Abstract
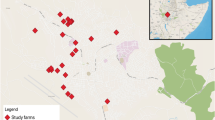
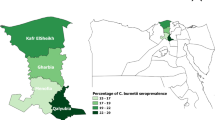
Coxiella burnetii, an obligate intracellular bacterium, is the causative agent of important zoonotic Q fever. It is the etiological agent of coxiellosis or Q fever in animals and human. This seroepidemiological survey was conducted to determine the seroprevalence of coxiellosis in increasingly camel raised population in vast area of Khorasan (North, South, and Razavi) provinces, northeast Iran. Using cluster random sampling strategy, 167 camels in 11 counties were selected as serum samples. Sera were assayed for antibody against C. burnetii using a Q fever ELISA kit. Logistic regression model was used to insight the contributing risk factor(s) of Q fever in the study area. C. burnetii was widely distributed throughout the study area. Seroprevalence of C. burnetii at animal level was 28.7 % [(95 % confidence interval (CI): 21.83, 35.56)] for camel populations. The proportion of seropositivity for camels in the studied counties ranged from 0 to 63.6 %. Logistic regression model showed that age correlated with seroprevalence of coxiellosis at the individual level in camels (P < 0.05). This study showed that a relatively high proportion of camels are seropositive to C. burnetii. Considering the economic, zoonotic, and public health importance of Q fever, percussion measures are to be implemented to prevent spreading of C. burnetii and zeroing the risk of Q fever in farm animals and human in this agro-ecologically and geopolitically important region.

Similar content being viewed by others
References
Angelakis, E., Raoult, D., 2010. Q fever. Veterinary Microbiology, 140, 297-309.
Aronson, N.E., 2008. Infections associated with war: the American forces experience in Iraq and Afghanistan. Clinical Microbiology Newsletter, 30, 135-40.
Arricau-Bouvery, N., Rodolakis, A., 2005. Is Q fever an emerging or reemerging zoonosis? Veterinary Research, 36,327-349.
Bailey, M.S., Trinick, T.R., Dunbar, J.A., Hatch, R., Osborne, J.C., Brooks, T.J., and Green, A.D., 2011. Undifferentiated febrile illnesses amongst British troops in Helmand, Afghanistan. Journal of the Royal Army Medical Corps, 157, 150-155.
Bielawska-Drózd, A., Cieślik, P., Mirski, T., Bartoszcze, M., Knap, J.P., Gaweł, J., and Żakowska, D., 2013. Q fever--selected issues. Annals of Agricultural and Environmental Medicine, 20, 222-32.
Doosti, A., Arshi, A., Sadeghi, M., 2014. Investigation on Coxiella burnetii Iranian camels. Comparative Clinical Pathology, 23, 43-46.
Ferrante, M.A., Dolan, M.J., 1993. Q fever meningoencephalitis in a soldier returning from the Persian Gulf War. Clinical Infectious Diseases, 16, 489-496.
Hartzell, J.D., Peng, S.W., Wood-Morris, R.N., Sarmiento, D.M., Collen, .F. Robben, P.M., Moran, K.A., 2007. Atypical Q fever in US Soldiers. Emerging Infectious Diseases, 13, 1247-1249.
Keyvani Rad, N., Azizzadeh, M., Taghavi Razavizadeh, A. R., Mehrzad, J., Rashtibaf, M., 2014. Seroepidemiology of coxiellosis (Q fever) in sheep and goat populations in the northeast of Iran. Iranian Journal of Veterinary Research, 15, 1- 6.
Khalili, M., Shahabi-Nejad, N., Golchin, M., 2010. Q fever serology in febrile patients in southeast Iran. Transactions of the Royal Society of Tropical Medicine and Hygiene, 104, 623- 624.
Luoto, L., 1960. Report on the nationwide occurrence of Q fever infections in cattle. Public health reports, 75, 135-140.
Lyytikäinen, O., Ziese, T., Schwartländer, B., Matzdorff, P., Kuhnhen, C., Jäger, C., Petersen, L., 1998. An outbreak of sheep-associated Q fever in a rural community in Germany. European Journal of Epidemiology, 14, 193-199.
Marrie, T.J., Stein, A., Janigan, D., Raoult, D., 1996. Route of infection determines the clinical manifestations of acute Q fever. The Journal of Infectious Diseases, 173, 484-487.
Mathur, K.N., Bhargava, S.C., 1979. Seroprevalence of Q fever and brucellosis in camels of Jorbeer and Bikaner, Rajasthan State. Indian Journal of Medical Research, 70, 391-393.
Maurin, M., Raoult, D., 1999. Q fever. Clinical Microbiology Reviews, 12, 518-53.
McCaughey, C., Murray, L.J., McKenna, J.P., Menzies, F.D., McCullough, S.J., O’Neill, H.J., Wyatt, D.E., Cardwell, C.R., Coyle, P.V., 2010. Coxiella burnetii (Q fever) Seroprevalence in cattle. Epidemiology and Infection, 138, 21-27.
McQuiston, J.H., Childs, J.E., 2002. Q fever in humans and animals in the United States. Vector Borne and Zoonotic Diseases, 2, 179-191.
Moffatt, J.H., Newton, P., and Newton, H.J. 2015. Coxiella burnetii: turning hostility into a home. Cell Microbiology, 17, 621-631.
Mohammed O.B., Jarelnabi A.A., Aljumaah R.S., Alshaikh M.A., Bakhiet A.O., Omer S.O., Alagaili A.N., Hussein M.F., 2014. Coxiella burnetii, the causative agent of Q fever in Saudi Arabia: molecular detection from camel and other domestic livestock. Asian Pacific Journal of Tropical Medicine, 715-719.
Norlander, L., 2000. Q fever epidemiology and pathogenesis. Microbes and Infection, 2, 417-424.
Nusinovici, S., Frössling, J., Widgren, S., Beaudeau, F., and Lindberg, A. 2015. Q fever infection in dairy cattle herds: increased risk with high wind speed and low precipitation. Epidemiology and Infection, 18, 1-11
O’Connor, B.A., Tribe, I.G., Givney, R., 2015. A windy day in a sheep sale yard: an outbreak of Q fever in rural South Australia. Epidemiology and Infection, 143, 391-398.
Ormsbee, R.A., Peacock, M.G., Gerloff, R., Tallent, G. and Wike, D., 1978. Limits of rickettsial infectivity. Infection and Immunity, 19, 239-245.
Pimenta, L., Alegria, N., Anastácio, S., Sidi-Boumedine, K., da Silva, G., Rabiço, Â. and Simões, J., 2015. Prevalence of Coxiella burnetii antibodies in Portuguese dairy cattle herds. Tropical Animal Health and Productio, 47, 227-230.
Ruiz-Fons, F., Astobiza, I., Barandika, J.F., Hurtado, A., Atxaerandio R., Juste, R.A., García-Pérez A.L., 2010. Seroepidemiological study of Q fever in domestic ruminants in semi-extensive grazing systems. BMC Veterinary Research, 6, 3.
Ruiz, S., and Wolfe, D.N., 2014. Vaccination against Q fever for biodefense and public health indications. Frontiers in Microbiology 5, 726.
Schimmer, B., Ter Schegget, R., Wegdam, M., Züchner, L., de Bruin, A., Schneeberger, P.M., Veenstra, T., Vellema, P., van der Hoek, W., 2010. The use of a geographic information system to identify a dairy goat farm as the most likely source of an urban Q fever outbreak. BMC Infectious Diseases, 10, 69.
Schimmer, B., Schotten, N., van Engelen, E., Hautvast, J.L., Schneeberger, P.M., and van Duijnhoven, Y.T. 2014. Coxiella burnetii seroprevalence and risk for humans on dairy cattle farms, the Netherlands, 2010-2011. Emerging Infectious Diseases, 20, 417-425.
Thrusfield, M., 2007. Veterinary Epidemiology, third edition (Blackwell Publishing: New York, USA).
Tissot-Dupont, H., Amadei, M.A., Nezri, M., Raoult, D., 2007. Wind in November, Q fever in December. Emerging Infectious Diseases, 10, 1264-1269.
Tozer, S.J., Lambert, S.B., Strong, C.L., Field, H.E., Sloots, T.P., Nissen, M.D., 2013. Potential Animal and Environmental Sources of Q fever Infection for Humans in Queensland. Zoonoses Public Health, 61, 105-112.
van den Brom, R., Roest, H.J., de Bruin, A., Dercksen, D., Santman-Berends, I., van der Hoek, W., Dinkla, A., Vellema, J., and Vellema, P., 2015. A Probably Minor Role for Land-Applied Goat Manure in the Transmission of Coxiella burnetii to Humans in the 2007-2010 Dutch Q Fever Outbreak. PLoS One, 10, e0121355.
van Loenhout, J.A., Hautvast, J.L., Vercoulen, J.H., Akkermans, R.P., Wijkmans, C.J., van der Velden, K., and Paget, W.J. 2015. Q-fever patients suffer from impaired health status long after the acute phase of the illness: results from a 24-month cohort study. Epidemiology and Infection, 13, 1-8.
Acknowledgments
We gratefully acknowledge the financial support from Ferdowsi University of Mashhad (grant #29353).
Conflict of interest
We declare that we have no conflict of interest.
Ethical standards
The manuscript does not contain clinical studies or patient data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Janati Pirouz, H., Mohammadi, G., Mehrzad, J. et al. Seroepidemiology of Q fever in one-humped camel population in northeast Iran. Trop Anim Health Prod 47, 1293–1298 (2015). https://doi.org/10.1007/s11250-015-0862-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-015-0862-z