Abstract
Objective
To evaluate the clinical performance of posterior resin composite restorations regarding experimental and regular adhesive protocols in caries affected-dentin (CAD).
Material and methods
A total of 92 class I and class II cavities with carious lesions were selected and randomly assigned to the following groups: (1) bioactive glass-ceramic powder/two-step etch and rinse system, (2) control/two-step etch and rinse system, (3) bioactive glass-ceramic powder/two-step self-etching system, and (4) control/two-step self-etching system. Two operators carried out the adhesive protocols and restored the cavities with a nano-hybrid resin composite. Participants were followed up at 1 week and 6, 12, and 18 months for clinical evaluation performed by two blinded examiners and calibrated according to FDI criteria. Data were analyzed using Kruskal-Wallis and Dunn tests with a confidence of 95%.
Results
The clinical performance of resin composite restoration was not affected by the experimental use of an adhesive protocol including a bioactive glass-ceramic powder for 18 months post-procedure. However, there was a significant difference between group 2 and group 4 (p < 0.05) for marginal adaptation (18 months). Group 2 was significantly different from group 3 (p < 0.05) for fracture of material/retention (18 months) and marginal adaptation (1 week); group 2 showed a better performance.
Conclusion
Adhesive protocols can alter the clinical performance of posterior restorations in terms of marginal adaptation and the fracture of material/retention in CAD. Clinical significance: Adhesive protocols may influence the success of resin composite restorations in CAD; this is important because failure can lead to caries, re-incidence, and/or clinical re-work.
Similar content being viewed by others
Introduction
Currently, minimally invasive restorative dentistry recognizes partial caries removal in an attempt to conserve tooth structure [1, 2]. Within this approach, only caries-infected dentin is removed, and demineralized caries-affected dentin (CAD), which is remineralizable tissue, is preserved. In this context, restoring CAD substrate is a challenge due not only to its organic and humid intrinsic nature but also because of alterations in chemical, morphological, and mechanical properties that negatively influence the bonding process [3]. Recently, systematic reviews have reported that CAD produces a lower bond strength and durability than sound dentin regardless of the adhesive system used [4, 5].
Even though significant progress has been made in adhesive technology, the major concern for contemporary adhesive systems (i.e., the etch and rinse and self-etching adhesive systems) is related to the degradation of the bonding interface over time [6]. Several in vitro [7,8,9,10] and in vivo [11, 12] studies have shown a significant lower bond strength in both the short and long term, affecting bonding effectiveness. Consequently, adhesion longevity and the clinical performance of resin composite restorations could be compromised. Indeed, clinical shortcomings such as secondary caries, fracture and loss of retention represent the main reasons for failures in posterior direct restorations [13, 14].
Different strategies for operative procedures have been proposed in order to minimize the degradation of the dentin-resin interface and improve its stability [6, 15]. These include the use of some agents such as inhibitors of proteolytic/collagenolytic activity (e.g., chlorhexidine) and protein cross-linking agents [16,17,18,19,20]. Moreover, the dentin remineralization approach using bioactive glass has also been proposed due to the capability of bioactive glass to promote hydroxyapatite precipitation and remineralization of the dentin at the adhesive interface [21,22,23]. Laboratory studies have revealed that the use of bioglass-containing experimental adhesives for the etch and rinse system promotes mineral formation on the hybrid layer, improves mechanical properties, and reduces nanoleakage [24, 25]. In addition, bioglasses used as dentin pretreatment in self-etching bonding can create a bioactive smear layer with therapeutic properties at the adhesive interface, improving the healing ability of the restorations [26].
In particular, researchers have developed Biosilicate, a fully crystallized bioactive glass-ceramic [27]. Due to its high bioactivity, antimicrobial, and mechanical properties, this new biomaterial has been suggested as an option in tissue regeneration applications [27,28,29]. In the field of dentistry, the first studies were conducted for the treatment of dentin hypersensitivity [30, 31]. It seems that the micro-sized particles of Biosilicate react rapidly with the surrounding tissue inside the dentin microchannels, resulting in an effective clinical reduction of hypersensitivity by promoting the occlusion of dentin tubules. Further, in vitro studies have demonstrated that the use of Biosilicate microparticles can favor remineralization in demineralized dental tissues by enhancing mineral gain (e.g., Ca and P) and increasing microhardness [32,33,34]. More recently, an in vitro study has revealed that adhesive systems can lead to higher bond strength values when Biosilicate is used as a dentin pretreatment [35]. Accordingly, the use of bioactive components capable of promoting remineralization of CAD and enhancing tissue repair would be clinically beneficial and serve as a useful clinical treatment strategy to avoid early failures in direct composite restorations.
Even though laboratory studies have supported adhesive procedures for decades, prospective clinical studies corroborating its effectiveness on affected-dentin in composite resin restorations are lacking. Thus, the aim of this study was to evaluate the effect of experimental adhesive protocols, which include the use of microparticles of Biosilicate on CAD, on the clinical performance of composite resin restorations. The null hypothesis stated that there is no difference in the clinical performance of composite resin restorations regardless of the experimental adhesive protocols with the two-step etch and rinse and two-step self-etching adhesive system.
Materials and methods
The study was conducted at the School of Dentistry of Ribeirão Preto - University of São Paulo, Brazil, following the guidelines published by the Consolidated Standards of Reporting Trials (CONSORT). The local ethics committee approved the study (Protocol number 27790214.1.0000.5419). The clinical trial was registered at the U.S. National Institutes of Health (ClinicalTrials.gov) No. NCT02389569.
Study design
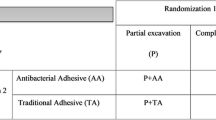
This study was a clinical, randomized, double blind, controlled trial that evaluated the performance of resin composite restorations using Biosilicate (as treatment of CAD) with two different adhesive systems in class I and class II restorations in permanent teeth in an 18-month follow-up. Both participants and examiners were blinded to treatment allocation. Operators were not blinded to treatment allocation, as they knew about the assigned interventions. The factors were the adhesive system on two levels (two-step total etching and two-step self-etching) and dentin treatment on two levels (dentin treatment with Biosilicate and control without dentin treatment). The products and their composition used in this study are described in Table 1.
Sample size
Sample size was calculated regarding the recommendations for conducting controlled clinical studies of dental restorative materials [36]. Based on the expected effect size of the treatment of 15% and using an α level of 0.05 and a power of 80%, the sample size resulted in n = 64 (taking into account four groups and four assessment points) as a minimal sample and considering one restoration per participant as the minimum and four as the maximum. [13]. In addition, in order to redress potential subject dropout during the trial period, we set an increase of 25%.
Participants
The study was carried out at the dental clinics of the Ribeirao Preto School of Dentistry (University of Sao Paulo, Ribeirao Preto, Sao Paulo, Brazil) from December 2014 to December 2016. Two operators evaluated 230 volunteers in order to check if they met the inclusion and exclusion criteria. Evaluation consisted of anamnesis, clinical examination, and radiographic examinations of teeth with evident caries or teeth that were suspected of caries (periapical or bitewing). A vitality test was performed to exclude caries-affected teeth that needed endodontic therapy.
Eligible participants were volunteers of both genders aged between 18 and 50 years. Participants were also required to have at least one active caries lesion in the medium third of the dentin of vital posterior teeth on the occlusal surface (class I) or proximal surface (class II), with no claim of spontaneous pain. Participants also had to be available for recalls. Exclusion criteria were non-vital teeth, teeth with cracks or fractures, an absence of occlusal contact or adjacent teeth, antagonist teeth with porcelain restoration, and teeth with an orthodontic appliance. Furthermore, volunteers with chronic use of medication, systemic diseases, poor oral hygiene, periodontal disease, and parafunctional habits and pregnant or lactating mothers and smokers were excluded. The selected participants received verbal and written information about the study and signed the term of consent form prior to any restorative procedure.
Intervention
Two operators performed all restoration procedures. To standardize the clinical treatment protocol for each group, operators were previously calibrated in a clinical training session. All restorations were performed after local anesthesia with 3% mepivacaine solution (Mepisv, Nova DFL, Rio de Janeiro, RJ, Brazil) and dental prophylaxis with pumice and water. Then, the operative field was isolated with a rubber dam (Duflex, SSWhite, Juiz de Fora, MG, Brazil).
After proper isolation, caries tissue removal was performed using the mechanical method. The caries lesions were accessed using round diamond burs 1012–1014 (KG Sorensen, Barueri, SP, Brazil) that were compatible with the lesion size and mounted in a high-speed turbine under constant water-cooling. In class II cavities, the access was performed with its orientation towards to the proximal surface. To protect the adjacent tooth, a metal band was inserted in the proximal area.
The cavity was prepared according to the select removal of carious tissue approach and based on clinical (tactile) criteria [37, 38]. On the pulpal wall, only softened dentin was removed; leathery dentin (which can still be easily without much force being required) and firm dentin (which is physically resistant to hand instrument) were left. On the axial walls, carious removal tissue was cleaned to hard dentin (similar to sound dentin). No additional retention or beveling on the enamel cavosurface margins was performed.
Subsequently, selected teeth were randomly assigned to one of the following adhesive protocols:
-
Group 1 (two-step etch and rinse + Biosilicate): 35% phosphoric acid gel (Etchant gel, VITAZahnfabrik) was applied on the enamel (15 s) and then onto the dentin surface (15 s). The acid gel was rinsed for 10 s, and excess water was removed using a sterile cotton pellet. Immediately after blotting, operators applied Biosilicate powder on the dentin surface using a microapplicator (~ 0.04 g), and the surface was scrubbed for 10 s. Then, 2–3 consecutive coats of Adper™ Single Bond 2 (3 M Oral Care, Sumaré, SP, Brazil) were applied to the etched enamel and dentin (15 s). The solvents were evaporated with a gentle stream of dry air (5 s). Light curing was performed for 20 s using an LED dental curing light unit of 800 mW/cm2 (Lec Plus, MMO, Sao Carlos, SP, Brazil).
-
Group 2 (two-step etch and rinse-control group): as described in group 1 but without the application of Biosilicate powder.
-
Group 3 (two-step self-etching adhesive + Biosilicate): dental tissues were etched with AdheSe Primer (AdheSE, IvoclarVivadent, Barueri, SP, Brazil) initiated at the enamel surface (15 s) and then at the dentin (15 s). The excess primer was dispersed with a strong stream of air until no further adhesive movement was noticeable. Then, operators applied Biosilicate powder on the dentin surface using a microapplicator (~ 0.04 g) and scrubbed the surface for 10 s. They then applied AdheSe Bond (15 s), beginning at the dentin and dispersing the bond with a stream of air to avoid pooling. Light curing was performed for 20 s using an LED dental curing light unit of 800 mW/cm2 (Lec Plus, MMO, Sao Carlos, SP, Brazil).
-
Group 4 (two-step self-etching adhesive-control group): as described in group 3 but without the application of Biosilicate powder.
After adhesive protocols, a resin composite (IPS Empress DirectIvoclar, Vivadent) was placed in 2-mm increments using the oblique layering technique and light-cured for 20 s using an LED dental curing light unit of 800 mW/cm2 (Lec Plus, MMO, Sao Carlos, SP, Brazil). Finally, occlusion was verified with articulating paper (Contacto, Angelus, PR, Brazil) and adjusted using diamond finishing burs (KG, Sorensen, Cotia, SP, Brazil). Polishing was accomplished with silicon polishers and silicon carbide brushes (Kit Viking, KG Sorensen, Cotia, SP, Brazil). Abrasive strips (Injecta, Itajai, SC, Brazil) were used for interproximal surface finishing.
Clinical evaluation
All restorations were clinically evaluated at baseline (1 week after the restoration placement) and after 6, 12, and 18 months. Two calibrated examiners who were not involved with the restoration procedures and were blinded to the treatment conditions independently evaluated each restoration according to FDI criteria [36, 39]. These included the following categories: esthetic properties (surface staining), functional properties (fracture and retention and marginal adaptation), and biological properties (postoperative hypersensitivity and recurrent caries). The examiners indicated each criterion with scores as follows: 1 = clinically excellent, 2 = clinically good, 3 = clinically satisfactory, 4 = clinically unsatisfactory, and 5 = clinically poor. Scores 1, 2, and 3 represented “clinically acceptable restorations,” while scores 4 and 5 represented “clinically unacceptable restorations” indicating the need for repair or replacement of the restoration.
For calibration, both examiners were trained in a clinical setting on patients for each criterion. They also used an online calibration system using FDI criteria (www.e-calib.info), where they trained on each criterion using high-quality photographs of clinical cases. A calibration at ≥ 85% was necessary prior to starting the evaluation [39].
Clinical evaluation was carried out using a mouth mirror, dental probe, and periodontal probe. For the evaluation of postoperative sensitivity, a cold air stimulus from a triple syringe was placed approximately 2.0 cm from the restored tooth surface. To assist in the evaluation process, restorations were documented with intraoral photographs at each recall appointment.
Randomization
The number of restorations was limited to one restoration per group per participant; thus, each participant received at least one restoration and at most four restorations. Simple randomization was performed, taking into consideration that (1) the observation unit was the restoration and (2) the selected teeth per participant (one to four restorations were allowed per participant) should be allocated in different groups. The operators had a box with 04 envelopes containing inside them numbers from 1 to 4 (each number in each envelope). For each tooth selected to be included in the study, one envelope was taken out of the box (the envelope removed was not inserted in the box again for the same patient), by the operator and the number inside the box indicated in which group that tooth was allocated. This procedure occurred for each tooth (minimum one and maximum four) and each patient.
Statistical analysis
Statistical analysis was initiated with Cohen’s Kappa statistic to test inter-examiner agreement. Descriptive analysis was used to describe the distribution of each criterion. The statistical unit was the restored tooth. Differences between groups in each assessment point were evaluated using Kruskal-Wallis and Dunn’s multiple comparison non-parametric tests with a significance level of 5%. All analyses were performed using IBM SPSS Statics version 20.0 for Windows (SPSS Inc., Armonk, NY, USA).
Results
Study population
Study subject recruitment and clinical examination were conducted from December 2014 to June 2015. A total of 230 volunteers were screened for eligibility, and 66 met the inclusion criteria that were selected for the study. Twenty-eight (42.4%) of the volunteers were men, and 38 (57.6%) were women. Participants were aged between 18 and 50 years (mean = 32.2 ± 7.8). Ninety-two restorations were placed in the selected participants that were randomly allocated into the groups. Each volunteer received at least one and no more than four restorations. Table 2 shows the baseline details of the subjects and distribution for the restored cavities.
Figure 1 shows the CONSORT flow diagram, which details the number of restorations allocated for intervention, the number lost, and the number of restorations available for analysis at each follow-up. At the end of the study, the recall rates for subjects and restorations were 72.7% and 77.2%, respectively. The Kappa statistic value indicated good and excellent agreement between the examiners (0.8–1.0) for all criteria evaluated.
Descriptive analysis
For marginal staining, all restorations received scores of 1, 2, or 3, i.e., clinically acceptable, with the exception of two restorations from group 3 (at 12 and 18 months), which received score of 5. For fracture and retention and marginal adaptation, most of the restorations received scores of 1 or 2; however, some restorations were scored as 4 or 5, i.e., clinically inacceptable, mainly for groups 3 and 4. For postoperative hypersensitivity and recurrence of caries, all restorations received scores of 1, 2, or 3. Only one restoration from group 1 received a score of 4 for postoperative hypersensitivity at 1 week. One restoration from group 4 also received a score of 4 for recurrence of caries at 6 months (Table 3).
Comparison analysis
For all criteria evaluated, no significant difference (p > 0.05) was found between experimental groups and their respective control groups (1 × 2 and 3 × 4). The results showed significant differences between the control groups (2 × 4) for marginal adaptation at the 18-month follow-up (p value 0.023). This difference was found within clinically acceptable parameters. Additionally, group 2 was significantly different from group 3 (p value 0.038) for marginal adaptation at 1 week and for fracture and retention at 18 months (p value 0.044) (Table 4).
Discussion
In an attempt to stabilize the dentin-resin interface, repair demineralized tissue and recover the biomechanical properties of CAD, new strategies in adhesion procedures were proposed, including remineralizing operative approaches with bioactive glass materials [23,24,25,26, 40, 41]. In this context, there is some evidence that the use of Biosilicate could be an option for improving the bond strength of adhesive materials for dental substrates, which suggests to be beneficial for the success of direct restorations [35, 42, 43]. Thus, this study aimed to evaluate the clinical performance of posterior resin composite restorations through experimental adhesive protocols that include the use of microparticles of Biosilicate on CAD.
In the present study, the use of Biosilicate microparticles in the adhesive protocol does not alter the clinical performance of the resin composite restoration at any assessment point. Thus, based on the results, we accept the null hypotheses (H0) that there would be no difference in the clinical performance of composite resin restorations regardless of the use of bioactive glass ceramic (Biosilicate) in adhesive protocols.
Concerning the adhesive protocol with the two-step etch and rinse adhesive system, both the experimental (group 1) and regular (group 2) groups generally showed an acceptable clinical performance for all categories (esthetic, functional, and biological) throughout the period of study. Previous randomized clinical trials have corroborated the satisfactory clinical performance observed in this study [44,45,46]. As mentioned, in our study, the clinical success of composite restorations does not seem to be compromised by the use of a two-step etch and rinse adhesive. Nonetheless, our results may be attributed to the short period of follow-up (18 months).
The clinical performance of restorations from an experimental group that used Biosilicate microparticles on CAD was no different from that of the regular protocol with two-step etch and rinse adhesive. In this regard, there is evidence that the use of Biosilicate after acid etching positively influences the bond strength to dentin and promotes a high mineral formation (i.e., calcium and phosphorus ions) contributing to the success and durability of composite resin restorations [35].
Concerning the adhesive protocol with the two-step self-etching adhesive system, no difference was observed between the experimental (group 3) and regular (group 4) protocols at any assessment point. Most restorations were clinically acceptable for all criteria (esthetic, functional and biological). However, unlike what was expected, five restorations (25%) from group 4 and nine (37%) from group 3 showed unacceptable clinical performance for the fracture of material criterion, which were considered clinical failures.
Other clinical short-term trials (1–2 years) have noted the satisfactory clinical performance of two-step self-etching adhesives, and they have also reported no failures in that period [45, 47]. Nevertheless, these studies included in the sample posterior teeth indicated for direct restoration for several reasons (e.g., caries lesions, fracture, esthetic reasons, etc.) and not exclusively for active caries lesions.
Additionally, for the fracture of material and retention criterion, there was a significant difference between the experimental protocol for self-etching adhesive (group 3) and the regular protocol for the etch and rinse adhesive (group 2) at the 18-month follow-up. The latter showed better clinical performance, as all restorations from this group received a score of 1 (clinically excellent). In contrast, group 3 varied in scores from 1 to 5 (clinically excellent to clinically poor) mainly for marginal staining, fracture and retention, and marginal adaptation. Probably, the difference between these two groups could be most likely related to the different etching protocols rather than to the use of the bioactive glass.
On the other hand, the results of this study indicated that there were significant differences between composite restorations that were used for different adhesive protocols for marginal adaptation. At the 1-week follow-up, a difference was found between group 2 and group 3, while at the 18-month follow-up, a difference was found between group 2 and group 4. In both cases, these differences occurred within clinically acceptable scores (1 to 3), and group 2 showed better clinical performance.
Alterations in marginal adaptation are associated with a thermal or mechanical stress on restorations in the oral environment as well as viscoelastic properties of the restoring material [48, 49]. Moreover, some alterations in marginal integrity may occur during the polymerization process due to a volumetric shrinkage of composite resins that depend on composition. In our study, IPS Empress Direct was used for all adhesive protocols. This nano-hybrid composite contains 75–79% fillers (wt.%) and presents reduced volumetric shrinkage [50]. Therefore, variations in marginal adaptation related to the resin used are unlikely.
Additionally, the influence of the adhesive system on the marginal adaptation of posterior composite restoration has been clinically reported and has shown divergent results. A review discussed that the clinical performance of self-etching adhesive was satisfactory in all criteria except for marginal adaptation, which showed inferior results compared to the etch and rinse adhesive system [51]. Further clinical studies reported some clinical shortcomings regarding marginal adaptation even in the first months when the one-step self-etching adhesives was used and significant deterioration after 4 years after the two-step-self-etching adhesive was used [44, 52]. Conversely, other clinical trials have indicated that there is no difference in the clinical performance of posterior composite restorations, even for marginal adaptation [45, 46, 53]. In this study, even though the differences detected for the marginal adaptation category among the adhesive protocols were within the clinically acceptable parameters, these alterations could lead to failure over time.
The results obtained in this study were based on FDI criteria that include three parameters (esthetic, functional, and biologic). Despite the fact that these criteria have been recently introduced, several studies have shown that they are more strict and sensitive to small variations in clinical outcomes than USPHS criteria [54, 55]. Moreover, in this study, all intervention procedures were performed by two operators, who were trained and calibrated with established adhesive protocols. Participants were also selected according to rigorous eligibility criteria. The low number of operators and the rigorous criteria applied in the selection of study subjects can be seen as limitations of this study.
Other limitations were the short follow-up period and relatively low recall rate. First, the recall rate after 18 months was 72.7%. Even though a great effort to maintain the number of participants up to the final assessment point was made, participants were not available for recalls. Second, the clinical performance of restorations was assessed in the short-term (18 months). Therefore, different results may be found during a longer follow-up period as failures are commonly reported after 3 years [13, 14].
Additional clinical studies should be conducted in order to compare and investigate the efficacy of new restorative strategies involving bioactive glasses. On the other hand, the execution of ex-vivo studies would be an interesting alternative to reveal important characteristics of the adhesive interface exposed to the oral environment.
Conclusions
The clinical performance of experimental adhesive protocols was similar to controls.
Etch and rinse adhesive protocol (group 2) showed higher clinical success of resin composite restoration in terms of marginal adaptation and fractures in caries-affected dentin after 18 months.
References
Tyas MJ, Anusavice KJ, Frencken JE, Mount GJ (2000) Minimal intervention dentistry--a review. FDI Commission Project 1-97. Int Dent J 50:1–12. https://doi.org/10.1111/j.1875-595X.2000.tb00540.x
FDI policy statement on Minimal Intervention Dentistry (MID) for managing dental caries: Adopted by the general assembly: September 2016, Poznan, Poland (2017). Int Dent J 67:6–7. https://doi.org/10.1111/idj.12308
Nakajima M, Kunawarote S, Prasansuttiporn T, Tagami J (2011) Bonding to caries-affected dentin. Jpn Dent Sci Rev 47:102–114. https://doi.org/10.1016/j.jdsr.2011.03.002
Pinna R, Maioli M, Eramo S, Mura I, Milia E (2015) Carious affected dentine: its behaviour in adhesive bonding. Aust Dent J 60:276–293. https://doi.org/10.1111/adj.12309
Ekambaram M, Yiu CKY, Matinlinna JP (2015) Bonding of resin adhesives to caries-affected dentin – a systematic review. Int J Adhes Adhes 61:23–34. https://doi.org/10.1016/j.ijadhadh.2015.04.011
Perdigão J, Reis A, Loguercio AD (2013) Dentin adhesion and MMPs: a comprehensive review. J Esthet Restor Dent 25:219–241. https://doi.org/10.1111/jerd.12016
Erhardt MC, Toledano M, Osorio R, Pimenta LA (2008) Histomorphologic characterization and bond strength evaluation of caries-affected dentin/resin interfaces: effects of long-term water exposure. Dent Mater 24:786–798. https://doi.org/10.1016/j.dental.2007.09.007
Armstrong SR, Vargas MA, Chung I, Pashley DH, Campbell JA, Laffoon JE, Qian F (2004) Resin-dentin interfacial ultrastructure and microtensile dentin bond strength after five-year water storage. Oper Dent 29:705–712. https://doi.org/10.3290/j.jad.a17543
Hashimoto M, Fujita S, Nagano F, Ohno H, Endo K (2010) Ten-years degradation of resin-dentin bonds. Eur J Oral Sci 118:404–410. https://doi.org/10.1111/j.1600-0722.2010.00744.x
Anchieta RB, Machado LS, Martini AP, Santos PH, Giannini M, Janal M et al (2015) Effect of long-term storage on nanomechanical and morphological properties of dentin-adhesive interfaces. Dent Mater 31:141–153. https://doi.org/10.1016/j.dental.2014.11.010
Hashimoto M, Ohno H, Kaga M, Endo K, Sano H, Oguchi H (2000) In vivo degradation of resin-dentin bonds in humans over 1 to 3 years. J Dent Res 79:1385–1391. https://doi.org/10.1177/00220345000790060601
Tam L, Jokstad A (2010) The bond between resin composite restorations and dentin may degrade in the mouth over time. J Evid Based Dent Pract 10:21–22. https://doi.org/10.1016/j.jebdp.2009.11.011
Opdam NJ, van de Sande FH, Bronkhorst E, Cenci MS, Bottenberg P, Pallesen U et al (2014) Longevity of posterior composite restorations: a systematic review and meta-analysis. J Dent Res 93:943–949. https://doi.org/10.1177/0022034514544217
Ástvaldsdóttir Á, Dagerhamn J, van Dijken JW, Naimi-Akbar A, Sandborgh-Englund G, Tranæus S et al (2015) Longevity of posterior resin composite restorations in adults – a systematic review. J Dent 43:934–954. https://doi.org/10.1016/j.jdent.2015.05.001
Reis A, Carrilho M, Breschi L, Loguercio AD (2013) Overview of clinical alternatives to minimize the degradation of the resin-dentin bonds. Oper Dent 38:E1–E25. https://doi.org/10.2341/12-258-LIT
Stanislawczuk R, Pereira F, Munoz MA, Luque I, Farago PV, Reis A et al (2014) Effects of chlorhexidine-containing adhesives on the durability of resin-dentine interfaces. J Dent 42:39–47. https://doi.org/10.1016/j.jdent.2013.11.002
Loguercio AD, Hass V, Gutierrez MF, Luque-Martinez IV, Szezs A, Stanislawczuk R, Bandeca MC, Reis A (2016) Five-year effects of chlorhexidine on the in vitro durability of resin/dentin interfaces. J Adhes Dent 18:35–42. https://doi.org/10.3290/j.jad.a35514
Zheng P, Zaruba M, Attin T, Wiegand A (2015) Effect of different matrix metalloproteinase inhibitors on microtensile bond strength of an etch-and-rinse and a self-etching adhesive to dentin. Oper Dent 40:80–86. https://doi.org/10.2341/13-162-L
Macedo GV, Yamauchi M, Bedran-Russo AK (2009) Effects of chemical cross-linkers on caries-affected dentin bonding. J Dent Res 88:1096–1100. https://doi.org/10.1177/0022034509351001
Chiang YS, Chen YL, Chuang SF, Wu CM, Wei PJ, Han CF, Lin JC, Chang HT (2013) Riboflavin-ultraviolet-A-induced collagen cross-linking treatments in improving dentin bonding. Dent Mater 29:682–692. https://doi.org/10.1016/j.dental.2013.03.015
Hernández M, Cobb D, Swift EJ (2014) Current strategies in dentin remineralization. J Esthet Restor Dent 26:139–145. https://doi.org/10.1111/jerd.12095
Zhong B, Peng C, Wang G, Tian L, Cai Q, Cui F (2015) Contemporary research findings on dentine remineralization. J Tissue Eng Regen Med 9:1004–1016. https://doi.org/10.1002/term.1814
Sauro S, Pashley DH (2016) Strategies to stabilise dentine-bonded interfaces through remineralising operative approaches – state of the art. Int J Adhes Adhes 69:39–57. https://doi.org/10.1016/j.ijadhadh.2016.03.014
Sauro S, Osorio R, Watson TF, Toledano M (2012) Therapeutic effects of novel resin bonding systems containing bioactive glasses on mineral-depleted areas within the bonded-dentine interface. J Mater Sci Mater Med 23:1521–1532. https://doi.org/10.1007/s10856-012-4606-6
Profeta AC, Mannocci F, Foxton RM, Thompson I, Watson TF, Sauro S (2012) Bioactive effects of a calcium/sodium phosphosilicate on the resin-dentine interface: a microtensile bond strength, scanning electron microscopy, and confocal microscopy study. Eur J Oral Sci 120:353–362. https://doi.org/10.1111/j.1600-0722.2012.00974.x
Sauro S, Watson TF, Thompson I, Banerjee A (2012) One-bottle self-etching adhesives applied to dentine air-abraded using bioactive glasses containing polyacrylic acid: an in vitro microtensile bond strength and confocal microscopy study. J Dent 40:896–905. https://doi.org/10.1016/j.jdent.2012.07.004
Crovace MC, Souza MT, Chinaglia CR, Peitl O, Zanotto ED (2016) Biosilicate® — a multipurpose, highly bioactive glass-ceramic. In vitro, in vivo and clinical trials. J Non-Crys Solids 432 Part A 432:90–110. https://doi.org/10.1016/j.jnoncrysol.2015.03.022
Renno AC, Bossini PS, Crovace MC, Rodrigues AC, Zanotto ED, Parizotto NA (2013) Characterization and in vivo biological performance of biosilicate. Biomed Res Int 2013:141427–141427. https://doi.org/10.1155/2013/141427
Martins CH, Carvalho TC, Souza MG, Ravagnani C, Peitl O, Zanotto ED et al (2011) Assessment of antimicrobial effect of Biosilicate® against anaerobic, microaerophilic and facultative anaerobic microorganisms. J Mater Sci Mater Med 22:1439–1446. https://doi.org/10.1007/s10856-011-4330-7
Tirapelli C, Panzeri H, Soares RG, Peitl O, Zanotto ED (2010) A novel bioactive glass-ceramic for treating dentin hypersensitivity. Braz Oral Res 24:381–387. https://doi.org/10.1590/S1806-83242010000400002
Tirapelli C, Panzeri H, Lara EH, Soares RG, Peitl O, Zanotto ED (2011) The effect of a novel crystallised bioactive glass-ceramic powder on dentine hypersensitivity: a long-term clinical study. J Oral Rehabil 38:253–262. https://doi.org/10.1111/j.1365-2842.2010.02157.x
Pinheiro H, Lopes B, Klautau E, Cardoso J, Silva B, Cardoso P (2010) Influence of bioactive materials used on the dentin surface whitened with carbamide peroxide 16%. Mat Res 13:273–278. https://doi.org/10.1590/S1516-14392010000200024
Pintado-Palomino K, Tirapelli C (2015) The effect of home-use and in-office bleaching treatments combined with experimental desensitizing agents on enamel and dentin. Eur J Dent 9:66–73. https://doi.org/10.4103/1305-7456.149645
Rastelli A, Nicolodelli G, Romano R, Milori D, Perazzoli I, Ferreira E et al (2016) After bleaching enamel remineralization using a bioactive glass-ceramic (BioSilicate®). Biomed glasses 2:1–9. https://doi.org/10.1515/bglass-2016-0001
de Morais RC, Silveira RE, Chinelatti MA, Pires-de-Souza FCP (2016) Biosilicate as a dentin pretreatment for total-etch and self-etch adhesives: in vitro study. Int J Adhes Adhes 70:271–276. https://doi.org/10.1016/j.ijadhadh.2016.07.007
Hickel R, Roulet JF, Bayne S, Heintze SD, Mjör IA, Peters M et al (2007) Recommendations for conducting controlled clinical studies of dental restorative materials. Science committee project 2/98--FDI world dental federation study design (part I) and criteria for evaluation (part II) of direct and indirect restorations including onlays and partial crowns. J Adhes Dent 9 Suppl 1:121–147. https://doi.org/10.3290/j.jad.a19262
Innes NP, Frencken JE, Bjørndal L, Maltz M, Manton DJ, Ricketts D et al (2016) Managing carious lesions: consensus recommendations on terminology. Adv Dent Res 28:49–57. https://doi.org/10.1177/0022034516639276
Massara ML, Alves JB, Brandão PR (2002) Atraumatic restorative treatment: clinical, ultrastructural and chemical analysis. Caries Res 36:430–436. https://doi.org/10.1159/000066534
Hickel R, Peschke A, Tyas M, Mjör I, Bayne S, Peters M, Hiller KA, Randall R, Vanherle G, Heintze SD (2010) FDI World Dental Federation: clinical criteria for the evaluation of direct and indirect restorations-update and clinical examples. Clin Oral Investig 14:349–366. https://doi.org/10.1007/s00784-010-0432-8
Profeta AC, Mannocci F, Foxton R, Watson TF, Feitosa VP, De Carlo B et al (2013) Experimental etch-and-rinse adhesives doped with bioactive calcium silicate-based micro-fillers to generate therapeutic resin-dentin interfaces. Dent Mater 29:729–741. https://doi.org/10.1016/j.dental.2013.04.001
Sauro S, Watson TF, Thompson I, Toledano M, Nucci C, Banerjee A (2012) Influence of air-abrasion executed with polyacrylic acid-Bioglass 45S5 on the bonding performance of a resin-modified glass ionomer cement. Eur J Oral Sci 120:168–177. https://doi.org/10.1111/j.1600-0722.2012.00939.x
Pires-de-Souza FC, de Marco FF, Casemiro LA, Panzeri H (2007) Desensitizing bioactive agents improves bond strength of indirect resin-cemented restorations: preliminary results. J Appl Oral Sci 15:120–126. https://doi.org/10.1590/S1678-77572007000200009
Osorio E, Fagundes T, Navarro MF, Zanotto ED, Peitl O, Osorio R, Toledano-Osorio M, Toledano M (2015) A novel bioactive agent improves adhesion of resin-modified glass-ionomer to dentin. J Adhes Sci Technol 29:1543–1552. https://doi.org/10.1080/01694243.2015.1030897
Boeckler A, Boeckler L, Eppendorf K, Schaller HG, Gernhardt CR (2012) A prospective, randomized clinical trial of a two-step self-etching vs two-step etch-and-rinse adhesive and SEM margin analysis: four-year results. J Adhes Dent 14:585–592. https://doi.org/10.3290/j.jad.a27796
Sundfeld RH, Scatolin RS, Oliveira FG, Machado LS, Alexandre RS, Sundefeld ML (2012) One-year clinical evaluation of composite restorations in posterior teeth: effect of adhesive systems. Oper Dent 37:E1–E8. https://doi.org/10.2341/10-375-C
Sundfeld RH, Machado LS, Pita DS, Franco LM, Sundfeld D, Sundefeld ML et al (2016) Three-year clinical evaluation of class I restorations in posterior teeth. Effects of two adhesive systems. Compend Contin Educ Dent 37:e1–e4
Boeckler A, Schaller HG, Gernhardt CR (2012) A prospective, double-blind, randomized clinical trial of a one-step, self-etch adhesive with and without an intermediary layer of a flowable composite: a 2-year evaluation. Quintessence Int 43:279–286
Wattanawongpitak N, Yoshikawa T, Burrow MF, Tagami J (2006) The effect of bonding system and composite type on adaptation of different C-factor restorations. Dent Mater J 25:45–50. https://doi.org/10.4012/dmj.25.45
Wattanawongpitak N, Yoshikawa T, Burrow MF, Tagami J (2007) The effect of thermal stress on bonding durability of resin composite adaptation to the cavity wall. Dent Mater J 26:445–450. https://doi.org/10.4012/dmj.25.45
Sideridou ID, Karabela MM, Vouvoudi E (2011) Physical properties of current dental nanohybrid and nanofill light-cured resin composites. Dent Mater 27:598–607. https://doi.org/10.1016/j.dental.2011.02.015
Krithikadatta J (2010) Clinical effectiveness of contemporary dentin bonding agents. J Conserv Dent 13:173–183. https://doi.org/10.4103/0972-0707.73376
Manchorova-Veleva NA, Vladimirov SB, Keskinova Dcapital AC (2015) Clinical effect of dental adhesive on marginal integrity in class I and class II resin-composite restorations. Folia Med (Plovdiv) 57:250–256. https://doi.org/10.1515/folmed-2015-0046
Boushell LW, Heymann HO, Ritter AV, Sturdevant JR, Swift EJ Jr, Wilder AD Jr et al (2016) Six-year clinical performance of etch-and-rinse and self-etch adhesives. Dent Mater 32:1065–1072. https://doi.org/10.1016/j.dental.2016.06.003
Mena-Serrano A, Kose C, De Paula EA, Tay LY, Reis A, Loguercio AD et al (2013) A new universal simplified adhesive: 6-month clinical evaluation. J Esthet Restor Dent 25:55–69. https://doi.org/10.1111/jerd.12005
Perdigao J, Kose C, Mena-Serrano AP, De Paula EA, Tay LY, Reis A et al (2014) A new universal simplified adhesive: 18-month clinical evaluation. Oper Dent 39:113–127. https://doi.org/10.2341/13-045-C
Funding
This study was funded by the São Paulo Research Foundation - FAPESP (grant number 2010/12032-6) and Center for Research, Teaching and Innovation in Glass (CeRTEV)/CEPID grant number 2013/07793-6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were conducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Pintado-Palomino, K., de Almeida, C.V.V.B., da Motta, R.J.G. et al. Clinical, double blind, randomized controlled trial of experimental adhesive protocols in caries-affected dentin. Clin Oral Invest 23, 1855–1864 (2019). https://doi.org/10.1007/s00784-018-2615-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-018-2615-7