Abstract
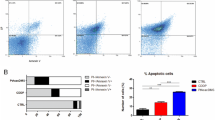
Patients treated with the cisplatin often develop strong resistance to the drug after prolonged treatments, ultimately resulting in limited clinical efficacy. One of the possible mechanisms is that the internalized compound may be inactivated before getting access to the nucleus where cisplatin forms a complex with the genomic DNA and triggers a cell death program. However, the nature and intracellular fate of inactivated cisplatin is poorly illustrated. In the present study, we reported for the first time the presence of platinum nanoparticles (Pt-NPs) in the cytoplasm of cells treated with cisplatin. Further analysis also evidenced a correlation of the increased intracellular Pt-NPs formation with cisplatin resistance, and confirmed the process was glutathione S-transferase relevant. Our data suggest that tumor cells may develop cisplatin resistance by converting the drug into less toxic intracellular Pt-NPs, thereby impeding the drug from targeting its substrates.
中文摘要
长期使用顺铂治疗的肿瘤患者容易产生肿瘤细胞耐药性, 影响顺铂的临床治疗效果. 顺铂进入肿瘤细胞后, 在到达靶向细胞 核之前可能会失去活性, 失活后的药物将无法与DNA络合以杀伤细胞, 这是细胞产生药物耐受性的可能机制之一. 然而失活后的顺铂 在细胞内以何种形式存在这一问题尚没有得到完全阐明. 本论文研究发现, 顺铂治疗后的细胞中有铂纳米颗粒存在, 而且该类颗粒的 形成与细胞对顺铂的耐药性有一定的关系. 进一步研究发现细胞内谷胱甘肽转移酶可能在这一机制中起到了重要作用. 数据表明, 细 胞可能通过将高细胞毒性的顺铂转化为低毒性的铂纳米颗粒, 起到阻止顺铂靶向其底物的作用.
Similar content being viewed by others
References
Prestayko AW, Daoust JC, Issell BF, et al. Cisplatin (cis-diamminedichloroplatinum-II). Cancer Treat Rev, 1979, 6: 17–39
Chen X, Wu Y, Dong H, et al. Platinum-based agents for individualized cancer treatment. Curr Mol Med, 2013, 13: 1603–1612
Maccio A, Madeddu C. Cisplatin: an old drug with a newfound efficacy—from mechanisms of action to cytotoxicity. Expert Opin Pharmaco, 2013, 14: 1839–1857
Stathopoulos GP. Cisplatin: process and future. J Buon, 2013, 18: 564–569
Perry DJ, Weltz MD, Brown AW, et al. Vinblastine, bleomycin and cisplatin for recurrent or metastatic squamous-cell carcinoma of the head and neck. Cancer, 1982, 50: 2257–2260
Leipzig B. Cisplatin sensitization to radiotherapy of squamous-cell carcinomas of the head and neck. Am J Surg, 1983, 146: 462–465
Brizel DM, Albers ME, Fisher SR, et al. Hyperfractionated irradiation with or without concurrent chemotherapy for locally advanced head and neck cancer. New Engl J Med, 1998, 338: 1798–1804
Bernier J, Domenge C, Ozsahin M, et al. Postoperative irradiation with or without concomitant chemotherapy for locally advanced head and neck cancer. New Engl J Med, 2004, 350: 1945–1952
Khuri FR, Nemunaitis J, Ganly I, et al. A controlled trial of intratumoral ONYX-015, a selectively-replicating adenovirus, in combination with cisplatin and 5-fluorouracil in patients with recurrent head and neck cancer. Nat Med, 2000, 6: 879–885
Posner MR, Hershock DM, Blajman CR, et al. Cisplatin and fluorouracil alone or with docetaxel in head and neck cancer. New Engl J Med, 2007, 357: 1705–1715
Jamieson ER, Lippard SJ. Structure, recognition, and processing of cisplatin-DNA adducts. Chem Rev, 1999, 99: 2467–2498
Jordan P, Carmo-Fonseca M. Molecular mechanisms involved in cisplatin cytotoxicity. Cell Mol Life Sci, 2000, 57: 1229–1235
Cepeda V, Fuertes MA, Castilla J, et al. Biochemical mechanisms of cisplatin cytotoxicity. Anticancer Agents Med Chem, 2007, 7: 3–18
Dasari S, Tchounwou PB. Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol, 2014, 740: 364–378
Siddik ZH. Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene, 2003, 22: 7265–7279
Shen DW, Pouliot LM, Hall MD, et al. Cisplatin resistance: a cellular self-defense mechanism resulting from multiple epigenetic and genetic changes. Pharmacol Rev, 2012, 64: 706–721
Galluzzi L, Senovilla L, Vitale I, et al. Molecular mechanisms of cisplatin resistance. Oncogene, 2012, 31: 1869–1883
Galluzzi L, Vitale I, Michels J, et al. Systems biology of cisplatin resistance: past, present and future. Cell Death Dis, 2014, 5: e1257
Arnesano F, Losacco M, Natile G. An updated view of cisplatin transport. Eur J Inorg Chem, 2013, 2701–2711
Katano K, Kondo A, Safaei R, et al. Acquisition of resistance to cisplatin is accompanied by changes in the cellular pharmacology of copper. Cancer Res, 2002, 62: 6559–6565
Tsai CY, Larson CA, Safaei R, et al. Molecular modulation of the copper and cisplatin transport function of CTR1 and its interaction with IRS-4. Biochem Pharmacol, 2014, 90: 379–387
Nishimura T, Newkirk K, Sessions RB, et al. Immunohistochemical staining for glutathione S-transferase predicts response to platinum- based chemotherapy in head and neck cancer. Clin Cancer Res, 1996, 2: 1859–1865
Cullen KJ, Newkirk KA, Schumaker LM, et al. Glutathione S-transferase p amplification is associated with cisplatin resistance in head and neck squamous cell carcinoma cell lines and primary tumors. Cancer Res, 2003, 63: 8097–8102
Shiga H, Heath EI, Rasmussen AA, et al. Prognostic value of p53, glutathione S-transferase p, and thymidylate synthase for neoadjuvant cisplatin-based chemotherapy in head and neck cancer. Clin Cancer Res, 1999, 5: 4097–4104
Chiu CY, Ruan L, Huang Y. Biomolecular specificity controlled nanomaterial synthesis. Chem Soc Rev, 2013, 42: 2512–2527
Faramarzi MA, Sadighi A. Insights into biogenic and chemical production of inorganic nanomaterials and nanostructures. Adv Colloid Interface Sci, 2013, 189–190: 1–20
Konishi Y, Ohno K, Saitoh N, et al. Bioreductive deposition of platinum nanoparticles on the bacterium shewanella algae. J Biotechnol, 2007, 128: 648–653
Song JY, Kwon EY, Kim BS. Biological synthesis of platinum nanoparticles using Diopyros kaki leaf extract. Bioprocess Biosyst Eng, 2010, 33: 159–164
Venu R, Ramulu TS, Anandakumar S, et al. Bio-directed synthesis of platinum nanoparticles using aqueous honey solutions and their catalytic applications. Colloids and Surfaces A: Physicochem Eng Aspects, 2011, 384: 733–738
Govender Y, Riddin TL, Gericke M, et al. On the enzymatic formation of platinum nanoparticles. J Nanopart Res, 2010, 12: 261–271
Aika K, Ban LL, Okura I, et al. Chemisorption and catalytic activity of a set of platinum catalysts. J Res Inst Catalysis Hokkaido Univ, 1976, 24: 54–64
Li M, Yang DP, Wang XS, et al. Mixed protein-templated luminescent metal clusters (Au and Pt) for H2O2 sensing. Nanoscale Res Lett, 2013, 8: 182
Li YJ, Whyburn GP, Huang Y. Specific peptide regulated synthesis of ultrasmall platinum nanocrystals. J Am Chem Soc, 2009, 131: 15998–15999
Konieczny P, Goralczyk AG, Szmyd R, et al. Effects triggered by platinum nanoparticles on primary keratinocytes. Int J Nanomed, 2013, 8: 3963–3975
Yamagishi Y, Watari A, Hayata Y, et al. Acute and chronic nephrotoxicity of platinum nanoparticles in mice. Nanoscale Res Lett, 2013, 8: 395
Elder A, Yang H, Gwiazda R, et al. Testing nanomaterials of unknown toxicity: an example based on platinum nanoparticles of different shapes. Adv Mater, 2007, 19: 3124–3129
Asharani PV, Xinyi N, Hande MP, et al. DNA damage and p53-mediated growth arrest in human cells treated with platinum nanoparticles. Nanomedicine, 2010, 5: 51–64
Asharani PV, Yi LW, Gong ZY, et al. Comparison of the toxicity of silver, gold and platinum nanoparticles in developing zebrafish embryos. Nanotoxicology, 2011, 5: 43–54
Gehrke H, Pelka J, Hartinger CG, et al. Platinum nanoparticles and their cellular uptake and DNA platination at non-cytotoxic concentrations. Arch Toxicol, 2011, 85: 799–812
Horie M, Kato H, Endoh S, et al. Evaluation of cellular influences of platinum nanoparticles by stable medium dispersion. Metallomics, 2011, 3: 1244–1252
Tsuchida S, Sato K. Glutathione transferases and cancer. Crit Rev Biochem Mol Biol, 1992, 27: 337–384
Hayes JD, Pulford DJ. The glutathione s-transferase supergene family: regulation of gst and the contribution of the isoenzymes to cancer chemoprotection and drug resistance. Crit Rev Biochem Mol Biol, 1995, 30: 445–600
Townsend DM, Tew KD. The role of glutathione-s-transferase in anti-cancer drug resistance. Oncogene, 2003, 22: 7369–7375
Henglein A, Ershov BG, Malow M. Absorption-spectrum and some chemical-reactions of colloidal platinum in aqueous-solution. J Phys Chem, 1995, 99: 14129–14136
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work.
Meng Cao was born in 1983. He received his PhD degree in biomedical engineering from the School of Biological Science & Medical Engineering, Southeast University, Nanjing, China, in 2013. Currently he is a postdoctoral fellow in Professor Ning Gu’s group. His research interests mainly focus on biochemistry and anti-tumor therapeutics.
Ning Gu was born in 1964. He received his PhD degree in biomedical engineering from the Department of Biomedical Engineering, Southeast University, Nanjing, China, in 1996. Currently he is a Changjiang Scholar Professor and NSFC Outstanding Young Investigator Fund Winner at the School of Biological Science and Medical Engineering, Southeast University. He also serves as the president of Jiangsu Society of Biomedical Engineering, the director of the Research Center for Nanoscale Science and Technology of Southeast University. His research interests include biomaterials, nanobiology, medical imaging, and advanced instrument development.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Cao, M., Liu, F., Zhang, X. et al. The formation of intracellular nanoparticles correlates with cisplatin resistance. Sci. China Mater. 58, 640–648 (2015). https://doi.org/10.1007/s40843-015-0073-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-015-0073-y