Abstract
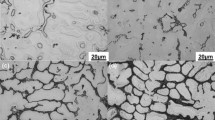
The influence of Nd and Gd additions on the evolution of microstructure, surface hardness and oxidation behavior of the extruded AZ91D magnesium alloy exposed to air was investigated in the temperature range from 290 to 410 °C. The kinetics of the oxidation process was obtained from gravimetric measurements performed during 28 days. Nd and Gd formed Al-rich intermetallic compounds (Al2Gd, Al2Nd, Al–Mn–Gd and Al–Mn–Nd) and reduced the volume fraction of β-Mg17Al12 phase. Surface degradation of the AZ91D alloy, with and without Gd or Nd, was negligible for temperatures below 370 °C. At 410 °C, the grain size and oxidation rate significantly increased, although lanthanide-containing alloys revealed lower degradation compared with the unmodified AZ91D alloy.














Similar content being viewed by others
References
J. Medved, P. Mrvar, and M. Voncina, Oxidation of Metals 71, 257 (2009).
F. Czerwinski, Acta Materialia 50, 2639 (2002).
F. Czerwinski, JOM Journal of the Minerals, Metals and Materials Society 56, 29 (2004).
D. Eliezer and H. Alves, in Handbook of Materials Selection, ed. by M. Kutz (John Wiley & Sons, New York, 2007).
M. D. López, C. J. Múnez, M. Carboneras, P. Rodrigo, M. D. Escalera, and E. Otero, Journal of Alloys and Compounds 491, 131 (2010).
C. Sanchez, H. Octor, G. Nussbaum, and P. Azavant, Materials Science and Engineering A 221, 48 (1996).
V. Neubert, I. Stulíková, B. Smola, B. L. Mordike, M. Vlach, A. Bakkar, and J. Pelcová, Materials Science and Engineering A 462, 329 (2007).
F. E. Horst and B. L. Mordike, Magnesium Technology—Metallurgy Applications, Design Data, (Springer, Berlin-Heidelberg, 2006).
M. O. Pekguleryuz and A. A. Kaya, Advanced Engineering Materials 5, 866 (2003).
S. Cheng, G. Yang, J. Fan, Y. Li, and Y. Zhou, Transactions of Nonferrous Metals Society of China 19, 299 (2009).
X. Zeng, Q. Wang, Y. Lu, Y. Zhu, W. Ding, and Y. Zhao, Journal of Materials Processing Technology 112, 17 (2001).
X. Zeng, Q. Wang, Y. Lu, W. Ding, C. Lu, Y. Zhu, C. Zhai, and X. Xu, Scripta Materialia 43, 403 (2000).
W. Li, H. Zhou, W. Zhou, W. P. Li, and M. X. Wang, Materials Letters 61, 2772 (2007).
P. Lin, H. Zhou, W.-P. Li, W. Li, M. Wang, Q. Guo, and H. Tang, Corrosion Science 51, 301 (2009).
H. Zhou, M. Wang, W. Li, L. Wang, and Y. Zhao, Materials Letters 60, 3238 (2006).
P. Lin, H. Zhou, W.-P. Li, M. Wang, Q. Guo, H. Tang, and W. Li, Journal of Alloys and Compounds 481, 373 (2009).
T. B. Massalski and H. Okamoto, in Binary Alloys Phase Diagrams, 2nd edn, ed. by P. R. Subramanian and L. Kacprzak (ASM, Metal Park, OH, 1990).
G. Zhang, H. Zhang, M. Gao, and D. Wei, Journal of Rare Earths 25, 348 (2007).
J. Zhang, S. Wang, J. Zhang, J. Zhang, and B. Xu, Rare Metal Materials and Engineering 38, 1141 (2009).
E. A. Gulbransen, Transactions of the Electrochemical Society 87, 589 (1945).
F. Czerwinski, Corrosion Science 46, 377 (2004).
W. P. Gilbreath, NASA Technical Note D-2723, 1965.
J. Medved, P. Mrvar, and M. Voncina, in Magnesium Alloys-corrosion, Surface Treatments Chapter 2, ed. by F. Czerwinski (Intechopen, India, 2011).
F. Czerwinski, Electrochimica Acta 44, 667 (1998).
T. D. Nguyen and D. B. Lee, Oxidation of Metals 73, 183 (2010).
Acknowledgments
The authors are grateful to the MCYT (Spain, Project MAT 2009-09845-C02-01), the Community of Madrid (Spain, S2009MAT-1585) and the MANOEQ of Departamento de Metalurgia Física of CENIM (CSIC) for supply of the test materials. R. Arrabal and E. Matykina are grateful to the MICINN (Spain) for financial support via the Ramon y Cajal Programme (RYC-2008-02038, RYC-2010-06749). K. Paucar is grateful to the Fundación Carolina for funding a grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arrabal, R., Pardo, A., Merino, M.C. et al. Oxidation Behavior of AZ91D Magnesium Alloy Containing Nd or Gd. Oxid Met 76, 433–450 (2011). https://doi.org/10.1007/s11085-011-9265-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11085-011-9265-3