Abstract
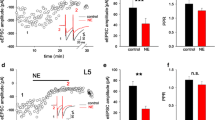
Although accumulative evidence indicates that the thalamocortical system is an important target for general anesthetics, the underlying mechanisms of anesthetic action on thalamocortical neurotransmission are not fully understood. The aim of the study is to explore the action of etomidate on glutamatergic and GABAergic transmission in rat thalamocortical slices by using whole cell patch-clamp recording. We found that etomidate mainly prolonged the decay time of spontaneous GABAergic inhibitory postsynaptic currents (sIPSCs), without changing the frequency. Furthermore, etomidate not only prolonged the decay time of miniature inhibitory postsynaptic currents (mIPSCs) but also increased the amplitude. On the other hand, etomidate significantly decreased the frequency of spontaneous glutamatergic excitatory postsynaptic currents (sEPSCs), without altering the amplitude or decay time in the absence of bicuculline. When GABAA receptors were blocked using bicuculline, the effects of etomidate on sEPSCs were mostly eliminated. These results suggest that etomidate enhances GABAergic transmission mainly through postsynaptic mechanism in thalamocortical neuronal network. Etomidate attenuates glutamatergic transmission predominantly through presynaptic action and requires presynaptic GABAA receptors involvement.






Similar content being viewed by others
Abbreviations
- GABA:
-
γ-Aminobutyric acid
- ACSF:
-
Artificial cerebrospinal fluid
- S1BF:
-
The primary somatosensory barrel cortex
- VPM:
-
The ventral posteromedial nucleus
- sEPSCs:
-
Spontaneous excitatory postsynaptic currents
- sIPSCs:
-
Spontaneous inhibitory postsynaptic currents
- mEPSCs:
-
Miniature excitatory postsynaptic currents
- mIPSCs:
-
Miniature inhibitory postsynaptic currents
- VB:
-
The ventrobasal complex
- RTN:
-
The reticular nucleus
References
Rudolph U, Antkowiak B (2004) Molecular and neuronal substrates for general anaesthetics. Nat Rev Neurosci 5:709–720
Franks NP (2008) General anaesthesia: from molecular targets to neuronal pathways of sleep and arousal. Nat Rev Neurosci 9:370–386
Alkire MT, Hudetz AG, Tononi G (2008) Consciousness and anesthesia. Science 322:876–880
Rath M, Fohr KJ, Weigt HU, Gauss A, Engele J, Georgieff M, Koster S, Adolph O (2008) Etomidate reduces glutamate uptake in rat cultured glial cells: involvement of PKA. Br J Pharmacol 155:925–933
Li Z, Luo C, Sun YY, Chen J (2004) Effects of etomidate on local synaptic transmission in substantia gelatinosa neurons of the adult rat spinal cord. Sheng Li Xue Bao 56:413–418
Edelman GM (2003) Naturalizing consciousness: a theoretical framework. Proc Natl Acad Sci USA 100:5520–5524
Tononi G, Koch C (2008) The neural correlates of consciousness: an update. Ann N Y Acad Sci 1124:239–261
Zhang Y, Li Z, Dong H, Yu T (2014) Effects of general anesthesia with propofol on thalamocortical sensory processing in rats. J Pharmacol Sci 126:370–381
Zhang Y, Wang C, Zhang L, Yu T (2013) GABAA receptor in the thalamic specific relay system contributes to the propofol-induced somatosensory cortical suppression in rat. PLoS One 8:e82377
Li Z, Liu X, Zhang Y, Shi J, Xie P, Yu T (2014) Connection changes in somatosensory cortex induced by different doses of propofol. PLoS One 9:e87829
Zhang Y, He JC, Liu XK, Wang Y, Yu T (2014) Assessment of the effect of etomidate on voltage-gated sodium channels and action potentials in rat primary sensory cortex pyramidal neurons. Eur J Pharmacol 736:55–62
Tu Y, Yu T, Fu XY, Xie P, Lu S, Huang XQ, Gong QY (2011) Altered thalamocortical functional connectivity by propofol anesthesia in rats. Pharmacology 88:322–326
Ying SW, Goldstein PA (2005) Propofol suppresses synaptic responsiveness of somatosensory relay neurons to excitatory input by potentiating GABA(A) receptor chloride channels. Mol Pain 1:2
Yang H, Wang Y, Zhang Y, Xu MS, Yuan J, Yu T (2016) Astrocytes contribute to the effects of etomidate on synaptic transmission in rat primary somatosensory cortex. Brain Res. doi:10.1016/j.brainres.2016.03.052
Kotani N, Wakita M, Shin MC, Ogawa S, Nonaka K, Akaike N (2012) Effects of halothane on GABAergic and glutamatergic transmission in isolated hippocampal nerve-synapse preparations. Brain Res 1473:9–18
Heinisch S, Kirby LG (2010) SDF-1alpha/CXCL12 enhances GABA and glutamate synaptic activity at serotonin neurons in the rat dorsal raphe nucleus. Neuropharmacology 58:501–514
Jang IS, Jeong HJ, Katsurabayashi S, Akaike N (2002) Functional roles of presynaptic GABA(A) receptors on glycinergic nerve terminals in the rat spinal cord. J Physiol 541:423–434
Akeju O, Loggia ML, Catana C, Pavone KJ, Vazquez R, Rhee J, Contreras Ramirez V, Chonde DB, Izquierdo-Garcia D, Arabasz G, Hsu S, Habeeb K, Hooker JM, Napadow V, Brown EN, Purdon PL (2014) Disruption of thalamic functional connectivity is a neural correlate of dexmedetomidine-induced unconsciousness. Elife 3:e04499
Liu X, Lauer KK, Ward BD, Li SJ, Hudetz AG (2013) Differential effects of deep sedation with propofol on the specific and nonspecific thalamocortical systems: a functional magnetic resonance imaging study. Anesthesiology 118:59–69
Alkire MT, Haier RJ, Fallon JH (2000) Toward a unified theory of narcosis: brain imaging evidence for a thalamocortical switch as the neurophysiologic basis of anesthetic-induced unconsciousness. Conscious Cogn 9:370–386
Boveroux P, Vanhaudenhuyse A, Bruno MA, Noirhomme Q, Lauwick S, Luxen A, Degueldre C, Plenevaux A, Schnakers C, Phillips C, Brichant JF, Bonhomme V, Maquet P, Greicius MD, Laureys S, Boly M (2010) Breakdown of within- and between-network resting state functional magnetic resonance imaging connectivity during propofol-induced loss of consciousness. Anesthesiology 113:1038–1053
White NS, Alkire MT (2003) Impaired thalamocortical connectivity in humans during general-anesthetic-induced unconsciousness. Neuroimage 19:402–411
Verdonck O, Reed SJ, Hall J, Gotman J, Plourde G (2014) The sensory thalamus and cerebral motor cortex are affected concurrently during induction of anesthesia with propofol: a case series with intracranial electroencephalogram recordings. Can J Anaesth 61:254–262
Dai S, Perouansky M, Pearce RA (2009) Amnestic concentrations of etomidate modulate GABAA, slow synaptic inhibition in hippocampus. Anesthesiology 111:766–773
Grasshoff C, Jurd R, Rudolph U, Antkowiak B (2007) Modulation of presynaptic beta3-containing GABAA receptors limits the immobilizing actions of GABAergic anesthetics. Mol Pharmacol 72:780–787
Banks MI, Pearce RA (1999) Dual actions of volatile anesthetics on GABA(A) IPSCs: dissociation of blocking and prolonging effects. Anesthesiology 90:120–134
Wakita M, Kotani N, Nonaka K, Shin MC, Akaike N (2013) Effects of propofol on GABAergic and glutamatergic transmission in isolated hippocampal single nerve-synapse preparations. Eur J Pharmacol 718:63–73
Jin YH, Zhang Z, Mendelowitz D, Andresen MC (2009) Presynaptic actions of propofol enhance inhibitory synaptic transmission in isolated solitary tract nucleus neurons. Brain Res 1286:75–83
Maldifassi MC, Baur R, Sigel E (2016) Functional sites involved in modulation of the GABA receptor channel by the intravenous anesthetics propofol, etomidate and pentobarbital. Neuropharmacology 105:207–214
Labrakakis C, Rudolph U, De Koninck Y (2014) The heterogeneity in GABAA receptor-mediated IPSC kinetics reflects heterogeneity of subunit composition among inhibitory and excitatory interneurons in spinal lamina II. Front Cell Neurosci 8:424
Mody I, Pearce RA (2004) Diversity of inhibitory neurotransmission through GABA(A) receptors. Trends Neurosci 27:569–575
Sassoe-Pognetto M, Panzanelli P, Sieghart W, Fritschy JM (2000) Colocalization of multiple GABA(A) receptor subtypes with gephyrin at postsynaptic sites. J Comp Neurol 420:481–498
Browne SH, Kang J, Akk G, Chiang LW, Schulman H, Huguenard JR, Prince DA (2001) Kinetic and pharmacological properties of GABA(A) receptors in single thalamic neurons and GABA(A) subunit expression. J Neurophysiol 86:2312–2322
Okada M, Onodera K, Van Renterghem C, Sieghart W, Takahashi T (2000) Functional correlation of GABA(A) receptor alpha subunits expression with the properties of IPSCs in the developing thalamus. J Neurosci 20:2202–2208
Pirker S, Schwarzer C, Wieselthaler A, Sieghart W, Sperk G (2000) GABA(A) receptors: immunocytochemical distribution of 13 subunits in the adult rat brain. Neuroscience 101:815–850
Kitamura A, Marszalec W, Yeh JZ, Narahashi T (2003) Effects of halothane and propofol on excitatory and inhibitory synaptic transmission in rat cortical neurons. J Pharmacol Exp Ther 304:162–171
Patel PM, Goskowicz RL, Drummond JC, Cole DJ (1995) Etomidate reduces ischemia-induced glutamate release in the hippocampus in rats subjected to incomplete forebrain ischemia. Anesth Analg 80:933–939
Xie F, Li X, Bao M, Shi R, Yue Y, Guan Y, Wang Y (2015) Anesthetic propofol normalized the increased release of glutamate and gamma-amino butyric acid in hippocampus after paradoxical sleep deprivation in rats. Neurol Res 37:1102–1107
McDougall SJ, Bailey TW, Mendelowitz D, Andresen MC (2008) Propofol enhances both tonic and phasic inhibitory currents in second-order neurons of the solitary tract nucleus (NTS). Neuropharmacology 54:552–563
Yamamoto S, Yoshimura M, Shin MC, Wakita M, Nonaka K, Akaike N (2011) GABA(A) receptor-mediated presynaptic inhibition on glutamatergic transmission. Brain Res Bull 84:22–30
Wakita M, Kotani N, Kogure K, Akaike N (2014) Inhibition of excitatory synaptic transmission in hippocampal neurons by levetiracetam involves Zn(2)(+)-dependent GABA type A receptor-mediated presynaptic modulation. J Pharmacol Exp Ther 348:246–259
Cheng VY, Martin LJ, Elliott EM, Kim JH, Mount HT, Taverna FA, Roder JC, Macdonald JF, Bhambri A, Collinson N, Wafford KA, Orser BA (2006) Alpha5GABAA receptors mediate the amnestic but not sedative-hypnotic effects of the general anesthetic etomidate. J Neurosci 26:3713–3720
Belelli D, Callachan H, Hill-Venning C, Peters JA, Lambert JJ (1996) Interaction of positive allosteric modulators with human and Drosophila recombinant GABA receptors expressed in Xenopus laevis oocytes. Br J Pharmacol 118:563–576
Zurek AA, Yu J, Wang DS, Haffey SC, Bridgwater EM, Penna A, Lecker I, Lei G, Chang T, Salter EW, Orser BA (2014) Sustained increase in alpha5GABAA receptor function impairs memory after anesthesia. J Clin Invest 124:5437–5441
Mantz J, Lecharny JB, Laudenbach V, Henzel D, Peytavin G, Desmonts JM (1995) Anesthetics affect the uptake but not the depolarization-evoked release of GABA in rat striatal synaptosomes. Anesthesiology 82:502–511
Hill-Venning C, Belelli D, Peters JA, Lambert JJ (1997) Subunit-dependent interaction of the general anaesthetic etomidate with the gamma-aminobutyric acid type A receptor. Br J Pharmacol 120:749–756
Antkowiak B, Rudolph U (2016) New insights in the systemic and molecular underpinnings of general anesthetic actions mediated by gamma-aminobutyric acid A receptors. Curr Opin Anaesthesiol 29:447–453
Amlong CA, Perkins MG, Houle TT, Miller KW, Pearce RA (2016) Contrasting effects of the gamma-aminobutyric acid type A receptor beta3 subunit N265M mutation on loss of righting reflexes induced by etomidate and the novel anesthetic barbiturate R-mTFD-MPAB. Anesth Analg. doi:10.1213/ANE.0000000000001358
Maldifassi MC, Baur R, Sigel E (2016) Functional sites involved in modulation of the GABAA receptor channel by the intravenous anesthetics propofol, etomidate and pentobarbital. Neuropharmacology 105:207–214
Acknowledgments
Supported by grants from the National Natural Science Foundation of China (NSFC, Grant No. 81060266 and 81571026). Thanks for Junwei Zeng (Zunyi Medical College, Zunyi, China) for carefully reading an early version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Additional information
Bao Fu and Yuan Wang have contributed to the work equally and should be regarded as co-first authors.
Rights and permissions
About this article
Cite this article
Fu, B., Wang, Y., Yang, H. et al. Effects of Etomidate on GABAergic and Glutamatergic Transmission in Rat Thalamocortical Slices. Neurochem Res 41, 3181–3191 (2016). https://doi.org/10.1007/s11064-016-2042-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-2042-6