Abstract
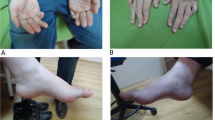
Hereditary spastic paraplegias (HSP) are a heterogeneous group of neurodegenerative disorders leading to progressive spasticity of the lower limbs. Here, we describe clinical and genetic features in an Italian family affected by autosomal recessive HSP (ARHSP) with mental impairment and thin corpus callosum (TCC). In both affected subjects, genetic analysis revealed the presence of a homozygous small deletion (733_734delAT) leading to a frameshift (M245VfsX) within the coding region of SPG11 gene, encoding spatacsin. This finding is the first independent confirmation that spatacsin loss of function mutations cause ARHPS-TCC.


Similar content being viewed by others
References
Tallaksen CM, Durr A, Brice A (2001) Recent advances in hereditary spastic paraplegia. Curr Opin Neurol 14(4):457–63 (Review).
Iwabuchi K, Kubota Y, Hanihara T, Nagatomo H (1994) Three patients of complicated form of autosomal recessive hereditary spastic paraplegia associated with hypoplasia of the corpus callosum. No To Shinkei 46:941–947
Martinez Murillo F, Kobayashi H, Pegoraro E, Galluzzi G, Creel G, Mariani C, Farina E, Ricci E, Alfonso G, Pauli RM, Hoffman EP (1999) Genetic localization of a new locus for recessive familial spastic paraparesis to 15q13–15. Neurology 53:50–56
Casali C, Valente EM, Bertini E, Montagna G, Criscuolo C, De Michele G, Villanova M, Damiano M, Pierallini A, Brancati F, Scarano V, Tessa A, Cricchi F, Grieco GS, Muglia M, Carella M, Martini B, Rossi A, Amabile GA, Nappi G, Filla A, Dallapiccola B, Santorelli FM (2004) Clinical and genetic studies in hereditary spastic paraplegia with thin corpus callosum. Neurology 62:262–268
Winner B, Uyanik G, Gross C, Lange M, Schulte-Mattler W, Schuierer G, Marienhagen J, Hehr U, Winkler J (2004) Clinical progression and genetic analysis in hereditary spastic paraplegia with thin corpus callosum in spastic gait gene 11 (SPG11). Arch Neurol 61:117–121
Olmez A, Uyanik G, Ozgul RK, Gross C, Cirak S, Elibol B, Anlar B, Winner B, Hehr U, Topaloglu H, Winkler J (2006) Further clinical and genetic characterization of SPG11: hereditary spastic paraplegia with thin corpus callosum. Neuropediatrics 37:59–66
Lossos A, Stevanin G, Meiner V, Argov Z, Bouslam N, Newman JP, Gomori JM, Klebe S, Lerer I, Elleuch N, Silverstein S, Durr A, Abramsky O, Ben Nariah Z, Brice A (2006) Hereditary spastic paraplegia with thin corpus callosum: reduction of the SPG11 interval and evidence for further genetic heterogeneity. Arch Neurol 63:756–60
Stevanin G, Montagna G, Azzedine H, Valente EM, Durr A, Scarano V, Bouslam N, Cassandrini D, Denora PS, Criscuolo C, Belarbi S, Orlacchio A, Jonveaux P, Silvestri G, Hernandez AM, De Michele G, Tazir M, Mariotti C, Brockmann K, Malandrini A, van der Knapp MS, Neri M, Tonekaboni H, Melone MA, Tessa A, Dotti MT, Tosetti M, Pauri F, Federico A, Casali C, Cruz VT, Loureiro JL, Zara F, Forlani S, Bertini E, Coutinho P, Filla A, Brice A, Santorelli FM (2006) Spastic paraplegia with thin corpus callosum: description of 20 new families, refinement of the SPG11 locus, candidate gene analysis and evidence of genetic heterogeneity. Neurogenetics 7:149–56
Shibasaki Y, Tanaka H, Iwabuchi K, Kawasaki S, Kondo H, Uekawa K, Ueda M, Kamiya T, Katayama Y, Nakamura A, Takashima H, Nakagawa M, Masuda M, Utsumi H, Nakamuro T, Tada K, Kurohara K, Inoue K, Koike F, Sakai T, Tsuji S, Kobayashi H (2000) Linkage of autosomal recessive hereditary spastic paraplegia with mental impairment and thin corpus callosum to chromosome 15q13–15. Ann Neurol 48:108–112
Stevanin G, Santorelli FM, Azzedine H, Coutinho P, Chomilier J, Denora PS, Martin E, Ouvrard-Hernandez AM, Tessa A, Bouslam N, Lossos A, Charles P, Loureiro JL, Elleuch N, Confavreux C, Cruz VT, Ruberg M, Leguern E, Grid D, Tazir M, Fontaine B, Filla A, Bertini E, Durr A, Brice A (2007) Mutations in SPG11, encoding spatacsin, are a major cause of spastic paraplegia with thin corpus callosum. Nat Genet 39(3):366–372
Maquat LE (2004) Nonsense-mediated mRNA decay: splicing, translation and mRNP dynamics. Nat Rev Mol Cell Biol 5:89–99
Acknowledgements
The authors are grateful to the patients and family members for their kind cooperation in this study. This work was supported by research grants to S.C. and N.B. from MIUR (Ministero Istruzione Università e Ricerca Scientifica) PRIN 2006 “Molecular pathogenesis of the motoneuron disorders as a tool for the identification of novel biomolecular and cellular therapeutic targets” and by a grant to G.P.C. from Italian Ministry of Health, “Progetti di Ricerca ex. Art. 56, 2005.” “The motor neuron disease: molecular and cellular pathways in neuronal and muscular degeneration as a cause of clinical and genetic heterogeneity.” The work of G.S. was supported by a “Groupement d’Intéret Scientifique, France” grant. We thank Prof. Alexis Brice for critical reading of the manuscript. We wish to thank the “Associazione Amici del Centro Dino Ferrari” for their support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Roberto Del Bo and Alessio Di Fonzo, These two authors equally contributed to the present work.
Rights and permissions
About this article
Cite this article
Del Bo, R., Di Fonzo, A., Ghezzi, S. et al. SPG11: a consistent clinical phenotype in a family with homozygous Spatacsin truncating mutation. Neurogenetics 8, 301–305 (2007). https://doi.org/10.1007/s10048-007-0095-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10048-007-0095-z