Abstract
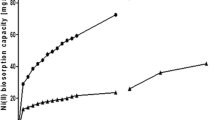
Lessonia nigrescens and Lessonia trabeculata kelps have been tested for the sorption of mercury from aqueous solutions. A pretreatment (using CaCl2) allowed stabilizing the biomass that was very efficient for removing Hg(II) at pH 6–7. Sorption isotherms were described by the Langmuir equation with sorption capacities close to 240–270 mg Hg g−1 at pH 6. The temperature had a negligible effect on the distribution of the metal at equilibrium. The presence of chloride anions had a more marked limiting impact than sulfate and nitrate anions. The uptake kinetics were modeled using the pseudo-second-order equation that fitted better experimental data than the pseudo-first-order equation. The particle size hardly influenced sorption isotherms and uptake kinetics, indicating that sorption occurs in the whole mass of the biosorbent and that intraparticle mass transfer resistance was not the limiting rate. Varying the sorbent dosage and the initial metal concentration influenced the equilibrium, but the kinetic parameters were not drastically modified. Metal can be eluted with hydrochloric acid, citric acid, or acidic KI solutions.











Similar content being viewed by others
References
Patterson, J. (1997). Aqueous Mercury Treatment. Washington: U.S.E.P.A.
Matlock, M. M., Howerton, B. S., & Atwood, D. A. (2001). Journal of Hazardous Materials, B84, 73–82.
Kuncoro, E. P., Roussy, J., & Guibal, E. (2005). Sep. Sci. Technol., 40, 659–684.
Duche, S. N., Pawar, S. D., & Dhadke, P. M. (2002). Separation Science and Technology, 37, 2215–2229.
Meera, R., Francis, T., & Reddy, M. L. P. (2001). Hydrometallurgy, 61, 93–103.
Vieira, R. S., Guibal, E., Silva, E. A., & Beppu, M. M. (2007). Adsorption-Journal of the International Adsorption Society, 13, 603–611.
Atia, A. A., Donia, A. M., & Elwakeel, K. Z. (2005). Reactive and Functional Polymers, 65, 267–275.
Guibal, E., Gavilan, K. C., Bunio, P., Vincent, T., & Trochimczuk, A. (2007). Separation Science and Technology, 43, 2406–2433.
Volesky, B., & Holan, Z. R. (1995). Biotechnology Progress, 11, 235–250.
Zeroual, Y., Moutaouakkil, A., Dzairi, F. Z., Talbi, M., Chung, P. U., Lee, K., et al. (2003). Bioresource Technology, 90, 349–351.
Vilar, V. J. P., Botelho, C. M. S., & Boaventura, R. A. R. (2008). Biochemical Engineering Journal, 38, 319–325.
Svecova, L., Spanelova, M., Kubal, M., & Guibal, E. (2006). Separation and Purification Technology, 52, 142–153.
Vijayaraghavan, K., & Yun, Y.-Y. (2008). Biotechnology Advances, 26, 266–291.
de França, F., Padilha, F., & da Costa, A. (2006). Applied Biochemistry and Biotechnology, 128, 23–32.
Michalak, I. & Chojnacka, K. (2009). Applied Biochemistry and Biotechnology. doi:10.1007/s12010-12009-18635-12017.
Picardo, M., de Melo Ferreira, A., & da Costa, A. (2006). Applied Biochemistry and Biotechnology, 134, 193–206.
Romera, E., González, F., Ballester, A., Blázquez, M. L., & Muñoz, J. A. (2007). Bioresource Technology, 98, 3344–3353.
Tajes-Martinez, P., Beceiro-Gonzalez, E., Muniategui-Lorenzo, S., & Prada-Rodriguez, D. (2006). Talanta, 68, 1489–1496.
Fourest, E., & Volesky, B. (1997). Applied Biochemistry and Biotechnology, 67, 215–226.
Dubois, M. A., Dozol, J. F., & Massiani, C. (1995). Journal of Analytical and Applied Pyrolysis, 31, 129–140.
Reategui, M., Maldonado, H., Ly, M., & Guibal, E. (2009). Advances in Materials Research, 71–73, 585–588.
Decarvalho, R. P., Chong, K. H., & Volesky, B. (1994). Biotechnological Letters, 16, 875–880.
Matheickal, J. T., Yu, Q., & Woodburn, G. M. (1999). Water Research, 33, 335–342.
Ho, Y. S. (2006). Water Research, 40, 119–125.
Liu, Y., & Liu, Y.-Y. (2007). Separation and Purification Technology, 61, 229–242.
Mata, Y. N., Blázquez, M. L., Ballester, A., González, F., & Muñoz, J. A. (2008). Journal of Hazardous Materials, 158, 316–323.
Davis, T. A., Volesky, B., & Mucci, A. (2003). Water Research, 37, 4311–4330.
Malik, D. J., Streat, M., & Greig, J. (1999). Institution of Chemical Engineers Translations, 77, 227–233.
Hansen, H. K., Ribeiro, A., & Mateus, E. (2006). Minerals Engineering, 19, 486–490.
Ghodbane, I., & Hamdaoui, O. (2008). Journal of Hazardous Materials, 160, 301–309.
Kaçar, Y., Arpa, C., Tan, S., Denizli, A., Genç, O., & Arica, Y. (2002). Process Biochemistry, 37, 601–610.
Bayramoglu, G., Tuzun, I., Celik, G., Yilmaz, M., & Arica, M. Y. (2006). International Journal of Mineral Processing, 81, 35–43.
Lloyd-Jones, P. J., Rangel-Mendez, J. R., & Streat, M. (2004). Institution of Chemical Engineers Translations, 82B, 301–311.
Pavasant, P., Apiratikul, R., Sungkhum, V., Suthiparinyanont, P., Wattanachira, S., & Marhaba, T. F. (2006). Bioresource Technology, 97, 2321–2329.
Prasanna Kumar, Y., King, P., & Prasad, V. S. R. K. (2007). Chemical Engineering Journal, 129, 161–166.
Make Equilibrium Diagrams Using Sophisticated Algorithms (MEDUSA) (2002) v. 3.1. http://www.kemi.kth.se/medusa. Accessed 7 February 2007.
Chen, J. Z., Tao, X. C., Xu, J., Zhang, T., & Liu, Z. L. (2005). Process Biochemistry, 40, 3675–3679.
Acknowledgements
Authors thank the European Commission for the funding of the project BIOPROAM (Contract no. AML/190901/06/18414/II-0548-FC-FA, in the framework of ALFA program). Authors acknowledge Jean-Marie Taulemesse (Centre des Matériaux de Grande Diffusion at Ecole des Mines d’Alès) for his technical support for SEM-EDAX analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reategui, M., Maldonado, H., Ly, M. et al. Mercury(II) Biosorption Using Lessonia sp. Kelp. Appl Biochem Biotechnol 162, 805–822 (2010). https://doi.org/10.1007/s12010-010-8912-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-010-8912-5