Summary
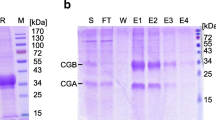
Many studies on the structure and function of human chorionic gonadotropin (hCG) have relied on purified hCG preparations obtained from pregnancy urine. In the present studies, in order to demonstrate possible differences between the hCG species present in serum and those released into urine, we examined serum and urinary samples derived from patients with trophoblastic tumors and of pregnancy origin by sodium dodecyl sulfate electrophoresis, isoelectric focusing, and two-dimensional electrophoresis including immunostaining with a specific hCG antibody and densitometry of the protein bands. This type of analysis was presently feasible only in patients presenting with extremely high serum levels of hCG and was therefore limited to seven patients with testicular cancers, one woman with a hydatidiform mole, and one pregnancy sample. We found marked differences in the isoelectric focusing pattern between urinary and serum hCG samples, with the urinary hCG consisting of more alkaline pl variants than that present in the serum of the same patients. Tumor hCG differed from pregnancy hCG in that it contained more acidic variants, and this was true for urinary and serum-derived materials. In some tumor and pregnancy samples an hCG immunoreactive material of lower molecular weight than hCG itself was found. In conclusion, the present studies, extending previous findings on the micro-heterogeneity of hCG, indicate that serum and urinary-derived hCG may differ in the composition of the isoform spectrum, as does tumor and pregancy hCG. Further, in some patients hCG immunoreactive molecules exist that differ markedly from hCG in size and charge. These observations suggest that, whenever possible, serum-derived hCG materials should be used to define the molecular structure of hCG and assess its biological activities.
Similar content being viewed by others
Abbreviations
- hCG:
-
human chorionic gonadotropin
- mAb:
-
monoclonal antibody
- SDS:
-
sodium dodecylsulfate
References
Amir SM, Kasagi K, Ingbar SH (1987) The role of subunit sialic acid in the thyrotropic and gonadotropic activities of human chorionic gonadotropin. Endocrinology 121:160–166
Bagshawe KD (1992) Choriocarcinoma. A model for tumour markers. Acta Oncol 31:99–106
Ballabio M, Poshyachinda M, Ekins RP (1991) Pregnancy-induced changes in thyroid function: role of human chorionic gonadotropin as putative regulator of maternal thyroid. J Clin Endocrinol Metab 73:824–831
Bartlett NL, Freiha FS, Torti FM (1991) Serum markers in germ cell neoplasms. Hematol Oncol Clin North Am 5:1245–1260
Blithe DL, Akar AH, Wehmann R, Birken S, Nisula B (1988) Purification of β-core fragment from pregnancy urine and demonstration that its carbohydrate moieties differ from those of native human chorionic gonadotropin-β. Endocrinology 122:173–180
Cassels Jr JW, Mann K, Blithe DL, Nisula BC, Wehmann RE (1989) Reduced metabolic clearance rate of acidic variants of human choriogonadotropin from patients with testicular cancer. Cancer 64:2313–2318
Cole LA (1987) The O-linked oligosacharide structures are striking different on pregnancy and choriocarcinoma hCG. J Clin Endocrinol Metab 65:811–813
Cole LA, Kardana A, Andrade-Gordon P, Gawinowicz MA, Morris JC, Bergert ER, O'Connor J, Birken S (1991) The heterogeneity of human chorionic gonadotropin (hCG). III. The occurrence and biological and immunological activities of nicked hCG. Endocrinology 129:1559–1567
Combarnous Y(1992) Molecular basis of the specificity of binding of glycoprotein hormones to their receptors. Endocr Rev 13:670–691
Goerg A, Postel W, Westermeier R, Gianazza E, Righetti PG (1980) Gel gradient electrophoresis, isoelectric focusing and two-dimensional techniques in horizontal, ultrathin polyacrylamide layers. J Biochem Biophys Methods 3:273–284
Good A, Ramos-Uribe M, Ryan RJ, Kempers RD (1977) Molecular forms of human chorionic gonadotropin in serum, urine and placental extracts. Fertil Steril 28:846–850
Graesslin D, Weise HC, Czygan PJ (1972) Isolation and partial characterization of several different chorionic gonadotropin (hCG) components. FEBS Lett 20:87–89
Hall van E, Vaitukaitis JL, Ross GT, Hickman JH, Ashwell GG (1971) Immunological and biological activities of hCG following progressive desialylation. Endocrinology 88:456–464
Hoermann R, Amir SM, Ingbar SH (1988) Evidence that partially desialylated variants of human chorionic gonadotropin (hCG) are the factors in crude hCG that inhibit the response to thyrotropin in human thyroid membranes. Endocrinology 123:1535–1543
Hoermann R, Spoettl G, Moncayo R, Mann (1990) Evidence for the presence of human chorionic gonadotropin (hCG) and free beta-subunit of hCG in the human pituitary. J Clin Endocrinol Metab 71:179–186
Hoermann R, Schumm-Draeger PM, Rehbach K, Mann K (1991) Asialoagalacto-human chorionic gonadotropin, a carbohydrate-modified variant of human chorionic gonadotropin, antagonizes the stimulatory actions of bovine thyroid-stimulating hormone on thyroid function and HLA-DR expression in human thyroid in vitro and in vivo. J Clin Invest 88:1947–1954
Hoermann R, Gerbes AL, Spoettl G, Jüngst D, Mann K (1992) Immunoreactive human chorionic gonadotropin and its free beta subunit in serum and ascites of patients with malignant tumors. Cancer Res 52:1520–1524
Hussa RO (1981) Human chorionic gonadotropin, a clinical marker: review of its biosynthesis. Ligand Review 3[Supp] 2]: 6–44
Kardana A, Elliot MM, Gawinowicz MA, Birken S, Cole LA (1991) The heterogeneity of human chorionic gonadotropin (hCG). I. Characterization of peptide heterogeneity in 13 individual preparations of hCG. Endocrinology 129:1541–1550
Kessler MJ, Reddy MS, Shah RH, Bahl OP (1979) Structures of N-glycosidic carbohydrate units of human chorionic gonadotropin. J Biol Chem 254:7901–7908
Kessler MJ, Mise T, Ghai RD, Bahl OP (1979) Structure and location of the O-glycosidic carbohydrate units of human chorionic gonadotropin. J Biol Chem 254:7909–7914
Kobata A (1988) Structures, function, and transformational changes of the sugar chains of glycohormones. J Cell Biochem 37:79–90
Kyhse-Andersen J (1984) A simple horizontal apparatus without buffer tank for electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose. J Biochem Biophys Meth 10:203–209
Manjunath P, Sairam MR (1982) Biochemical, biological and immunological properties of chemically deglycosylated human chorionic gonadotropin. J Biol Chem 257:7109–7115
Mann K, Schneider N, Hoermann R (1986) Thyrotropic activity of isoelectric variants of human chorionic gonadotropin from trophoblastic tumors. Endocrinology 118:1558–1566
Mann K, Saller B, Hoermann R (1993) Clinical use of hCG and hCGβ determinations. Scand J Clin Lab Invest 53 [Suppl 216]: 97–104
Mazuk MM, Keene JL, Boime I (1989) Site specifity of the chorionic gonadotropin N-linked oligosacharides in signal transdction. J Biol Chem 264:2409–2414
Merz WE, Hilgenfeldt U, Doerner M, Brossmer R (1974) Biological, immunological and physical investigations on human chorionic gonadotropin. Hoppe-Seyler's Z Physiol Chem 355:1035–1045
Morell AG, Gregoriadis G, Ito K, Scheinberg IH, Hickman J, Ashwell G (1971) The role of sialic acid in determining the survival of glycoproteins in the circulation. J Biol Chem 246:1461–1467
Moyle WR, Bahl OP, Maerz L (1975) Role of the carbohydrate of human chorionic gonadotropin in the mechanism of hormone action. J Biol Chem 250:9163–9169
Ozturk M (1991) Human chorionic gonadotropin, its free subunits and gestational trophoblastic disease. J Reprod Med 36:21–26
Pierce JG, Parsons TF (1981) Glycoprotein hormones, structure and function. Ann Rev Biochem 50:465–495
Rao Thotakura N, Weintraub B, Bahl OP (1990) The role of carbohydrate in human choriogonadotropin (hCG) action. Effects of N-linked carbohydrate chains from hCG and other glycoproteins on hormonal activity. Mol Cell Endocrinol 70:263–272
Rosa C, Amr S, Birken S, Wehmann R, Nisula B (1984) Effect of desialylation of human chorionic gonadotropin on its metabolic clearance rate in humans. J Clin Endocrinol Metab 59:1215–1219
Siddle K, Gard T, Thomas D, Cranage MP, Coombs RRA (1984) Red cell-labelled monoclonal antibodies for assay of human chorionic gonadotropin and luteinising hormone by reverse passive haemagglutination. J Immunol Methods 73:169–176
Taliadouros GS, Amr S, Louvet JP, Birken S, Canfield RE, Nisula BC (1982) Biological and immunological characterization of crude commercial human choriongonadotropin. J Clin Endocrinol Metab 54:1002–1009
Tsuruhara T, Dufau ML, Hickman J Catt KJ (1972) Biological properties of hCG after removal of terminal sialic acid and galactose residues. Endocrinology 91:296–301
Yazaki K, Yazaki C, Wakabayashi K, Igarashi M (1980) Isoelectric heterogeneity of human chorionic gonadotropin: presence of choriocarcinoma specific components. Am J Obstet Gynecol 138:189–194
Yoshimoto Y, Wolfsen AR, Odell WD (1979) Glycosylation, a variable in the production of hCG by cancers. Am J Med 67:415–420
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. Dr. G. Paumgartner on the occasion of his 60th birthday
Rights and permissions
About this article
Cite this article
Hoermann, R., Spoettl, G., Grossmann, M. et al. Molecular heterogeneity of human chorionic gonadotropin in serum and urine from patients with trophoblastic tumors. Clin Investig 71, 953–960 (1993). https://doi.org/10.1007/BF00185610
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00185610