Abstract
This study investigated for the first time the effects of ZnO nanoparticle (NP) chronic exposure (28 days) on Tigriopus fulvus. Acute toxicity (48 h) of three Zn chemical forms was assessed as well including the following: (a) ZnO nanoparticles (NPs), (b) Zn2+ from ZnO NP suspension after centrifugation (supernatant) and (c) ZnSO4 H2O. Physical-chemical and electronic microscopies were used to characterize spiked exposure media. Results showed that the dissolution of ZnO NPs was significant, with a complete dissolution at lowest test concentrations, but nano- and micro-aggregates were always present. Acute test evidenced a significant higher toxicity of Zn2+ and ZnSO4 compared to ZnO NPs. The chronic exposure to ZnO NPs caused negative effects on the reproductive traits, i.e. brood duration, brood size and brood number at much lower concentrations (≥ 100 μg/L). The appearance of ovigerous females was delayed at higher concentrations of ZnO NPs, while the time required for offspring release and the percentage of non-viable eggs per female were significantly increased. ZnO NP subchronic exposure evidenced its ability to reduce T. fulvus individual reproductive fitness, suggesting that ZnO NPs use and release must be carefully monitored.

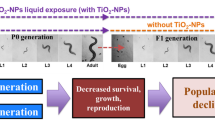
Graphical Abstract.





Similar content being viewed by others
References
Ates M, Daniels J, Arslan Z, Farah IO, Félix-Rivera H (2013) Comparative evaluation of impact of Zn and ZnO nanoparticles on brine shrimp (Artemia salina) larvae: effects of particle size and solubility on toxicity. Environ Sci Process Impacts 15:225–233. https://doi.org/10.1039/c2em30540b
Barata C, Baird DJ, Medina M, Albalat A, Soares AMVM (2002) Determining the ecotoxicological mode of action of toxic chemicals in meiobenthic marine organisms: stage-specific short tests with Tisbe battagliai. Mar Ecol Prog Ser 230:183–194. https://doi.org/10.3354/meps230183
Baun A, Hartmann NB, Grieger K, Kusk KO (2008) Ecotoxicity of engineered nanoparticles to aquatic invertebrates: a brief review and recommendations for future toxicity testing. Ecotoxicology 17:387–395. https://doi.org/10.1007/s10646-008-0208-y
Bernhardt ES, Colman BP, Hochella MF Jr, Cardinale BJ, Nisbet RM, Richardson CJ, Yin L (2010) An ecological perspective on nanomaterial impacts in the environment. J Environ Qual 39:1–12. https://doi.org/10.2134/jeq2009.0479
Biandolino F, Parlapiano I, Faraponova O, Prato E (2018) Effects of short-and long-term exposures to copper on lethal and reproductive endpoints of the harpacticoid copepod Tigriopus fulvus. Ecotoxicol Environ Saf 147:327–333. https://doi.org/10.1016/j.ecoenv.2017.08.041
Blinova I, Ivask A, Heinlaan M, Mortimer M, Kahru A (2010) Ecotoxicity of nanoparticles of CuO and ZnO in natural water. Environ Pollut 158(1):41–47. https://doi.org/10.1016/j.envpol.2009.08.017
Buerki-Thurnherr T, Xiao L, Diener L, Arslan O, Hirsch C, Maeder-Althaus X, Grieder K, Wampfler B, Mathur S, Wick P, Krug H (2013) In vitro mechanistic study towards a better understanding of ZnO nanoparticle toxicity. Nanotoxicology 7:402–416. https://doi.org/10.3109/17435390.2012.666575
Cairns JJ (1992) The threshold problem in ecotoxicology. Ecotoxicology 1:3–16. https://doi.org/10.1007/BF00702652
Cheng JP, Flahaut E, Cheng SH (2007) Effect of carbon nanotubes on developing zebrafish (Danio rerio) embryos. Environ Toxicol Chem 26(4):708–716. https://doi.org/10.1897/06-272R.1
Choi O, Hu Z (2009) Role of reactive oxygen species in determining nitrification inhibition by metallic/oxide nanoparticles. J Environ Eng 135:1365–1370. https://doi.org/10.1061/(ASCE)EE.1943-7870.0000103
EPA, 2014. Method 6020b inductively coupled plasma—mass spectrometry. www.epa.gov
Espanani HR, Faghfoori Z, Izadpanah M, Babadi VY (2015) Toxic effect of nano-zinc oxide. Bratislava Med J 116(10):616–620. https://doi.org/10.4149/BLL_2015_119
Fabrega J, Tantra R, Amer A, Stolpe B, Tomkins J, Fry T, Lead JR, Tyler CR, Galloway TS (2011) Sequestration of zinc from zinc oxide nanoparticles and life cycle effects in the sediment dweller amphipod Corophium volutator. Environ Sci Technol 46(2):1128–1135. https://doi.org/10.1021/es202570g
Faggio C, Tsarpali V, Dailianis S (2018) Mussel digestive gland as a model for assessing xenobiotics: an overview. Sci Total Environ 613:220–229. https://doi.org/10.1016/j.scitotenv.2018.04.264
Fairbairn EA, Keller AA, Madler L, Zhou D, Pokhrel S, Cherr GN (2011) Metal oxide nanomaterials in seawater: linking physicochemical characteristics with biological response in sea urchin development. J Hazard Mater 192:1565–1571. https://doi.org/10.1016/j.jhazmat.2011.06.080
Faraponova O, De Pascale D, Onorati F, Finoia MG (2005) Tigriopus fulvus (Copepoda, Harpacticoida) as a target species in biological assays. Meiofauna Marina 14:91–95
Faraponova O, Giacco E, Biandolino F, Prato E, Del Prete F, Valenti A, Sarcina S, Pasteris A, Montecavalli A, Comin S, Cesca C, Francese M, Cigar M, Piazza V, Falleni F, Lacchetti I (2016) Tigriopus fulvus: the interlaboratory comparison of the acute toxicity test. Ecotoxicol Environ Saf 124:309–314. https://doi.org/10.1016/j.ecoenv.2015.10.013
Franklin NM, Rogers NJ, Apte SC, Batley GE, Gadd GE, Casey PS (2007) Comparative toxicity of nanoparticulate ZnO, bulk ZnO, and ZnCl2 to a freshwater microalga (Pseudokirchneriella subcapitata): the importance of particle solubility. Environ Sci Technol 41:8484–8490. https://doi.org/10.1021/es071445r
Fulda S, Gorman AM, Hori O, Samali A (2010) Cellular stress responses: cell survival and cell death. Int J Cell Biol ID214074:1–23. https://doi.org/10.1155/2010/214074
Garbutt JS, Little TJ (2014) Maternal food quantity affects offspring feeding rate in Daphnia magna. Biol Lett 10:20140356. https://doi.org/10.1098/rsbl.2014.0356
Gerloff K, Albrecht C, Boots AW, Förster I, Schins RPF (2009) Cytotoxicity and oxidative DNA damage by nanoparticles in human intestinal Caco-2 cells. Nanotoxicology 3:355–364. https://doi.org/10.3109/17435390903276933
Greenstein D, Bay S, Anderson B, Chandler G, Farrar J, Keppler C, Phillips B, Ringwood A, Young D (2008) Comparison of methods for evaluating acute and chronic toxicity in marine sediments. Environ Toxicol Chem 27:933–944. https://doi.org/10.1897/07-285.1
Hanna SK, Miller RJ, Muller EB, Nisbet RM, Lenihan HS (2013) Impact of engineered zinc oxide nanoparticles on the individual performance of Mytilus galloprovincialis. PLoS One 8(4):e61800. https://doi.org/10.1371/journal.pone.0061800
Heinlaan M, Ivask A, Blinova I, Dubourguier HC, Kahru A (2008) Toxicity of nanosized and bulk ZnO, CuO and TiO2 to bacteria Vibrio fischeri and crustaceans Daphnia magna and Thamnocephalus platyurus. Chemosphere 71:1308–1316. https://doi.org/10.1016/j.chemosphere.2007.11.047
Huang CW, Li SW, Liao VHC (2017) Chronic ZnO NPs exposure at environmentally relevant concentrations results in metabolic and locomotive toxicities in Caenorhabditis elegans. Environ Pollut 220:1456–1464. https://doi.org/10.1016/j.envpol.2016.10.086
Ingersoll CG, Hutchinson TH, Crane M, Dodson S, DeWitt T, Geis A, Huet MC, McKenney CL, Oberdorster E, Pascoe D, Versteeg D, Warwick O (1999) Laboratory tests for evaluating potential effects of endocrine disrupting compounds. In: de Fur PJ, Crane M, Ingersoll CG, Tattersfield LJ (eds) Endocrine disruption in invertebrates. SETAC Technical Publication, Brussels, pp 107–109 ISBN 1-880611-27-9
Inohaya K, Yasumasu S, Araki K, Naruse K, Yamazaki K, Yasumasu I, Iuchi I, Yamagami K (1997) Species dependent migration of fish hatching gland cells that commonly express astacin-like proteases in common. Develop Growth Differ 39(2):191–197. https://doi.org/10.1046/j.1440-169X.1997.t01-1-00007.x
Jarvis TA, Miller RJ, Lenihan HS, Bielmyer GK (2013) Toxicity of ZnO nanoparticles to the copepod Acartia tonsa, exposed through a phytoplankton diet. Environ Toxicol Chem 32:1264–1269. https://doi.org/10.1002/etc.2180
Kahru A, Dubourguier HC (2010) From ecotoxicology to nanoecotoxicology. Toxicology 269:105–119. https://doi.org/10.1016/j.tox.2009.08.016
Keller AA, Mc Ferran S, Lazareva A, Suh S (2013) Global life cycle releases of engineered nanomaterials. J Nanopart Res 15(6):1–17. https://doi.org/10.1007/s11051-013-1692-4
Keller AA, Vosti W, Wang H, Lazareva A (2014) Release of engineered nanomaterials from personal care products throughout their life cycle. J Nanopart Res 16(7):1–10. https://doi.org/10.1007/s11051-014-2489-9
Khosravi-Katuli K, Lofrano G, Pak Nezhad H, Giorgio A, Guida M, Aliberti F, Siciliano A, Carotenuto M, Galdiero E, Rahimi E, Libralato G (2018) Effects of ZnO nanoparticles in the Caspian roach (Rutilus rutilus caspicus). Sci Total Environ 626:30–41
Kohen R, Nyska A (2002) Oxidation of biological systems: oxidative stress phenomena, antioxidants, redox reactions, and methods for their quantification. Toxicol Pathol 6:620–650. https://doi.org/10.1080/01926230290166724
Kwok KWH, Souissi S, Dur G, Won EJ, Lee JS (2015) Copepods as references species in estuarine and marine waters. In: Amirard-Triquet C, Amiard JC, Mouneyrac C (eds) Aquatic ecotoxicology: advancing tools for dealing with emerging risks. Academic Press, London, pp 281–308. https://doi.org/10.1016/B978-0-12-800949-9.00012-7
Lagido C, McLaggan D, Flett A, Petitt J, Glover LA (2009) Rapid sublethal toxicity assessment using bioluminescent Caenorhabditis elegans, a novel whole-animal metabolic biosensor. Toxicol Sci 109:88–95. https://doi.org/10.1093/toxsci/kfp058
Laufer H, Borst DW (1988) Juvenile hormone in Crustacea. In Invertebrate endocrinology. In: Laufer H, Downer RGH (eds) Endocrinology of selected invertebrate types, vol 2. A. R. Liss, Inc., New York, pp 305–3I3
Li J, Schiavo S, Xiangli D, Rametta G, Miglietta ML, Oliviero M, Changwen W, Manzo S (2018) Early ecotoxic effects of ZnO nanoparticle chronic exposure in Mytilus galloprovincialis revealed by transcription of apoptosis and antioxidant-related genes. Ecotoxicology 27:369–384. https://doi.org/10.1007/s10646-018-1901-0
Ma H, Williams PL, Diamond SA (2013) Ecotoxicity of manufactured ZnO nanoparticles - a review. Environ Pollut 172:76–85. https://doi.org/10.1016/j.envpol.2012.08.011
Mangiapia G, Vitiello G, Irace C, Santamaria R, Colonna A, Angelico R, Radulescu A, D'Errico G, Montesarchio D, Paduano L (2013) Anticancer cationic ruthenium nanovectors: from rational molecular design to cellular uptake and bioactivity. Biomacromolecules 14(8):2549–2560
Manzo S, Miglietta ML, Rametta G, Buono S, Di Francia G (2013a) Toxic effects of ZnO nanoparticles towards marine algae Dunaliella tertiolecta. Sci Total Environ 445–446:371–376. https://doi.org/10.1016/j.scitotenv.2012.12.051
Manzo S, Miglietta ML, Rametta G, Buono S, Di Francia G (2013b) Embryotoxicity and spermiotoxicity of nanosized ZnO for Mediterranean Sea urchin Paracentrotus lividus. J Hazard Mater 254:1–9. https://doi.org/10.1016/j.jhazmat.2013.03.027
Mariani L, De Pascale D, Faraponova O, Tornambè A, Sarni A, Giuliani S, Ruggiero G, Onorati F, Magaletti E (2006) The use of a battery test in marine ecotoxicology: the acute toxicity of sodium dodecyl sulfate. Environ Toxicol 21:373–379. https://doi.org/10.1002/tox.20204
Matranga V, Corsi I (2012) Toxic effects of engineered nanoparticles in the marine environment: model organisms and molecular approaches. Mar Environ Res 76:32–40. https://doi.org/10.1016/j.marenvres.2012.01.006
Mortimer M, Kasemets K, Kahru A (2010) Toxicity of ZnO and CuO nanoparticles to ciliated protozoa Tetrahymena thermophila. Toxicology 269(2–3):182–189. https://doi.org/10.1016/j.tox.2009.07.007
Nawaz HR, Solangi BA, Zehra B, Nadeem U (2011) Preparation of nano zinc oxide and its application in leather as a retanning and antibacterial agent. Can J Sci Ind Res 2(4):164–170
Nel A, Xia T, Mädler L, Li N (2006) Toxic potential of materials at the nanolevel. Science 311(5761):622–627. https://doi.org/10.1126/science.1114397
Nowack B, Bucheli TD (2007) Occurrence, behavior and effects of nanoparticles in the environment. Environ Pollut 150:5–22. https://doi.org/10.1016/j.envpol.2007.06.006
Oberdörster G, Oberdörster E, Oberdörster J (2005) Nanotoxicology: an emerging discipline evolving from studies of ultra fine particle. Environ Health Perspect 113:823–839. https://doi.org/10.1289/ehp.7339
OECD, Organization for Economic Cooperation and Development. 2004 Guideline for the testing of chemicals 202: Daphnia sp., acute immobilisation test. Adopted 13 April 2004. Paris, France
Oliviero M, Schiavo S, Rametta G, Miglietta ML, Manzo S (2017) Different sizes of ZnO diversely affected the cytogenesis of the sea urchin Paracentrotus lividus. Sci Total Environ 2017:176–183. https://doi.org/10.1016/j.scitotenv.2017.07.038
Oukarroum A, Bras S, Perreault F, Popovic R (2012) Inhibitory effects of silver nanoparticles in two green algae, Chlorella vulgaris and Dunaliella tertiolecta. Ecotoxicol Environ Saf 78:80–85. https://doi.org/10.1016/j.ecoenv.2011.11.012
Papageorgiou I, Brown C, Schins R, Singh S, Newson R, Davis S, Fisher J, Ingham E, Case CP (2007) The effect of nano- and micron-sized particles of cobalt-chromium alloy on human fibroblasts in vitro. Biomaterials 28:2946–2958. https://doi.org/10.1016/j.biomaterials.2007.02.034
Park J, Kim S, Yoo J, Lee JS, Park JW, Jung J (2014) Effect of salinity on acute copper and zinc toxicity to Tigriopus japonicus: the difference between metal ions and nanoparticles. Mar Pollut Bull 85:526–531. https://doi.org/10.1016/j.marpolbul.2014.04.038
Parlapiano I, Biandolino F, Russo V, Manfra L, Rotini A, Migliore L, Prato E (2017) Chronic toxicity of ZnO nanoparticles: effects on survival and reproductive traits of the marine copepod Tigriopus fulvus. PeerJ Preprints 5:e3276v1. https://doi.org/10.7287/peerj.preprints.3276v1
Peng X, Palma S, Fisher NS, Wong SS (2011) Effect of morphology of ZnO nanostructures on their toxicity to marine algae. Aquat Toxicol 102:186–196. https://doi.org/10.1016/j.aquatox.2011.01.014
Piccinno F, Gottschalk F, Seeger S, Nowack B (2012) Industrial production quantities and uses of ten engineered nanomaterials for Europe and the world. J Nanopart Res 14:1109. https://doi.org/10.1007/s11051-012-1109-9
Poland CA, Duffin R, Kinloch I, Maynard A, Wallace WA, Seaton A, Stone V, Brown S, Macnee W, Donaldson K (2008) Carbon nanotubes introduced into the abdominal cavity of mice show asbestos-like pathogenicity in a pilot study. Nat Nanotechnol 3:423–428. https://doi.org/10.1038/nnano.2008.111
Poynton HC, Lazorchak JM, Impellitteri CA, Smith ME, Rogers K, Patra M, Hammer KA, Allen HJ, Vulpe CD (2011) Differential gene expression in Daphnia magna suggests distinct modes of action and bioavailability for ZnO nanoparticles and Zn ions. Environ Sci Technol 45(2):762–768. https://doi.org/10.1021/es102501z
Prato E, Biandolino F, Bisci AP, Caroppo C (2011) Preliminary assessment of Ostreopsis cfr. ovata acute toxicity by using a battery bioassay. Chem Ecol 27:117–125. https://doi.org/10.1080/02757540.2011.625930
Prato E, Parlapiano I, Biandolino F (2012) Evaluation of a bioassays battery for ecotoxicological screening of marine sediments. Environ Monit Assess 9:5225–5238. https://doi.org/10.1007/s10661-011-2335-9
Prato E, Parlapiano I, Biandolino F (2013) Assessment of individual and combined toxicities of three metals (Cu, Cd and Hg) by using Tigriopus fulvus. Chem Ecol 29(7):635–642. https://doi.org/10.1080/02757540.2013.817561
Prato E, Parlapiano I, Biandolino F (2015) Ecotoxicological evaluation of sediments by battery bioassays: application and comparison of two integrated classification systems. Chem Ecol 31(7):661–678. https://doi.org/10.1080/02757540.2015.1069278
Raisuddin S, Kwok KWH, Leung KMY, Schlenk D, Lee JS (2007) The copepod Tigriopus: a promising marine model organism for ecotoxicology and environmental genomics. Aquat Toxicol 83:161–173. https://doi.org/10.1016/j.aquatox.2007.04.005
Rotini A, Manfra L, Spanu F, Pisapia M, Cicero AM, Migliore L (2017) Ecotoxicological method with marine bacteria Vibrio anguillarum to evaluate the acute toxicity of environmental contaminants. J Vis Exp 123:e55211. https://doi.org/10.3791/55211
Rotini A, Gallo A, Parlapiano I, Berducci MT, Boni F, Tosti E, Prato E, Maggi C, Cicero AM, Migliore L, Manfra L (2018) Insights into the CuO nanoparticles’ ecotoxicity with suitable marine model species. Ecotoxicol Environ Saf 147:852–860. https://doi.org/10.1016/j.ecoenv.2017.09.053
Scarlett A, Rowland SJ, Canty M, Smith EL, Galloway TS (2007) Method for assessing the chronic toxicity of marine and estuarine sediment-associated contaminants using the amphipod Corophium volutator. Mar Environ Res 63:457–447. https://doi.org/10.1016/j.marenvres.2006.12.006
Schiavo S, Oliviero M, Miglietta M, Rametta G, Manzo S (2016) Genotoxic and cytotoxic effects of ZnO nanoparticles for Dunaliella tertiolecta and comparison with SiO 2 and TiO effects at population growth inhibition levels. Sci Total Environ 550:619–627. https://doi.org/10.1016/j.scitotenv.2016.01.135
Schiavo S, Oliviero M, Li J, Manzo S (2018) Testing ZnO nanoparticles ecotoxicity: linking time variable exposure to effects on different marine model organisms. Environ Sci Pollut Res 25(5):4871–4880. https://doi.org/10.1007/s11356-017-0815-3
Schrurs F, Lison D (2012) Focusing the research efforts. Nat Nanotechnol 7:546–548
Singh S, Shi T, Duffin R, Albrecht C, van Berlo D, Hohr D, Fubini B, Martra G, Fenoglio I, Borm PJ, Schins RP (2007) Endocytosis, oxidative stress and IL-8 expression in human lung epithelial cells upon treatment with fine and ultrafine TiO2: role of the specific surface area and of surface methylation of the particles. Toxicol Appl Pharmacol 222:141–151. https://doi.org/10.1016/j.taap.2007.05.001
Sirelkhatim A, Mahmud S, Seeni A, Kaus NHM, Ann LC, Bakhori SKM, Hasan H, Mohamad D (2015) Review on zinc oxide nanoparticles: antibacterial activity and toxicity mechanism. Nano Micro Lett 7(3):219–242. https://doi.org/10.1007/s40820-015-0040-x
Tornambè A, Manfra L, Mariani L, Faraponova O, Onorati F, Savorelli F, Cicero AM, Virno Lamberti C, Magaletti E (2012) Toxicity evaluation of diethylene glycol and its combined effects with produced waters of off-shore gas platforms in the Adriatic Sea (Italy): bioassays with marine/estuarine species. Mar Environ Res 77:141–149. https://doi.org/10.1016/j.marenvres.2011.12.006
U.S. Environmental Protection Agency/U.S. Army Corps of Engineers (USEPA/USACE). 2001. Methods for assessing the chronic toxicity of marine and estuarine sediment-associated contaminants with the Amphipod Leptocheirus plumulosus. EPA/600/R-01/020. Washington
UNICHIM 2396. 2014. Qualità dell’acqua-Determinazione della tossicità letale a 24 h, 48 h e 96 h di esposizione con naupli di Tigriopus fulvus (Fischer, 1860) (Crustacea: Copepoda) [Water quality-Determination of lethal toxicity at 24, 48, 96 hrs exposition in Tigriopus fulvus nauplii]
Vajargah MF, Yalsuyi AM, Hedayati A, Faggio C (2018) Histopathological lesions and toxicity in common carp (Cyprinus carpio L. 1758) induced by copper nanoparticles. Microsc Res Tech 81(7):724–729. https://doi.org/10.1002/jemt.23028
Wang H, Wick RL, Xing B (2009) Toxicity of nanoparticulate and bulk ZnO, Al2O3 and TiO2 to the nematode Caenorhabditis elegans. Environ Pollut 157:1171–1177. https://doi.org/10.1016/j.envpol.2008.11.004
Warnau M, Temara A, Jangoux M, Dubois P, Iaccarino M, De Biase A, Pagano G (1996) Spermiotoxicity and embryotoxicity of heavy metals in the echinoid Paracentrotus lividus. Environ Toxicol Chem 15:1931–1936. https://doi.org/10.1002/etc.5620151111
Wong SWY, Leung PTY, Djurišić AB, Leung KMY (2010) Toxicities of nano zinc oxide to five marine organisms: influences of aggregate size and ion solubility. Anal Bioanal Chem 396:609–618. https://doi.org/10.1007/s00216-009-3249-z
Xia T, Kovochich M, Liong M, Mädler L, Gilbert B, Shi H, Yeh JI, Zink JI, Nel AE (2008) Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS Nano 2:2121–2134. https://doi.org/10.1021/nn800511k
Yi X, Chi T, Liu B, Liu C, Feng G, Dai X, Zhou H (2019) Effect of nano zinc oxide on the acute and reproductive toxicity of cadmium and lead to the marine copepod Tigriopus japonicus. Comp Biochem Physiol C Toxicol Pharmacol 222:118–124
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Cinta Porte
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Acute LC50s: ZnO NPs < Zn2+~ZnSO4.
• Sublethal ZnO NPs caused a delay appearance of ovigerous females.
• Chronic exposure reduced nauplii per brood
• Sublethal concentration of ZnO NPs could affect the population by reducing individual reproductive fitness.
Electronic supplementary material
Supplementary Figure 1
(DOCX 87 kb)
Supplementary Figure 2
(DOCX 51 kb)
Rights and permissions
About this article
Cite this article
Prato, E., Parlapiano, I., Biandolino, F. et al. Chronic sublethal effects of ZnO nanoparticles on Tigriopus fulvus (Copepoda, Harpacticoida). Environ Sci Pollut Res 27, 30957–30968 (2020). https://doi.org/10.1007/s11356-019-07006-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-07006-9