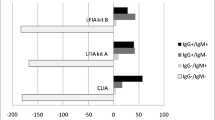
Abstract
Laboratory indicators can be used as the basis for the diagnosis, treatment, judgment of treatment efficacy, and prognosis of patients infected with SARS-CoV-2. Currently, the general laboratory indicators include nucleic acid tests, serum antibody tests, immunological tests, hematological tests, and bacteriological and mycological tests. The rational use of laboratory indicators is of great significance in guiding clinical diagnosis and treatment. Meanwhile, the laboratory staff should be careful to determine biosecurity measures to avoid infection based on the operational risk level of different test items.
The corresponding author of Sects. 3.1 and 3.2 is Hui Xing, Email: xhxytt080@sina.com;
The corresponding author of Sect. 3.3 is Lian Yang, Email: yanglian@hust.edu.cn;
The corresponding author of Sect. 3.4 is MingxingXie, Email: xiemx64@126.com;
The corresponding author of Sect. 3.5 is Yadan Wang, Email: yadan_wang@hust.edu.cn;
The corresponding author of Sect. 3.6 is Weimin Xiao, Email: xiaoweimin199968@163.com;
The corresponding author of Sect. 3.7 is Fanjun Cheng, Email: chengfanjun001@sina.com;
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Yang Y, Yang M, Shen C, et al. Evaluating the accuracy of different respiratory specimens in the laboratory diagnosis and monitoring the viral shedding of 2019-nCoV infections. med Rxiv preprint: https://doi.org/10.1101/2020.02.11.20021493
To KK, Tsang OT, Chik-Yan Yip C, et al. Consistent detection of 2019 novel coronavirus in saliva [published on February 12, 2020]. Clin Infect Dis. 2020. https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciaa149/5734265
Real-Time RT-PCR Panel for detection 2019-Novel coronavirus. (https://www.cdc.gov/coronavirus/2019-ncov/lab/index.html).
National Health Commission of the People’s Republic of China. COVID-19 diagnosis and treatment plan (Trial Version 7) (GWBYH [2020] No.184) [EB / OL]. [2020-03-03].
Liu J, Li S, Liu J, et. al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. medRxiv preprint: https://doi.org/10.1101/2020.02.16.20023671
Liu J, Liu Y, Pan X et al. Neutrophil-to-lymphocyte ratio predicts severe illness patients with 2019 novel coronavirus in the early stage. medRxiv preprint: https://doi.org/10.1101/2020.02.10.20021584
Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18(5):1233–4. https://doi.org/10.1111/jth.14768.
Shi H, et al. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis. 2020;20(4):425–34.
World Health Organization. Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected. Published on March 13, 2020. https://apps.who.int/iris/rest/bitstreams/1272156/retrieve.
Pan F, et al. Time course of lung changes on chest CT during recovery from 2019 novel coronavirus (COVID-19) pneumonia. Radiology. 2020;295(3):200370.
Xu Z, Shi L, Wang Y, et al. Pathological findings of NCP associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–2. https://doi.org/10.1016/s2213-2600(20)30076-x.
Rouby JJ, Arbelot C, Gao Y, et al. Training for lung ultrasound score measurement in critically ill patients. Am J Respir Crit Care Med. 2018;198(3):498–01.
Lichtenstein DA, Meziere GA. Relevance of lung ultrasound in the diagnosis of acute respiratory failure: the BLUE protocol. Chest. 2008;134:117–25.
Wang G, Ji X, Xu Y, et al. Lung ultrasound: a promising tool to monitor ventilator-associated pneumonia in critically ill patients. Crit Care. 2016;20(1):320.
Zhang Q, Liu D, Wang X, et al. Inferior vena cava diameter and variability on longitudinal plane measured through ultrasonography from different sites: a comparison study. Zhonghua Nei Ke Za Zhi. 2014;53(11):880–3.
Shrestha GS. Longing for better ultrasound-guided subclavian/axillary venous cannulation. Crit Care. 2018;22(1):148.
Wu Y, Luo L, Niu T, et al. Evaluation of the new Chinese disseminated intravascular coagulation scoring system in critically ill patients: a multicenter prospective study. Sci Rep. 2017;7(1):9057.
Wang YD, Zhang SP, Wei QZ, Zhao MM, Mei H, Zhang ZL, Hu Y. COVID-19 complicated with DIC: 2 cases report and literatures review. Zhonghua Xue Ye Xue Za Zhi. 2020;41(3):245–7. https://doi.org/10.3760/cma.j.issn.0253-2727.2020.0001.
Mei H, Hu Y. Characteristics, causes, diagnosis and treatment of coagulation dysfunction in patients with COVID-19. Zhonghua Xue Ye Xue Za Zhi. 2020;41(3):185–91. https://doi.org/10.3760/cma.j.issn.0253-2727.2020.0002.
Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–13.
Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–9. https://doi.org/10.1001/jama.2020.1585.
Guan W, Ni Z, Hu Y, et al. Clinical characteristics of 2019 novel coronavirus infection in China. medRxiv preprint. https://doi.org/10.1101/2020.02.06.20020974.
Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506.
Tang N, Li D, Wang X, et al. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18(4):844–7. https://doi.org/10.1111/jth.14768.
Dukic L, Kopcinovic LM, Dorotic A, Barsic I. Blood gas testing and related measurements: national recommendations on behalf of the Croatian Society of Medical Biochemistry and Laboratory Medicine. Biochem Med (Zagreb). 2016;26(3):318–36.
Hill S, Moore S. Arterial blood gas sampling: using a safety and pre-heparinised syringe. Br J Nurs. 2018;27(14):S20–6.
Han D, Li Z, Li R, Tan P, Zhang R, Li J. mNGS in clinical microbiology laboratories: on the road to maturity. Crit Rev Microbiol. 2019;45(5–6):668–85. https://doi.org/10.1080/1040841X.2019.1681933. Epub 2019 Nov 6
Chiu CY, Miller SA. Clinical metagenomics. Nat Rev Genet. 2019 Jun;20(6):341–55. https://doi.org/10.1038/s41576-019-0113-7.
Wilson MR, O’Donovan BD, Gelfand JM, Sample HA, et al. Chronic meningitis investigated via metagenomic next-generation sequencing. JAMA Neurol. 2018;75(8):947–55. https://doi.org/10.1001/jamaneurol.2018.0463.
Chen L, Liu W, Zhang Q, et al. RNA based mNGS approach identifies a novel human coronavirus from two individual pneumonia cases in 2019 Wuhan outbreak. Emerg Microbes Infect. 2020;9(1):313–9. https://doi.org/10.1080/22221751.2020.1725399. eCollection 2020
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 People's Medical Publishing House, PR of China
About this chapter
Cite this chapter
Xing, H. et al. (2020). COVID-19 Diagnosis. In: Cheng, F., Zhang, Y. (eds) The Clinical Diagnosis and Treatment for New Coronavirus Pneumonia. Springer, Singapore. https://doi.org/10.1007/978-981-15-5975-4_3
Download citation
DOI: https://doi.org/10.1007/978-981-15-5975-4_3
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-5974-7
Online ISBN: 978-981-15-5975-4
eBook Packages: MedicineMedicine (R0)