ABSTRACT
Background
The presence of tumor-infiltrating lymphocytes (TILs) in breast tumors is prognostic and predictive, suggesting that TILs may be an important biomarker. Recently, an international TILs working group formulated consensus recommendations for TIL evaluation. The current study was performed to determine interobserver agreement using that methodology.
Methods
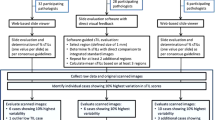
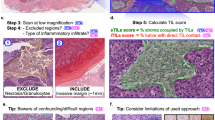
Tumor-infiltrating lymphocytes were assessed on a single hematoxylin and eosin (H&E)-stained slide obtained from the core biopsy of 75 triple-negative breast cancers. Four pathologists independently reviewed each slide and evaluated stromal TILs (sTILs) and intratumoral TIL (iTILs). The kappa statistic was used to estimate interobserver agreement for identification of sTILs, and the intraclass correlation coefficient (ICC) was used to estimate the agreement among observers for iTILs. Cases with poor agreement were reviewed to identify pathologic factors that may contribute to the lack of agreement.
Results
The kappa statistic for sTIL evaluation was 0.57 (standard error, 0.04). For iTILs, the ICC calculated to determine internal consistency within raters was 0.65 (95 % confidence interval [CI] 0.56–0.74; p < 0.0001), and the ICC calculated to determine agreement among raters was 0.62 (95 % CI 0.50–0.72; p < 0.0001). In 10 cases (13 %), there was not agreement between three of four pathologists. The pathologic features contributing to difficulty in TIL enumeration included marked individual tumor cell necrosis or apoptosis, the presence of reactive plasma cells mimicking tumor cells, plasmatoid tumor cells, and accurate quantification of TILs in specimens with focal areas of heavy immune infiltrate.
Conclusion
Acceptable agreement in TIL enumeration was observed, suggesting that the proposed methodology can be used to facilitate the use of TILs as a biomarker in research and clinical trial settings.




Similar content being viewed by others
References
Adams S, Gray RJ, Demaria S, et al. Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J Clin Oncol. 2014;32:2959–66.
Ali HR, Provenzano E, Dawson SJ, et al. Association between CD8+ T-cell infiltration and breast cancer survival in 12,439 patients. Ann Oncol. 2014;25:1536–43.
Dieci MV, Criscitiello C, Goubar A, et al. Prognostic value of tumor-infiltrating lymphocytes on residual disease after primary chemotherapy for triple-negative breast cancer: a retrospective multicenter study. Ann Oncol. 2015;26:1518.
Perez EA, Ballman KV, Tenner KS, et al. Association of stromal tumor-infiltrating lymphocytes with recurrence-free survival in the N9831 adjuvant trial in patients with early-stage HER2-positive breast cancer. JAMA Oncol. 2016;2:56–64.
Salgado R, Denkert C, Campbell C, et al. Tumor-infiltrating lymphocytes and associations with pathological complete response and event-free survival in HER2-positive early-stage breast cancer treated with lapatinib and trastuzumab: a secondary analysis of the NeoALTTO Trial. JAMA Oncol. 2015;1:448–54.
Loi S, Michiels S, Salgado R, et al. Tumor-infiltrating lymphocytes are prognostic in triple-negative breast cancer and predictive for trastuzumab benefit in early breast cancer: results from the FinHER trial. Ann Oncol. 2014;25:1544–50.
Loi S, Sirtaine N, Piette F, et al. Prognostic and predictive value of tumor-infiltrating lymphocytes in a phase III randomized adjuvant breast cancer trial in node-positive breast cancer comparing the addition of docetaxel to doxorubicin with doxorubicin-based chemotherapy: BIG 02-98. J Clin Oncol. 2013;31:860–7.
Denkert C, Loibl S, Noske A, et al. Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J Clin Oncol. 2010;28:105–13.
Denkert C, von Minckwitz G, Brase JC, et al. Tumor-infiltrating lymphocytes and response to neoadjuvant chemotherapy with or without carboplatin in human epidermal growth factor receptor 2-positive and triple-negative primary breast cancers. J Clin Oncol. 2015;33:983–91.
Issa-Nummer Y, Darb-Esfahani S, Loibl S, et al. Prospective validation of immunological infiltrate for prediction of response to neoadjuvant chemotherapy in HER2-negative breast cancer–a substudy of the neoadjuvant GeparQuinto trial. PloS One. 2013;8:e79775.
West NR, Milne K, Truong PT, Macpherson N, Nelson BH, Watson PH. Tumor-infiltrating lymphocytes predict response to anthracycline-based chemotherapy in estrogen receptor-negative breast cancer. Breast Cancer Res. 2011;13:R126.
Salgado R, Denkert C, Demaria S, et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an International TILs Working Group 2014. Ann Oncol. 2015;26:259–71.
Gamer M, Lemon J, Puspendra Singh IF. irr: Various coefficients of interrater reliability and agreement. R package version 0.84; 2012.
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33:159–74.
Hida AI, Ohi Y. Evaluation of tumor-infiltrating lymphocytes in breast cancer; proposal of a simpler method. Ann Oncol. 2015;26:2351.
Hornychova H, Melichar B, Tomsova M, Mergancova J, Urminska H, Ryska A. Tumor-infiltrating lymphocytes predict response to neoadjuvant chemotherapy in patients with breast carcinoma. Cancer Invest. 2008;26:1024–31.
Lee HJ, Seo JY, Ahn JH, Ahn SH, Gong G. Tumor-associated lymphocytes predict response to neoadjuvant chemotherapy in breast cancer patients. J Breast Cancer. 2013;16:32–9.
Nabholtz JM, Abrial C, Mouret-Reynier MA, et al. Multicentric neoadjuvant phase II study of panitumumab combined with an anthracycline/taxane-based chemotherapy in operable triple-negative breast cancer: identification of biologically defined signatures predicting treatment impact. Ann Oncol. 2014;25:1570–7.
Oda N, Shimazu K, Naoi Y, et al. Intratumoral regulatory T cells as an independent predictive factor for pathological complete response to neoadjuvant paclitaxel followed by 5-FU/epirubicin/cyclophosphamide in breast cancer patients. Breast Cancer Res Treat. 2012;136:107–16.
Seo AN, Lee HJ, Kim EJ, et al. Tumour-infiltrating CD8 + lymphocytes as an independent predictive factor for pathological complete response to primary systemic therapy in breast cancer. Br J Cancer. 2013;109:2705–13.
Acknowledgment
This work was supported in part by a Sister Institution Network Fund Grant (C.C. and E.A.M.) from the University of Texas MD Anderson Cancer Center. The statistical analyses were supported in part by a Cancer Center Support Grant awarded to the University of Texas MD Anderson Cancer Center (CA016672) and in part by the Hamill Foundation. Dr. Elizabeth Mittendorf is an R. Lee Clark Fellow of The University of Texas MD Anderson Cancer Center supported by the Jeanne F. Shelby Scholarship Fund. The authors acknowledge the administrative and financial support provided by Dr. Kelly K. Hunt, Hamill Foundation Professor of Surgery, Department of Breast Surgical Oncology at the University of Texas MD Anderson Cancer Center.
Conflict of interest
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Swisher, S.K., Wu, Y., Castaneda, C.A. et al. Interobserver Agreement Between Pathologists Assessing Tumor-Infiltrating Lymphocytes (TILs) in Breast Cancer Using Methodology Proposed by the International TILs Working Group. Ann Surg Oncol 23, 2242–2248 (2016). https://doi.org/10.1245/s10434-016-5173-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5173-8