Abstract
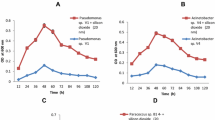
In this study the biodegradation characteristics of aromatic aliphatic copolyester Ecoflex was assayed with five different bacterial strains isolated from different kinds of environments and living at diverse optimum of temperature. The biodegradation properties of selected strains (Brevibacterium luteolum, Bacillus thuringiensis, Aeromonas media, Geobacillus kaustophilus and Serratia sp.) were assessed through several plate assays oriented to their proteolytic, lipolytic, esterolytic and cellulolytic abilities; catalase activities were measured too. In addition the bacterial hydrolytic abilities for polyhydroxybutyrate, polylactic acid and polybutyrate adipate terephthalate were obtained using specific culture media including also a specific Ecoflex agar. The biodegradation dynamics of selected strains were evaluated mainly by the incubation of Ecoflex foils with a specific bacterium during a period from 3 days until 16 weeks and the change of the degradability of the Ecoflex surface in nitrogen has been assessed by nonisothermal chemiluminescence measurements from 40 to 250 °C. Scanning electron microscope observation of inoculated foils was applied too. The comparison of the results showed the complexity of biodegradation phenomena. The concentration of oxygenated products measured by reached the maximum after 2 weeks of cultivation for strains G. kaustophilus and B. thurigiensis, after 4 weeks of cultivation for strain B. luteolum, and 8 weeks of cultivation for strains Serratia sp. and A. media. Each strain evidenced its own hydrolytic optimum at different periods, but almost all tested strains demonstrated a decline of hydrolysis up to 16 weeks of incubation. This study demonstrated that nonisothermal chemiluminescence examining momentaneous oxidation state of the polymer surface is a valuable technique to follow the biodegradation dynamics on polymeric film, while the Ecoflex agar is able to select useful polymer-degrading bacteria.










Similar content being viewed by others
References
Artham T, Sudhakar M, Venkatesan R et al (2009) Biofouling and stability of synthetic polymers in sea water. Int Biodeterior Biodegrad 63:884–890
Novotný Č, Erbanová P, Sezimová H et al (2015) Biodegradation of aromatic-aliphatic copolyesters and polyesteramides by esterase activity-producing microorganisms. Int Biodeterior Biodegrad 97:25–30
Witt U, Einig T, Yamamoto M et al (2001) Biodegradation of aliphatic-aromatic copolyesters: evaluation of the final biodegradability and ecotoxicological impact of degradation intermediates. Chemosphere 44:289–299
Chen Y, Tan L, Chen L et al (2008) Study on biodegradable aromatic/aliphatic copolyesters. Braz J Chem Eng 25:321–335
Müller RJ, Kleeberg I, Deckwer WD (2001) Biodegradation of polyesters containing aromatic constituents. J Biotechnol 86:87–95
Mostafa HM, Sourell H, Bockisch FJ (2010) The mechanical properties of some bioplastics under different soil types for use as a biodegradable drip tubes. Agric Eng Int 12:1–16
Shah AA, Hasan F, Hameed A, Ahmed S (2008) Biological degradation of plastics: a comprehensive review. Biotechnol Adv 26:246–265
Allen AD, Anderson WA, Ayorinde F, Eribo BE (2011) Isolation and characterization of an extracellular thermoalkanophilic P(3HB-co-3HV) depolymerase from Streptomyces sp. IN1. Int Biodeterior Biodegrad 65:777–785
Bornscheuer UT (2002) Microbial carboxyl esterases: classification, properties and application in biocatalysis. FEMS Microbiol Rev 26:73–81
Trinh TF, Cooper DG, Marić M, Nicell JA (2008) Biodegradation of a synthetic co-polyester by aerobic mesophilic microorganisms. Polym Degrad Stab 93:1479–1485
Hu X, Thumarat U, Zhang X, Tang M, Kawai F (2010) Diversity of polyester-degrading bacteria in compost and molecular analysis of a thermoactive esterase from Thermobifida alba AHK119. Appl Microbiol Biotechnol 87:771–779
Shah AA, Kato S, Shintani N et al (2014) Microbial degradation of aliphatic and aliphatic-aromatic co-polyesters. Appl Microbiol Biotechnol 98:3437–3447
Albertsson ACH, Karlsson S (1993) Aspects of biodeterioration of inert and degradable polymers. Int Biodeterior Biodegrad 31:161–170
Belal EB (2013) Biodegradation of aliphatic-aromatic coplyester under thermophilic conditions. Res J Environ Earth Sci 5:677–690
Lee SH, Kim IY, Song WS (2014) Biodegradation of polylactic acid (PLA) fibers using different enzymes. Macromol Res 22:657–663
Li R, Gao B, Sun J et al (2016) Synthesis, characterization of a novel lignin-based polymer and its behavior as a coagulant aid in coagulation/ultrafiltration hybrid process. Int Biodeterior Biodegrad 113:334–341.
Abrusci C, Marquina D, Santos A et al (2009) Biodeterioration of cinematographic cellulose triacetate by Sphingomonas paucimobilis using indirect impedance and chemiluminescence techniques. Int Biodeterior Biodegrad 63:759–764
Matisová-Rychlá L, Rychlý J (1996) Inherent relations of chemiluminescence and thermooxidation of polymers, Chapter 12. In: Clough RL, Billingham NC, Gillen KT (eds) Polymer durability. ACS advances in chemistry. Washington DC, p 175–193
Zlatkevich L (1989) Luminescence techniques in solid state polymer research, Marcel Dekker Inc., New York
Matisová-Rychlá L, Bukovský V, Rychlý J, Pleteníková M (2007) Chemiluminescence—a novel method in the research of degradation of paper. I: the effect of light on stacked sheets of paper. Macromol Symp 247:340–349
Rychlý J, Rychlá L, Stloukal P et al (2013) UV initiated oxidation and chemiluminescence from aromatic-aliphatic co-polyesters and polylactic acid. Polym Degrad Stab 98:2556–2563
Abrusci C, Marquina D, Santos A et al (2007) A chemiluminescence study on degradation of gelatine biodegradation by bacteria and fungi isolated from cinematographic films. J Photochem Photobiol A Chem 185:188–197
Abrusci C, Pablos JL, Corrales T et al (2011) Biodegradation of photo-degraded mulching fi lms based on polyethylenes and stearates of calcium and iron as pro-oxidant additives. Int Biodeterior Biodegrad 65:451–459
Abrusci C, Pablos JL, Marin I et al (2012) Photodegradation and biodegradation by bacteria of mulching films based on ethylene-vinyl acetate copolymer: effect of pro-oxidant additives. J Appl Polym Sci 126:664–1675
Pangallo D, Kraková L, Chovanová K et al (2013) Disclosing a crypt: microbial diversity and degradation activity of the microflora isolated from funeral clothes of Cardinal Peter Pázmány. Microbiol Res 168:289–299
Pangallo D, Bučková M, Kraková L et al (2015) Biodeterioration of epoxy resin: a microbial survey through culture-independent and culture-dependent approaches. Environ Microbiol 17:462–479
Grivalský T, Bučková M, Puškárová A et al (2016) Water-related environments: a multistep procedure to assess the diversity and enzymatic properties of cultivable bacteria. World J Microbiol 32:42
Roggenkamp R, Sahm H, Wagner F (1974) Microbial assimilation of methanol induction and function of catalase in Candida boidinii. FEBS Lett 41:283–286
Ishigaki T, Sugano W, Nakanishi A et al (2004) The degradability of biodegradable plastics in aerobic and anaerobic waste landfill model reactors. Chemosphere 54:225–233
Díaz A, Katsarava R, Puiggalí J (2014) Synthesis, properties and applications of biodegradable polymers derived from diols and dicarboxylic acids: from polyesters to poly(ester amide)s. Int J Mol Sci 15:7064–7123
Amjadi M, Yoon YJ, Park I (2015) Ultra-stretchable and skin-mountable strain sensors using carbon nanotubes–Ecoflex nanocomposites. Nanotechnology 26:375501
Tachibana Y, Maeda T, Ito O et al (2009) Utilization of a biodegradable mulch sheet produced from poly(lactic acid)/Ecoflex®/modified starch in Mandarin orange groves. Int J Mol Sci 10:3599–3615
Acknowledgements
The acknowledgements are due to Grant Agency VEGA 2/0122/15, 2/0161/14 and 2/0167/14, and also to Slovak Research and Development Agency for the project APVV-15-0528.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grivalský, T., Rychlý, J., Rychlá, L. et al. Aerobic Biodegradation of Aromatic Aliphatic Copolyester Induced by Bacteria Obtained from Different Environments. J Polym Environ 26, 680–690 (2018). https://doi.org/10.1007/s10924-017-0980-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-017-0980-y