Abstract
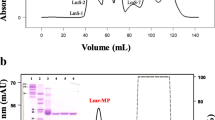
A serine protease from Bothrops atrox (Peruvian specimen’s venom) was isolated in two chromatographic steps in LC molecular exclusion and reverse phase-HPLC. This protein was denominated Ba III-4 (33,080.265 Da determinated by MALDI-TOF mass spectrometry) and showed pI of 5.06, Km 0.2 × 10−1 M and the V máx 4.1 × 10−1 nmoles p-NA/lt/min on the synthetic substrate BapNA. Ba III-4 also showed ability to coagulate bovine fibrinogen. The serine protease was inhibited by soyben trypsin inhibitor and DA2II, which is an anti-hemorrhagic factor isolated from the opossum specie Didelphis albiventris. The primary structure of Ba III-4 showed the presence of His(44), Asp(94) and Ser(193) residues in the corresponding positions to the catalytic triad established in the serine proteases and Ser(193) are inhibited by phenylmethylsulfonylfluoride (PMSF). Amino acid analysis showed a high content of Asp, Glu, Gly, Ser, Ala and Pro, as well as 12 half-cysteine residues. Ba III-4 contained 293 amino acid residues and the primary structure of VIGGDECDIN EHPFLAFMYY SPRYFCGMTL INQEWVLTAA HCRYFCGMTL IHLGVHRESE KANYDEVRRF PKEKYFIFCD NNFTDDEVDK DIMLIRLDKP VSNSEHIAPL SLPSNPPSVG SVCRIMGWGQ TTTSPIDVLS PDEPHCANIN LFDNTVCHTA HPQVANTRTS TDTLCAGDLQ GGRDTCNGDS GGPLICNEQL HGILSWGGDP CAQPNKPAFY TKVYYFDHPW IKSIIAGNKK TVNFTCPPLR SDAKDDSTTY INQEWDWVLT AEHCDRTHMR NSFYDYSSIN SDS. Titration experiments did not show the presence of free sulfhydryl groups after 4 h incubation, nor were differences found in relation to titration kinetics in the presence of nondenaturating buffer. The isolation of this protein, Ba III-4, is of potential interest for the understanding of the pathomechanism of the snake venom action and for the identification of new blood coagulation enzymes of natural sources.






Similar content being viewed by others
Abbreviations
- RP-HPLC:
-
reverse phase high performance liquid chromatography
- DTT:
-
dithiothreitol
- BaIII-4:
-
serine protease from Bothrops atrox
- MALDI-TOF:
-
matrix assisted laser desorption ionization time-of-flight
- K m :
-
Michaelis constant
- V máx :
-
maximum velocity
- DA2II:
-
anti-hemorrhagic factor Didelphis albiventris
- PMSF:
-
phenylmethylsulfonyl fluoride
- BapNA:
-
N-benzoyl-l-arginine-p-nitroanilide
- SBTI:
-
soybean trypsin
- SDS-PAGE:
-
sodium dodecyl sulpate-polyacrilamide gel electrophoresis
- DTNB:
-
dithiobis-(2-nitrobenzoate)
- EDTA:
-
ethylenedinitro tetraacetic acid
- RC-BaIII-4:
-
reduced carboxymethylated-serine protease from B. atrox
- SV8:
-
staphylococcus aureus protease
References
Au L. C., Lin S. B., Chou J. S., Teh G. W., Chang K. J., Shih C. M. (1993). J. Biochem. 294: 387–390
Burkhart W., Smith G. F., Su J. L., Parikh I., LeVine H. (1992). FEBS Lett. 297(3): 297–301
Campbell, J. A., and Lamar, W. (1989). The Venomous Reptiles of Latin America, Comstock Publ. Assoc., Cornell University Press, Ithaca, New York, 425 pp
Castro H. C., Silva D. M., Craik C., Zingali R. B. (2001). Biochem. Biophys. Acta 1547: 183–195
Cavinato R. A., Remold H., Kipnis T. L. (1998). Toxicon 36: 257–267
Deshimaru M., Ogawa T., Nakasjima K., Nobuhisa I., Chijiwa T., Shimohigashi Y., Fukumaki Y., Niwa M., Yamashina I., Hattori S., Ohno M. (1996). FEBS Lett. 397: 83–88
Ellman G. L. (1959). Arch. Biochem. Biophys. 82: 70–77
Erlanger B. F., Kokowsky N., Cohen W. (1961). Biochim. Biophys. Acta 95: 271–278
Fan H. W., Cardoso J. L. (1995). In: Méier J., White J. (Eds) Handbook of Clinical Toxicology of Animal Venoms and Poisons. CRC Press, Boca Raton, pp. 667–688
Francis S. M. (1998). Toxicon 36: 1749–1800
Goñi M., Vaisberg A., Zavaleta A. (1992). Rev. Biol. Trop. 40(1):143–145
Hahn B. S., Yang K. Y., Park E. M., Chang I. M., Kim Y. S. (1996). J. Biochem. (Tokyo) 119: 835–843
Heinrikson R. L., Meridith S. C. (1984). Anal. Biochem. 13: 65–72
Incio-Ruiz R., Incio-Ruiz L., Zavaleta Martinez-Vargas A., Salas-Arruz M., Gutierrez J. M., (1993). Rev. Biol. Trop. 41: 351–357
Itoh N., Tanaka N., Mihashi S., Yamashina I. (1987). J. Biol. Chem. 262: 3132–3135
Kawasaki T., Fujimura Y., Usami Y., Suzuki M., Miura S., Sakuri Y., Makita K., Taniuchi Y., Hirano K., Titani K. (1996). J. Biol. Chem. 271: 10635–10639
Kini R. M. (2005). Pathophysiol Haemost. Thromb. 34(4–5): 200–204
Kumasaka T., Yamamoto M., Moriyama H., Tanaka N., Sato M., Katsube Y., Yamakawa Y Omori-Satoh T., Iwanaga S., Ueki T. (1996). J. Biochem. 119: 49–57
Nikai T., Ohara A., Komori Y., Fox J. W., Sugihara H. (1995). Arch. Biochem. Biophys. 318: 89–96
Petreski J. H., Kanashiro M., Silva C. P., Alves E. W., Kinipis T. L. (2000). Braz. J. Med. Biol. Res. 33: 1293–3000
Pirkle H., Stocker K. (1991). Thromb. Haemost. 65: 444–450
Rojas E., Quesada L., Arce V., Lomonte B., Rojas G., Gutierrez J. M. (2005). Acta Trop. 93(1):85–95
Schagger H. A., Von Jagow G. (1987). Anal. Biochem. 166: 368–379
Smolka M. B., Marangoni S., Oliveira B., Novello J. C. (1998). Toxicon 36:1059–1063
Toyama M. H., Mancuso L. C., Giglio J. R., Novello J. C., Oliveira B., Marangoni S. (1995). Biochem. Mol. Biol. Int. 37: 1047–1055
Toyama M. H., Soares A. M., Wen-Hwa L., Polikarpov I., Giglio J. R., Marangoni S. (2000). Biochemie 82: 189–264
Waheed A., Van Etten R. L. (1985). Int. J. Pept. Protein Res. 26: 362–372
Walter, K., Shütt, C. (1974). In: Bergmeyer, H. U. (ed.), Methods of Enzymatic Analysis, Verlang Chemic, Weinheim, pp. 856–861
Zavaleta A., Salas M. (1996). Emergencias en Medicina Interna. Lima, Peru, pp. 241–260
Acknowledgements
The authors thank Marino Forner and Paulo A. Baldasso for technical assistance. This work was supported by the Fundação de Amparo a Pesquisa, São Paulo (FAPESP).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ponce-Soto, L.A., Bonfim, V.L., Novello, J.C. et al. Isolation and Characterization of a Serine Protease, Ba III-4, from Peruvian Bothrops atrox venom. Protein J 26, 387–394 (2007). https://doi.org/10.1007/s10930-007-9078-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-007-9078-z