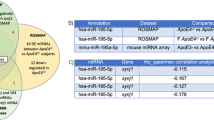
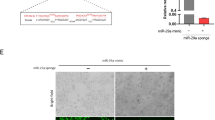
Abstract
Aging is characterized by progressive memory decline that can lead to dementia when associated with neurodegeneration. Here, we show in mice that aging-related memory decline involves defective biogenesis of microRNAs (miRNAs), in particular miR-183/96/182 cluster, resulting from increased protein phosphatase 1 (PP1) and altered receptor SMAD (R-SMAD) signaling. Correction of the defect by miR-183/96/182 overexpression in hippocampus or by environmental enrichment that normalizes PP1 activity restores memory in aged animals. Regulation of miR-183/96/182 biogenesis is shown to involve the neurodegeneration-related RNA-binding proteins TDP-43 and FUS. Similar alterations in miR-183/96/182, PP1, and R-SMADs are observed in the brains of patients with amyotrophic lateral sclerosis (ALS) or frontotemporal lobar degeneration (FTLD), two neurodegenerative diseases with pathological aggregation of TDP-43. Overall, these results identify new mechanistic links between miR-183/96/182, PP1, TDP-43, and FUS in age-related memory deficits and their reversal.





Similar content being viewed by others
References
Blahna MT, Hata A (2012) Smad-mediated regulation of microRNA biosynthesis. FEBS Lett 586:1906–1912
Bilan V, Allegra D, Kuchenbauer F, Mertens D (2014) In vivo processing assay based on a dual-luciferase reporter system to evaluate DROSHA enzymatic activity. Methods Mol Biol 1095:87–93
Bishop NA, Lu T, Yankner BA (2010) Neural mechanisms of ageing and cognitive decline. Nature 464:529–535
Capitano F, Camon J, Licursi V, Ferretti V, Maggi L, Scianni M, del Vecchio G, Rinaldi A et al (2017) MicroRNA-335-5p modulates spatial memory and hippocampal synaptic plasticity. Neurobiol Learn Mem 139:63–68
Dannhauser TM, Cleverley M, Whitfield TJ, Fletcher B, Stevens T, Walker Z (2014) A complex multimodal activity intervention to reduce the risk of dementia in mild cognitive impairment--ThinkingFit: pilot and feasibility study for a randomized controlled trial. BMC Psychiatry 14:129
Davis BN, Hilyard AC, Lagna G, Hata A (2008) SMAD proteins control DROSHA-mediated microRNA maturation. Nature 454:56–61
Davis BN, Hilyard AC, Nguyen PH, Lagna G, Hata A (2010) Smad proteins bind a conserved RNA sequence to promote microRNA maturation by Drosha. Mol Cell 39:373–384
De Conti L et al (2015) TDP-43 affects splicing profiles and isoform production of genes involved in the apoptotic and mitotic cellular pathways. Nucleic Acids Res 43:8990–9005
Di Carlo V et al (2013) TDP-43 regulates the microprocessor complex activity during in vitro neuronal differentiation. Mol Neurobiol 48:952–963
Dong C-M et al (2014) A stress-induced cellular aging model with postnatal neural stem cells. Cell Death Dis 5:e1116
Ford L, Crossley M, Williams T, Thorpe JR, Serpell LC, Kemenes G (2015) Effects of Aβ exposure on long-term associative memory and its neuronal mechanisms in a defined neuronal network. Sci Rep 5:10614
Genoux D, Haditsch U, Knobloch M, Michalon A, Storm D, Mansuy IM (2002) Protein phosphatase 1 is a molecular constraint on learning and memory. Nature 418:970–975
Gräff J, Rei D, Guan JS, Wang WY, Seo J, Hennig KM, Nieland TJF, Fass DM et al (2012) An epigenetic blockade of cognitive functions in the neurodegenerating brain. Nature 483:222–226
Hedden T, Gabrieli JDE (2004) Insights into the ageing mind: a view from cognitive neuroscience. Nat Rev Neurosci 5:87–96
Hernandez-Rapp J, Smith PY, Filali M, Goupil C, Planel E, Magill ST, Goodman RH, Hébert SS (2015) Memory formation and retention are affected in adult miR-132/212 knockout mice. Behav Brain Res 287:15–26
Hernandez-Rapp J, Rainone S, Hébert SS (2017) MicroRNAs underlying memory deficits in neurodegenerative disorders. Prog Neuro-Psychopharmacology Biol Psychiatry 73:79–86
Hudson MB et al (2014) miR-182 attenuates atrophy-related gene expression by targeting FoxO3 in skeletal muscle. Am J Physiol Cell Physiol 307:C314–C319
Inukai S, de Lencastre A, Turner M, Slack F (2012) Novel microRNAs differentially expressed during aging in the mouse brain. PLoS One 7:e40028
Ishihara H, Martin BL, Brautigan DL, Karaki H, Ozaki H, Kato Y, Fusetani N, Watabe S et al (1989) Calyculin A and okadaic acid: inhibitors of protein phosphatase activity. Biochem Biophys Res Commun 159:871–877
Kawahara Y, Mieda-Sato A (2012) TDP-43 promotes microRNA biogenesis as a component of the Drosha and Dicer complexes. Proc Natl Acad Sci U S A 109:3347–3352
Kremer EA, Gaur N, Lee MA, Engmann O, Bohacek J, Mansuy IM (2018) Interplay between TETs and microRNAs in the adult brain for memory formation. Sci Rep 8:1678
Koshibu K, Graff J, Beullens M, Heitz FD, Berchtold D, Russig H, Farinelli M, Bollen M et al (2009) Protein phosphatase 1 regulates the histone code for long-term memory. J Neurosci 29:13079–13089
Kosik KS, Rapp PR, Raz N, Small SA, Sweatt JD, Tsai LH (2012) Mechanisms of age-related cognitive change and targets for intervention: epigenetics. J Gerontol A Biol Sci Med Sci 67:741–746
Kye MJ, Niederst ED, Wertz MH, Gonçalves ICG, Akten B, Dover KZ, Peters M, Riessland M et al (2014) SMN regulates axonal local translation via miR-183/mTOR pathway. Hum Mol Genet 23:6318–6331
Li Y et al (2016) miR-182 (microRNA-182) suppression in the hippocampus evokes antidepressant-like effects in rats. Prog Neuropsychopharmacol Biol Psychiatry 65:96–103
Luttenberger K, Hofner B, Graessel E (2012) Are the effects of a non-drug multimodal activation therapy of dementia sustainable? Follow-up study 10 months after completion of a randomised controlled trial. BMC Neurol 12:151
Meerson A, Cacheaux L, Goosens KA, Sapolsky RM, Soreq H, Kaufer D (2010) Changes in brain MicroRNAs contribute to cholinergic stress reactions. J Mol Neurosci 40:47–55
Michely J, Kraft S, Müller U (2017) miR-12 and miR-124 contribute to defined early phases of long-lasting and transient memory. Sci Rep 7:7910
Morlando M, Dini Modigliani S, Torrelli G, Rosa A, di Carlo V, Caffarelli E, Bozzoni I (2012) FUS stimulates microRNA biogenesis by facilitating co-transcriptional Drosha recruitment. EMBO J 31:4502–4510
Munton RP, Vizi S, Mansuy IM (2004) The role of protein phosphatase-1 in the modulation of synaptic and structural plasticity. FEBS Lett 567:121–128
Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, Bruce J, Schuck T et al (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314:130–133
Neumann M, Rademakers R, Roeber S, Baker M, Kretzschmar HA, Mackenzie IRA (2009) A new subtype of frontotemporal lobar degeneration with FUS pathology. Brain 132:2922–2931
Perera ND, Sheean RK, Scott JW, Kemp BE, Horne MK, Turner BJ (2014) Mutant TDP-43 deregulates AMPK activation by PP2A in ALS models. PLoS One 9:e90449
Pavlopoulos E et al (2013) Molecular mechanism for age-related memory loss: the histone-binding protein RbAp48. Sci Transl Med 5:200ra115
Rabkin JG, Albert SM, del Bene ML, O'Sullivan I, Tider T, Rowland LP, Mitsumoto H (2005) Prevalence of depressive disorders and change over time in late-stage ALS. Neurology 65:62–67
Ringholz GM, Appel SH, Bradshaw M, Cooke NA, Mosnik DM, Schulz PE (2005) Prevalence and patterns of cognitive impairment in sporadic ALS. Neurology 65:586–590
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C (T) method. Nat Protoc 3:1101–1108
Schwartz JC, Ebmeier CC, Podell ER, Heimiller J, Taatjes DJ, Cech TR (2012) FUS binds the CTD of RNA polymerase II and regulates its phosphorylation at Ser2. Genes Dev 26:2690–2695
Spires TL, Orne JD, SantaCruz K, Pitstick R, Carlson GA, Ashe KH, Hyman BT (2006) Region-specific dissociation of neuronal loss and neurofibrillary pathology in a mouse model of tauopathy. Am J Pathol 168:1598–1607
Stanford PM, Brooks WS, Teber ET, Hallupp M, McLean C, Halliday GM, Martins RN, Kwok JB et al (2004) Frequency of tau mutations in familial and sporadic frontotemporal dementia and other tauopathies. J Neurol 251:1098–1104
Witgert M, Salamone AR, Strutt AM, Jawaid A, Massman PJ, Bradshaw M, Mosnik D, Appel SH et al (2010) Frontal-lobe mediated behavioral dysfunction in amyotrophic lateral sclerosis. Eur J Neurol 17:103–110
Woldemichael BT, Jawaid A, Kremer EA, Gaur N, Krol J, Marchais A, Mansuy IM (2016) The microRNA cluster miR-183/96/182 contributes to long-term memory in a protein phosphatase 1-dependent manner. Nat Commun 7:12594
Woldemichael BT, Mansuy IM (2016) Micro-RNAs in cognition and cognitive disorders: Potential for novel biomarkers and therapeutics. Biochem Pharmacol 104:1–7
Xu XF et al (2017) miR-181a participates in contextual fear memory formation via activating mTOR signaling pathway. Cereb Cortex 22:1–13
Acknowledgments
We thank John Ravits (University of California, San Diego) for generously providing human autopsy tissues for analysis; Vinnie Kandra, Jennifer Brown, and Mattia Privitera for technical help; Jean-Charles Paterna and Jacek Krol for virus production, animal caretakers for mouse colony management.
Funding
This work was supported by the University of Zurich, the Swiss Federal Institute of Technology, the Swiss National Science Foundation (grant No. 31003A_153147/1). M.P. is supported by the Swiss National Science Foundation (grant No. PP00P3_144862 and NCCR RNA & Disease) and a Clinical Research Priority Program on Small RNAs by the University of Zurich.
Author information
Authors and Affiliations
Contributions
A. J. and I. M. M. conceived and designed the study, analyzed the results, and wrote the manuscript. A. J conducted behavioral experiments, molecular analyses of brain samples, and qPCR assays, and designed and executed in vitro experiments. B. T. W. performed stereotaxic injections and helped in the interpretation and drafting of in vivo data. E. A. K. conducted environmental enrichment and luciferase assays with A. J. F. L. performed Western blots and immunohistochemistry. N. G. assisted A. J. and E. A. K. in qPCR and luciferase assays. T. A. extracted RNA from human samples. M. P. provided human samples with TDP-43 pathology and critical input to the manuscript.
Corresponding author
Electronic Supplementary Material
ESM 1
(DOCX 607 kb)
Rights and permissions
About this article
Cite this article
Jawaid, A., Woldemichael, B.T., Kremer, E.A. et al. Memory Decline and Its Reversal in Aging and Neurodegeneration Involve miR-183/96/182 Biogenesis. Mol Neurobiol 56, 3451–3462 (2019). https://doi.org/10.1007/s12035-018-1314-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1314-3