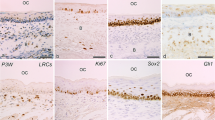
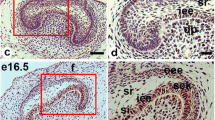
Abstract
The Nestin gene encodes type VI intermediate filament and is known to be expressed in undifferentiated cells during neurogenesis and myogenesis. To regulate Nestin expression, the first or second intron enhancer is activated in a tissue-dependent manner, for example, the former in mesodermal cells and the latter in neural stem cells. Although Nestin has also been used as a differentiation marker for odontoblasts during tooth development, how Nestin expression is regulated in odontoblasts remains unclear. Therefore, this study aimed to compare the expression patterns of Nestin-GFP (green fluorescent protein) with that of endogenous Nestin in developing teeth of Nestin-EGFP (enhanced GFP) transgenic mice, in which the second intron enhancer is connected with the EGFP domain, at postnatal 7d, 3w, and 8w. Immunohistochemical and in situ hybridization analyses revealed that endogenous Nestin protein and Nestin mRNA were intensely expressed in differentiated odontoblasts, while GFP immunoreactivity, which reflects the activity of Nestin second intron enhancer-mediated transcription, was mainly observed in the subodontoblastic layer. These results indicate that the first intron enhancer may be activated in differentiated odontoblasts. Intriguingly, Nestin-GFP expression in the subodontoblastic layer was found to be restricted to the coronal pulp of molars, which is susceptible to tooth injuries. Because the subodontoblastic layer serves as a reservoir of newly differentiated odontoblast-like cells upon exogenous stimuli to dentin, our findings suggest that the original odontoblasts and regenerated odontoblast-like cells may differently regulate Nestin expression.







Similar content being viewed by others
References
Berkovitz BKB, Holland GR, Moxham BJ (2009) Oral anatomy, embryology, and histology. Mosby, Edinburgh
Harada M, Kenmotsu S, Nakasone N, Nakakura-Ohshima K, Ohshima H (2008) Cell dynamics in the pulpal healing process following cavity preparation in rat molars. Histochem Cell Biol 130:773–783
Hasegawa T, Suzuki H, Yoshie H, Ohshima H (2007) Influence of extended operation time and of occlusal force on determination of pulpal healing pattern in replanted mouse molars. Cell Tissue Res 329:259–272
Ishikawa Y, Ida-Yonemochi H, Suzuki H, Nakakura-Ohshima K, Jung HS, Honda MJ, Ishii Y, Watanabe N, Ohshima H (2010) Mapping of BrdU label-retaining dental pulp cells in growing teeth and their regenerative capacity after injuries. Histochem Cell Biol 134:227–241
Ishikawa Y, Ida-Yonemochi H, Nakakura-Ohshima K, Ohshima H (2012) The relationship between cell proliferation and differentiation and mapping of putative dental pulp stem/progenitor cells during mouse molar development by chasing BrdU-labeling. Cell Tissue Res 348:95–107
Izumi T, Kobayashi I, Okamura K, Matsuo K, Kiyoshima T, Ishibashi Y, Inoue H, Sakai H (1996) An immunohistochemical study of HLA-DR and alpha 1-antichymotrypsin-positive cells in the pulp of human non-carious and carious teeth. Arch Oral Biol 41:627–630
Jussila M, Thesleff I (2012) Signaling networks regulating tooth organogenesis and regeneration, and the specification of dental mesenchymal and epithelial cell lineages. Cold Spring Harb Perspect Biol 4:a008425
Kamal AM, Okiji T, Kawashima N, Suda H (1997) Defense responses of dentin/pulp complex to experimentally induced caries in rat molars: an immunohistochemical study on kinetics of pulpal Ia antigen-expressing cells and macrophages. J Endod 23:115–120
Kawagishi E, Nakakura-Ohshima K, Nomura S, Ohshima H (2006) Pulpal responses to cavity preparation in aged rat molars. Cell Tissue Res 326:111–122
Kawaguchi A, Miyata T, Sawamoto K, Takashita N, Murayama A, Akamatsu W, Ogawa M, Okabe M, Tano Y, Goldman SA, Okano H (2001) Nestin-EGFP transgenic mice: visualization of the self-renewal and multipotency of CNS stem cells. Mol Cell Neurosci 17:259–273
Keyoung HM, Roy NS, Benraiss A, Louissaint A Jr, Suzuki A, Hashimoto M, Rashbaum WK, Okano H, Goldman SA (2001) High-yield selection and extraction of two promoter-defined phenotypes of neural stem cells from the fetal human brain. Nat Biotechnol 19:843–850
Kibe K, Nakatomi M, Kataoka S, Toyono T, Seta Y (2018) Hey1 and Hey2 are differently expressed during mouse tooth development. Gene Expr Patterns GEP 27:99–105
Lendahl U, Zimmerman LB, McKay RD (1990) CNS stem cells express a new class of intermediate filament protein. Cell 60:585–595
Li J, Huang X, Xu X, Mayo J, Bringas P Jr, Jiang R, Wang S, Chai Y (2011) SMAD4-mediated WNT signaling controls the fate of cranial neural crest cells during tooth morphogenesis. Development 138:1977–1989
Mendez-Ferrer S, Michurina TV, Ferraro F, Mazloom AR, Macarthur BD, Lira SA, Scadden DT, Ma’ayan A, Enikolopov GN, Frenette PS (2010) Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature 466:829–834
Mitsiadis TA, Fried K, Goridis C (1999) Reactivation of Delta–Notch signaling after injury: complementary expression patterns of ligand and receptor in dental pulp. Exp Cell Res 246:312–318
Morshead CM, Reynolds BA, Craig CG, McBurney MW, Staines WA, Morassutti D, Weiss S, van der Kooy D (1994) Neural stem cells in the adult mammalian forebrain: a relatively quiescent subpopulation of subependymal cells. Neuron 13:1071–1082
Nakakura-Ohshima K, Watanabe J, Kenmotsu S, Ohshima H (2003) Possible role of immunocompetent cells and the expression of heat shock protein-25 in the process of pulpal regeneration after tooth injury in rat molars. J Electron Microsc (Tokyo) 52:581–591
Nakatomi M, Morita I, Eto K, Ota MS (2006) Sonic hedgehog signaling is important in tooth root development. J Dent Res 85:427–431
Nanci A (2013) Ten Cate’s oral histology: development, structure, and function. Elsevier, St. Louis
Ohshima H, Sato O, Kawahara I, Maeda T, Takano Y (1995) Responses of immunocompetent cells to cavity preparation in rat molars: an immunohistochemical study using OX6-monoclonal antibody. Connect Tissue Res 32:303–311
Ohshima H, Nakakura-Ohshima K, Takeuchi K, Hoshino M, Takano Y, Maeda T (2003) Pulpal regeneration after cavity preparation, with special reference to close spatio-relationships between odontoblasts and immunocompetent cells. Microsc Res Tech 60:483–490
Quispe-Salcedo A, Ida-Yonemochi H, Nakatomi M, Ohshima H (2012) Expression patterns of nestin and dentin sialoprotein during dentinogenesis in mice. Biomed Res 33:119–132
Reynolds BA, Weiss S (1992) Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science 255:1707–1710
Roy NS, Wang S, Jiang L, Kang J, Benraiss A, Harrison-Restelli C, Fraser RA, Couldwell WT, Kawaguchi A, Okano H, Nedergaard M, Goldman SA (2000) In vitro neurogenesis by progenitor cells isolated from the adult human hippocampus. Nat Med 6:271–277
Ruch JV, Lesot H, Begue-Kirn C (1995) Odontoblast differentiation. Int J Dev Biol 39:51–68
Rungvechvuttivittaya S, Okiji T, Suda H (1998) Responses of macrophage-associated antigen-expressing cells in the dental pulp of rat molars to experimental tooth replantation. Arch Oral Biol 43:701–710
Saito K, Nakatomi M, Ohshima H (2013) Dynamics of bromodeoxyuridine label-retaining dental pulp cells during pulpal healing after cavity preparation in mice. J Endod 39:1250–1255
Saito K, Nakatomi M, Kenmotsu S, Ohshima H (2014) Allogenic tooth transplantation inhibits the maintenance of dental pulp stem/progenitor cells in mice. Cell Tissue Res 356:357–367
Saito K, Nakatomi M, Ida-Yonemochi H, Ohshima H (2016) Osteopontin is essential for type I collagen secretion in reparative dentin. J Dent Res 95:1034–1041
Sakurai K, Okiji T, Suda H (1999) Co-increase of nerve fibers and HLA-DR- and/or factor-XIIIa-expressing dendritic cells in dentinal caries-affected regions of the human dental pulp: an immunohistochemical study. J Dent Res 78:1596–1608
Shimizu A, Nakakura-Ohshima K, Noda T, Maeda T, Ohshima H (2000) Responses of immunocompetent cells in the dental pulp to replantation during the regeneration process in rat molars. Cell Tissue Res 302:221–233
Sunabori T, Tokunaga A, Nagai T, Sawamoto K, Okabe M, Miyawaki A, Matsuzaki Y, Miyata T, Okano H (2008) Cell-cycle-specific nestin expression coordinates with morphological changes in embryonic cortical neural progenitors. J Cell Sci 121:1204–1212
Tanaka S, Kamachi Y, Tanouchi A, Hamada H, Jing N, Kondoh H (2004) Interplay of SOX and POU factors in regulation of the Nestin gene in neural primordial cells. Mol Cell Biol 24:8834–8846
Tucker A, Sharpe P (2004) The cutting-edge of mammalian development; how the embryo makes teeth. Nat Rev Genet 5:499–508
Yoshiba N, Yoshiba K, Nakamura H, Iwaku M, Ozawa H (1996) Immunohistochemical localization of HLA-DR-positive cells in unerupted and erupted normal and carious human teeth. J Dent Res 75:1585–1589
Zimmerman L, Parr B, Lendahl U, Cunningham M, McKay R, Gavin B, Mann J, Vassileva G, McMahon A (1994) Independent regulatory elements in the nestin gene direct transgene expression to neural stem cells or muscle precursors. Neuron 12:11–24
Acknowledgements
This work was supported by the Japan Society for the Promotion of Science KAKENHI (Grant Numbers 25293371 and 17H04366 to H.O.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Hideyuki Okano is a founding Scientist of SanBio Co Ltd. and K Pharma Inc. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Nakatomi, M., Quispe-Salcedo, A., Sakaguchi, M. et al. Nestin expression is differently regulated between odontoblasts and the subodontoblastic layer in mice. Histochem Cell Biol 149, 383–391 (2018). https://doi.org/10.1007/s00418-018-1651-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-018-1651-3