis was confirmed by the detection of the SCA2-specific pathological protein using the 1C2 monoclonal antibody which selectively recognizes large polyglutamine expansions and the characterization of a CAG expansion in the patients. Clinically, the families were characterized by progressive ataxia of stance, gait and limbs. Saccade velocity was markedly reduced in SCA2. Further oculomotor findings were gaze palsy, impaired smooth pursuit and reduced optokinetic reflex. Dementia and pyramidal tract signs were rather rare, while peripheral involvement (reduced or absent ankle reflexes, fasciculation-like movements, amyotrophy) was a prominent feature. Electrophysiological investigations provided evidence of sensory neuropathy of the axonal type and degeneration of the posterior columns. Imaging studies demonstrated severe shrinkage of brain-stem structures even in early stages of the disease.
Similar content being viewed by others
References
Belal S, Cancel G, Stevanin G, Hentati F, Khati C, et al (1994) Clinical and genetic analysis of a Tunisian family with autosomal dominant cerebellar ataxia type I linked to the SCA2 locus. Neurology 44:1423–1426
Biirk K, Abele M, Fetter M, Dichgans J, Skalej M, Laccone F, Didierjean O, Brice A, Klockgether T (1996) Autosomal dominant cerebellar ataxia type I. Clinical features and magnetic resonance imaging findings in families with SCA1, SCA2 and SCA3 mutation. Brain 119:1497–1505
Büttner-Ennever JA, Wadia NH, Sakai H, Schwendemann G (1985) Neuroanatomy of oculomotor structures in olivopontocerebellar atrophy (OPCA) patient with slow saccades (abstract). J Neurol 232[Suppl]:285
Chung MY, Ranum LP, Duvick LA, Servadio A, Zoghbi HY (1993) Evidence for a mechanism predisposing intergenerational CAG repeat instability in spinocerebellar ataxia type I. Nature Genet 5:254–258
Dürr A, Brice A, Lepage-Lezin A, Cancel G, et al (1995) Autosomal dominant cerebellar ataxia type I linked to chromosome 12q (SCA2: spinocerebellar ataxia type 2). Clin Neurosci 3:12–16
Dürr A, Smadja D, Cancel G, Lezin A, Stevanin G, Mikol J, Beilance R, et al (1995) Autosomal dominant cerebellar ataxia type I in Martinique (French West Indies). Clinical and neuropathological analysis of 53 patients from three unrelated SCA 2 families. Brain 118:1573–1581
Fetter M, Klockgether T, Schulz JB, Faiss J, König E, Dichgans J (1993) Oculomotor abnormalities and MRI findings in idiopathic cerebellar ataxia. J Neurol 241:234–241
Filla A, De Michele G, Banfi S, Santoro L, Perretti A, et al (1995) Has spinocerebellar ataxia type 2 a distinct phenotype? Genetic and clinical study of an Italian family. Neurology 45: 793–796
Gardner K, Alderson K, Galster B, et al (1994) Autosomal dominant spinocerebellar ataxia: clinical description of a distinct hereditary ataxia and localization to chromosome 16 (SCA4) in a Utah kindred. Neurology 44:A361
Gispert S, Twells R, Orozco G, Brice A, Weber J, et al (1993) Chromosomal assignment of the second locus for autosomal dominant cerebellar ataxia (SCA 2) to chromosome 12q23-24. 1. Nature Genet 4:295–299
Gispert S, Lunkes A, Santos N, Orozco G, Ha-Hao D, Ratzlaff T, et al (1995) Localization of the candidate gene Damino acid oxidase outside the refined I-cm region of spinocerebellar ataxia 2 (letter). Am J Hum Genet 57:972–975
Harding AE (1982) Clinical features and classification of the late onset autosomal dominant cerebellar ataxias, including descendants of ’The Drew Family of Walworth’. Brain 105:1–28
Hazan J, Dubay C, Pankowiak MP, et al (1992) A genetic linkage map of human chromosome 20 composed entirely of microsatellite markers. Genomics 12:183–189
Imbert G, Saudou F, Yvert G, Devys D, Trottier Y, Garnier JM, et al (1996) Cloning of the gene for spinocerebellar ataxia 2 reveals a locus with high sensitivity to expanded CAG/glutamine repeats. Nature Genet 14:285–291
Kawaguchi Y, Okamoto T, Taniwaki M, Aizawa M, Inoue M, Katayama S, et al (1994) CAG expansions in a novel gene for Machado-Jospeh disease at chromosome 14q32. 1. Nature Genet 8:221–228
Klockgether T, Schroth G, Diener HC, Dichgans J (1990) Idiopathic cerebellar ataxia of late onset: natural history and MRI morphology. J Neurol Neurosurg Psychiatry 53:297–305
Klockgether T, Wüllner U, Dichgans J (1993) Clinical and imaging correlations in inherited ataxias. Adv Neurol 61:77–96
Lathrop GM, Laouel JM (1991) Statistical methods for linkage analysis. In: Rao CR, Chakraborty D (eds) Handbook of statistics, vol 8, Elsevier, Amsterdam, pp 81–123
Lima L, Coutinho P (1980) Clinical criteria for diagnosis of Machado-Joseph disease: report of a non-Azorean Portuguese family. Neurology 30: 319–322
Lezin A, Cancel G, Stevanin G, Smadja D, Vernant JC, Dürr A, Martial J, Buisson GG, Bellance R, Chneiweiss H, Agid Y, Brice A (1996) Autosomal dominant cerebellar ataxia type I in Martinique (French West Indies): genetic analysis of three unrelated SCA2 families. Hum Genet 97:671–676
Lopes-Cendes I, Andermann E, Attig E, Cendes F, et al (1994) Confirmation of the SCA2 locus as an alternative locus for dominantly inherited spinocerebellar ataxias and refinement of the candidate region. Am J Hum Genet 54:774–781
Orozco Diaz G, Estrada R, Perry TL (1989) Dominantly inherited olivopontocerebellar atrophy from eastern Cuba: clinical, neuropathological, and biochemical findings. J Neurol Sci 93: 37–50
Orozco Diaz G, Nodarse Fleites A, Cordoves Sagaz R, Auburger G (1990) Autosomal dominant cerebellar ataxia: clinical analysis of 263 patients from a homogeneous population in Holguin, Cuba. Neurology 40:1369–1375
Orr HT, Chung M, Banfi S, et al (1993) Expansion of an unstable trinucleotide repeat in spinocerebellar ataxia type I. Nature Genet 4:221–226
Ott J (1991) Analysis of human genetic linkage, revised edition. The Johns Hopkins University Press, Baltimore
Pulst SM, Nechiporuk A, Nechiporuk T, Gispert S, Chen XN, Lopes-Cendes I, et al (1996) Moderate expansion of a normally biallelic trinucleotide repeat in spinocerebellar ataxia type 2. Nature Genet 14:269–276
Ranum L, Schut L, Lundgren J, Orr D (1994) Spinocerebellar ataxia type 5 in a family descended from the grandparents of President Lincoln maps to chromosome 11. Nature Genet 8:280–284
Sanpei K, Takano H, Igarashi S, Sato T, Oyake M, Sasaki H, et al (1996) Identification of the spinocerebellar ataxia type 2 gene using a direct identification of repeat expansion and cloning technique, DIRECT. Nature Genet 14:277–284
Trottier Y, Lutz Y, Stevanin G, Imbert G, Devys D, et al (1995) Polyglutamine expansion as a pathological epitope in Huntington’s disease and four dominant cerebellar ataxias. Nature 378:403–406
Wüllner U, Klockgether T, Petersen D, Naegele T, Dichgans J (1993) Magnetic resonance imaging in hereditary and idiopathic ataxia. Neurology 43: 318–325
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bürk, K., Stevanin, G., Didierjean, O. et al. Clinical and genetic analysis of three German kindreds with autosomal dominant cerebellar ataxia type I linked to the SCA2 locus. J Neurol 244, 256–261 (1997). https://doi.org/10.1007/s004150050081
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s004150050081