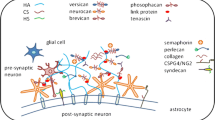
Abstract
The brain extracellular matrix (ECM) is involved in crucial processes of neural support, neuronal and synaptic plasticity, extrasynaptic transmission, and neurotransmission. ECM is a tridimensional fibrillary meshwork composed of macromolecules that determine its bioactivity and give it unique characteristics. The characterization of the brain ECM is critical to understand its dynamic in SZ. Thus, a comparative study was developed with 71 patients with schizophrenia (SZ) and 70 healthy controls. Plasma of participants was analysed by label-free liquid chromatography–tandem mass spectrometry, and the results were validated using the classical western blot method. Lastly, immunostaining of post-mortem human brain tissue was performed to analyse the distribution of the brain ECM proteins by confocal microscopy. The analysis identified four proteins: fibronectin, lumican, nidogen-1, and secreted protein acidic and rich in cysteine (SPARC) as components of the brain ECM. Statistical significance was found for fibronectin (P = 0.0166), SPARC (P = 0.0003), lumican (P = 0.0012), and nidogen-1 (P < 0.0001) that were decreased in the SZ group. Fluorescence imaging of prefrontal cortex (PFC) sections revealed a lower expression of ECM proteins in SZ. Our study proposes a pathophysiological dysregulation of proteins of the brain ECM, whose abnormal composition leads to a progressive neuronal impairment and consequently to neurodegenerative processes due to lack of neurophysiological support and dysregulation of neuronal homeostasis. Moreover, the brain ECM and its components are potential pharmacological targets to develop new therapeutic approaches to treat SZ.



Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Barros CS, Franco SJ, Müller U (2011) Extracellular Matrix: Functions in the nervous system. Cold Spring Harb Perspect Biol 3:1–24. https://doi.org/10.1101/cshperspect.a005108
Benias PC, Wells RG, Sackey-Aboagye B et al (2018) Structure and distribution of an unrecognized interstitium in human tissues. Sci Rep 8:1–8. https://doi.org/10.1038/s41598-018-23062-6
Berretta S (2012) Extracellular Matrix Abnormalities in Schizophrenia. Neuropharmacology 62:1584–1597. https://doi.org/10.2217/FON.09.6.Dendritic
Bikbaev A, Frischknecht R, Heine M (2015) Brain extracellular matrix retains connectivity in neuronal networks. Sci Rep 5:1–12. https://doi.org/10.1038/srep14527
Bonneh-Barkay D, Wiley CA (2009) Brain Extracellular Matrix in Neurodegeneration. Brain Pathol 19:573–585. https://doi.org/10.1111/j.1750-3639.2008.00195.x
Bradshaw AD, Sage EH (2001) SPARC, a matricellular protein that functions in cellular differentiation and tissue response to injury. J Clin Investig 107:1049–1054
Byron A, Humphries JD, Humphries MJ (2013) Defining the extracellular matrix using proteomics. Int J Exp Pathol 94:75–92
D’Amelio M, Cavallucci V, Cecconi F (2010) Neuronal caspase-3 signaling: Not only cell death. Cell Death Differ 17:1104–1114. https://doi.org/10.1038/cdd.2009.180
Dauth S, Grevesse T, Pantazopoulos H et al (2016) Extracellular matrix protein expression is brain region dependent. J Comp Neurol 524:1309–1336. https://doi.org/10.1002/cne.23965
Dong L, Chen Y, Lewis M et al (2002) Neurologic defects and selective disruption of basement membranes in mice lacking entactin-1/nidogen-1. Lab Investig 82:1617–1630. https://doi.org/10.1097/01.LAB.0000042240.52093.0F
Engebretsen KVT, Lunde IG, Strand ME et al (2013) Lumican is increased in experimental and clinical heart failure, and its production by cardiac fibroblasts is induced by mechanical and proinflammatory stimuli. FEBS J 280:2382–2398. https://doi.org/10.1111/febs.12235
Fleischhacker WW, Arango C, Arteel P et al (2014) Schizophrenia-Time to commit to policy change. Schizophr Bull 40:S165–S194. https://doi.org/10.1093/schbul/sbu006
Gongidi V, Ring C, Moody M et al (2004) SPARC-like 1 Regulates the Terminal Phase of Radial Glia-Guided Migration in the Cerebral Cortex. Neuron 41:57–69
Gupta S, Kulhara P (2010) What is schizophrenia: A neurodevelopmental or neurodegenerative disorder or a combination of both? A critical analysis. Indian J Psychiatry 52:21–27. https://doi.org/10.4103/0019-5545.58891
Henrich-Noack P, Nikitovic D, Neagu M et al (2019) The blood–brain barrier and beyond: Nano-based neuropharmacology and the role of extracellular matrix. Nanomedicine Nanotechnology, Biol Med 17:359–379. https://doi.org/10.1016/j.nano.2019.01.016
Iozzo RV, Gubbiotti MA (2018) Extracellular matrix: The driving force of mammalian diseases. Matrix Biol 71–72:1–9. https://doi.org/10.1016/j.matbio.2018.03.023
Järveläinen H, Sainio A, Koulu M et al (2009) Extracellular matrix molecules: potential targets in pharmacotherapy. Pharmacol Rev 61:198–223. https://doi.org/10.1124/pr.109.001289.provided
Jones EV, Bernardinelli Y, Tse YC et al (2011) Astrocytes Control Glutamate Receptor Levels at Developing Synapses through SPARC- -Integrin Interactions. J Neurosci 31:4154–4165. https://doi.org/10.1523/JNEUROSCI.4757-10.2011
Kapitein LC, Hoogenraad CC (2015) Building the Neuronal Microtubule Cytoskeleton. Neuron 87:492–506
Kochunov P, Hong LE (2014) Neurodevelopmental and neurodegenerative models of schizophrenia: White matter at the center stage. Schizophr Bull 40:721–728. https://doi.org/10.1093/schbul/sbu070
Köhling R, Nischt R, Vasudevan A et al (2006) Nidogen and nidogen-associated basement membrane proteins and neuronal plasticity. Neurodegener Dis 3:56–61. https://doi.org/10.1159/000092094
Kruegel J, Miosge N (2010) Basement membrane components are key players in specialized extracellular matrices. Cell Mol Life Sci 67:2879–2895. https://doi.org/10.1007/s00018-010-0367-x
Kurian SM, Le-Niculescu H, Patel SD et al (2011) Identification of blood biomarkers for psychosis using convergent functional genomics. Mol Psychiatry 16:37–58. https://doi.org/10.1038/mp.2009.117
Lampi MC, Reinhart-King CA (2018) Targeting extracellular matrix stiffness to attenuate disease: From molecular mechanisms to clinical trials. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aao0475
Lau LW, Cua R, Keough MB et al (2013) Pathophysiology of the brain extracellular matrix: A new target for remyelination. Nat Rev Neurosci 14:722–729
Long KR, Huttner WB (2019) How the extracellular matrix shapes neural development. Open Biol 9:1–12. https://doi.org/10.1098/rsob.180216
Long KR, Newland B, Florio M et al (2018) Extracellular Matrix Components HAPLN1, Lumican, and Collagen I Cause Hyaluronic Acid-Dependent Folding of the Developing Human Neocortex. Neuron 99:702-719.e7. https://doi.org/10.1016/j.neuron.2018.07.013
López-Murcia FJ, Terni B, Llobet A (2015) SPARC triggers a cell-autonomous program of synapse elimination. Proc Natl Acad Sci 112:13366–13371. https://doi.org/10.1073/pnas.1512202112
Maeda N (2015) Proteoglycans and neuronal migration in the cerebral cortex during development and disease. Front Neurosci 9:1–15. https://doi.org/10.3389/fnins.2015.00098
Mauney SA, Athanas KM, Pantazopoulos H et al (2013) Developmental pattern of perineuronal nets in the human prefrontal cortex and their deficit in schizophrenia. Biol Psychiatry 74:427–435. https://doi.org/10.1016/j.biopsych.2013.05.007
Miyamae Y, Nakamura Y, Kashiwagi Y et al (1998) Altered adhesion efficiency and fibronectin content in fibroblasts from schizophrenic patients. Psychiatry Clin Neurosci 52:345–352. https://doi.org/10.1046/j.1440-1819.1998.00386.x
Mouw JK, Ou G, Weaver VM (2014) Extracellular matrix assembly: a multiscale deconstruction. Nat Rev Mol Cell Biol 15:771–785. https://doi.org/10.1038/nrm3902.Extracellular
Naba A, Clauser KR, Hoersch S et al (2011) The Matrisome. In Silico Definition and In Vivo Characterization by Proteomics of Normal and Tumor Extracellular Matrices. Mol Cell Proteomics 11:1–18. https://doi.org/10.1074/mcp.m111.014647
Nau R, Sörgel F, Eiffert H (2010) Penetration of drugs through the blood-cerebrospinal fluid/blood-brain barrier for treatment of central nervous system infections. Clin Microbiol Rev 23:858–883
Neame PJ, Kay CJ, McQuillan DJ et al (2000) Independent modulation of collagen fibrillogenesis by decorin and lumican. Cell Mol Life Sci 57:859–863. https://doi.org/10.1007/s000180050048
Novak U, Kaye AH (2000) Extracellular matrix and the brain: Components and function. J Clin Neurosci 7:280–290. https://doi.org/10.1054/jocn.1999.0212
Pantazopoulos H, Woo TUW, Lim MP et al (2010) Extracellular matrix-glial abnormalities in the amygdala and entorhinal cortex of subjects diagnosed with schizophrenia. Arch Gen Psychiatry 67:155–166. https://doi.org/10.1001/archgenpsychiatry.2009.196
Paylor JW, Wendlandt E, Freeman TS et al (2018) Impaired cognitive function after perineuronal net degradation in the medial prefrontal cortex. eNeuro 5:1–15. https://doi.org/10.1523/ENEURO.0253-18.2018
Rao J, Chiappelli J, Kochunov P et al (2015) Is Schizophrenia a Neurodegenerative Disease? Evidence from of Brain-Derived Neurotrophic Factor in the Brains of Schizophrenia Patients and Matched Non-Psychiatric Controls. Neurodegener Dis 15:38–44. https://doi.org/10.1159/000369214
Rodrigues-Amorim D, Rivera-Baltanas T, Del Carmen V-C et al (2019) Proteomics in Schizophrenia : A Gateway to Discover Potential Biomarkers of Psychoneuroimmune Pathways. Front Psychiatry 10:1–10. https://doi.org/10.3389/fpsyt.2019.00885
Rodrigues-Amorim D, Rivera-Baltanás T, del Carmen V-C et al (2020) Plasma β-III tubulin, neurofilament light chain and glial fibrillary acidic protein are associated with neurodegeneration and progression in schizophrenia. Sci Rep 10:1–10. https://doi.org/10.1038/s41598-020-71060-4
Rouslahti E (1996) Brain extracellular matrix. Glycobiology 6:489–492
Sethi M, Zaia J (2017) Extracellular Matrix Proteomics in Schizophrenia and Alzheimer’s Disease. Anal Chem 409:379–394. https://doi.org/10.1109/EMBC.2016.7590696.Upper
Shaw LM, Korecka M, Clark CM et al (2007) Biomarkers of neurodegeneration for diagnosis and monitoring therapeutics. Nat Rev Drug Discov 6:295–303
Tonge DA, De Burgh HT, Docherty R et al (2012) Fibronectin supports neurite outgrowth and axonal regeneration of adult brain neurons in vitro. Brain Res 1453:8–16. https://doi.org/10.1016/j.brainres.2012.03.024
Tyanova S, Cox J (2018) Perseus: A Bioinformatics Platform for Integrative Analysis of Proteomics Data in Cancer Research. In: Tyanova S, Cox J (eds) Cancer Systems Biology: Methods and Protocols, Methods in Molecular Biology. Springer, New York, New York, pp 133–148
Tyanova S, Temu T, Cox J (2016) The MaxQuant computational platform for mass spectrometry-based shotgun proteomics. Nat Protoc 11:2301–2319. https://doi.org/10.1038/nprot.2016.136
Wang J, Yin L, Chen Z (2011) New insights into the altered fibronectin matrix and extrasynaptic transmission in the aging brain. J Clin Gerontol Geriatr 2:35–41. https://doi.org/10.1016/j.jcgg.2010.12.002
Wang JT, Yin L, Chen Z (2013) Neuroprotective role of fibronectin in neuron-glial extrasynaptic transmission. Neural Regen Res 8:376–382. https://doi.org/10.3969/j.issn.1673-5374.2013.04.010
Willis CM, Crocker SJ (2016) The Mosaic of Extracellular Matrix in the Central Nervous System as a Determinant of Glial Heterogeneity. In: Willis CM, Crocker SJ (eds) Composition and Function of the Extracellular Matrix in the Human Body. BoD, Germany, pp 115–141
Yang Z, Wang KKW (2015) Glial fibrillary acidic protein: From intermediate filament assembly and gliosis to neurobiomarker. Trends Neurosci 38:364–374
Acknowledgement
The authors would like to thank the Galicia Sur Health Research Institute (Instituto de Investigación Sanitaria Galicia Sur) and the University Hospital Complex of Vigo (Complexo Hospitalario Universitario de Vigo) for their support. In addition, the authors are especially thankful for the aid provided by the Psychiatric Nursing and Psychiatry Services of the Álvaro Cunqueiro and Nicolás Peña Hospitals.
Funding
This research was financially backed by the Foundation for Science and Technology (FCT, Fundação para a Ciência e Tecnologia) within the framework of grant SFRH/BD/135623/2018 awarded to Daniela Rodrigues-Amorim. Our research was further supported by the Carlos III Health Institute (ISCIII, Instituto Carlos III) through grant P16/00405, and the Ministry of Health, Equality, and Social Policy (Ministerio de Sanidad, Servicios Sociales e Igualdad) – Government Delegation for the National Plan on Drugs (Delegación del Gobierno para el Plan Nacional sobre Drogas) through grant number 2017I054 awarded to José Manuel Olivares, the Health Knowledge Agency (ACIS, Axencia de Coñecementoen Saúde) grant number PRIS2-17, and the Galician Network for Dementia Research (GAIN, Red Gallega de Investigaciónen Demencias) through grant number IN607C-2017/02 awarded to Carlos Spuch, and Luis Freiría Martínez (GAIN, AxenciaGalega de Innovación) with grant number IN606A-2019/025. Finally, this work was also supported by the Consolidation and structure programme of competitive research units (Consolidación y estructuración de unidades de investigacióncompetitivas) through grant number IN607B 2018/17.
Author information
Authors and Affiliations
Contributions
DRA, TRB, and CS designed the study and acquired data to analysis. DRA, TRB, and CS wrote the article. PFP, MIMA, LFM, CRJ, MCVC, MAA, MLG, EH, AGC, and JMO made substantial contributions to conception and design of the study. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
Jose Manuel Olivares declared paid positions, honoraria and advisory boards by Angelini, AstraZeneca, Bristol-Myers, Casen Ricordati, GSK, Janssen, Lilly, Lundbeck, Novartis, Otsuka, Pfizer, Sanofi. The rest of the authors declare no conflict of interest.
Ethical Approval
Ethical approval for this study was obtained from Ethics Committee (Galician Network of Research Ethics Committees and Scientific and Ethics Committee of the IISGS Biobank).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rodrigues-Amorim, D., Rivera-Baltanás, T., Fernández-Palleiro, P. et al. Changes in the Brain Extracellular Matrix Composition in schizophrenia: A Pathophysiological Dysregulation and a Potential Therapeutic Target. Cell Mol Neurobiol 42, 1921–1932 (2022). https://doi.org/10.1007/s10571-021-01073-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-021-01073-8