Abstract
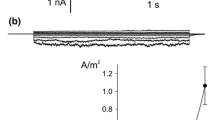
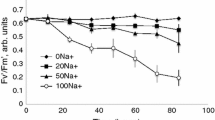
An anion channel that only allows outward current flow (anion influx) has been identified in protoplasts derived from wheat (Triticum aestivum L., Triticum turgidum L.) roots. The anion outward rectifier (anion OR) measured by patch-clamp of whole cells activated very quickly, usually reaching a steady-state level in less than 100 ms and was easily distinguished from the cation outward rectifier (cation OR) which activated more slowly during membrane depolarisation. The anion OR is permeable to NO −3 and Cl−, moderately permeable to I−, and relatively impermeable to H2PO4/− and ClO4/−. An anomalous mole-fraction effect between ClO4/ − and Cl− was observed on the outward current, indicating that the channel is a multi-ion pore. The anion OR is gated by both voltage and external anion concentration such that it activates near to the equilibrium potential for the permeant anion. It activated at more negative membrane potentials when NO −3 was substituted for Cl− in the external medium, indicating that the channel may function to allow NO −3 influx under luxuriant external NO −3 concentrations. For most experiments, K+ and Cl− were the main cation and anion in solution, and under these conditions it appeared likely that the anion OR functioned in membrane-potential regulation by facilitating a Cl− influx at membrane potentials more positive than the chloride reversal potential (ECl). If ECl was more negative than the K+ reversal potential (EK) then the anion OR dominated but both the anion and cation ORs occurred together when the membrane potential difference (Vm) was positive of both ECl and EK. The cation OR was inhibited by increasing external Cl− concentrations, but the anion OR was not affected by external K+ or Na+ concentration. The anion-transport inhibitors, zinc and phenylglyoxal were ineffective in blocking the anion OR. 4,4′-Di-isothiocyanostilbene-2, 2′-disulfonic acid (DIDS) irreversibly blocked about 34% of the current when applied extracellularly at a concentration of 25 μM, and about 69% at a concentration of 200 μM. However, DIDS (200 μM) also occasionally acted as an irreversible blocker of the cation OR. Perchlorate blocked irreversibly 75% of the current at an external concentration of 10 mM and did not block the cation OR. Whole-cell currents also indicated that the anion OR was insensitive to external pH (pH=5–7) and calcium concentration ([Ca2+]=0.1–10 mM). Increasing intracellular calcium concentration significantly increased the occurrence of the fast outward current in whole cells (P < 0.005, X2 test). With approximately 10 nM calcium inside the cell the anion outward current was observed in 64% (n = 45) of cells and with 50 nM calcium inside the cell the anion current was observed in 88% (n = 69) of cells. Single-anion OR channels observed in outside-out patches had a conductance in 300 mM KCl (external) of about 4 pS. When voltage pulses were applied to outside-out patches the average currents were similar to those observed in whole cells. The significance of the anion OR as a likely route for Cl uptake in high salinities is discussed.
Similar content being viewed by others
Abbreviations
- Bath:
-
solution bathing the extracellular face of the membrane
- DIDS:
-
(4,4′-diisothiocyanostilbene-2,2′-disulfonic acid)
- Ex :
-
reversal potential for ion x
- OR:
-
outward rectifier
- Pip :
-
solution inside the pipette
- TEACl:
-
(tetraethyl-ammonium chloride)
- Vm :
-
membrane potential difference
References
Binzel, M.L., Hess, D., Bressan, R.A., Hasegawa, P.M. (1988) Intracellular compartmentation of ions in salt adapted tobacco cells. Plant Physiol. 86, 607–614
Blatt, M.R. (1988) Potassium-dependent, bipolar gating of K+ channels in guard cells. J. Membr. Biol. 102, 235–246
Blatt, M.R. (1992) K+ channels of stomatal guard cells. J. Gen. Physiol. 99, 615–644
Bretag, A.H. (1987) Muscle chloride channels. Physiol. Rev. 67, 618–724
Cakirlar, H., Bowling, D.J.F. (1981) The effect of salinity of the membrane potential of sunflower roots. J. Exp. Bot. 32, 479–485
Cerana, R., Colombo, R. (1992) K+ and Cl− conductance of Arabidopsis thaliana plasma membrane at depolarised voltages. Bot. Acta 105, 273–277
Cheeseman, J.M., Hanson, J.B. (1979) Mathematical analysis of the dependence of cell potential on external potassium in corn roots. Plant Physiol. 63, 1–4
Corey, D.P., Stevens, C.F. (1983) Science and technology of patchrecording electrodes. In: Single-channel recording, pp. 53–68, Sakmann, B., Neher, E., eds. Plenum Press, New York London
Cosgrove, D.J., Hedrich, R. (1991) Stretch-activated chloride, potassium, and calcium channels coexisting in plasma membranes of guard cells of Vicia faba L. Planta 186, 143–153
Coyaud, L., Kurkdjian, A., Kado, R., Hedrich, R. (1987) Ion channels and ATP-driven transport across the tonoplast of sugarbeet vacuoles. Biochim. Biophys. Acta. 902, 263–268
Cram, W.J. (1973) Chloride fluxes in cells of the isolated root cortex of Zea mays. Aust. J. Biol. Sci. 26, 757–79
Dhugga, K.S., Waines, J.G., Leonard, R.T. (1988) Nitrate absorption by corn roots. Plant Physiol. 86, 759–763
Fairley, K., Laver, D., Walker, N.A. (1991) Whole-cell and single channel currents across the plasmalemma of corn shoot suspension cells. J. Membr. Biol. 121, 11–22
Falke, L.C., Edwards, K.L., Pickard, B.G., Misler, S. (1988) A stretch-activated anion channel in tobacco protoplasts. FEBS Lett. 237, 141–144
Findlay, G.P., Coleman, H.A. (1983) Potassium channels in the membrane of Hydrodictyon africanum. J. Membr. Biol. 75, 241–251
Gradmann, D., Mayer, W.-E. (1977) Membrane potentials and ion permeabilities in flexor cells of the laminar pulvini of Phaseolus coccineus L. Planta 137, 19–24
Hagiwara, S., Takahashi, K. (1974) Mechanism of anion permeation through the muscle fibre membrane of an elasmobranch fish, Taeniura lymma. J. Physiol. London 238, 109–127
Hamill, O.P., Marty, A., Neher, E., Sakmann, B., Sigworth, F.J. (1981) Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pfluegers Arch. 391, 85–100
Harris, E.J. (1958) Anion interaction in frog muscle. J. Physiol London 141, 351–365
Hedrich, R., Kurkdjian, A. (1988) Characterisation of an anion-permeable channel from sugar beet vacuoles: effect of inhibitors. EMBO J. 7, 3661–3666
Hedrich, R., Neher, E. (1987) Cytoplasmic calcium regulates voltage-dependent ion channels in plant vacuoles. Nature 329, 833–836
Hedrich, R. Schroeder, J. (1989) The physiology of ion channels and electrogenic pumps in higher plants. Annu. Rev. Plant Physiol. 40, 539–69
Hedrich, R., Busch, H., Raschke, K. (1990) Ca2+ and nucleotide dependent regulation of voltage dependent anion channels in the plasma membrane of guard cells. EMBO J. 9, 3889–3892
Hille, B., Schwarz, W. (1978) Potassium channels as multi-ion singlefile pores. J. Gen Physiol. 72, 409–442
Iijima, T., Hagiwara, S. (1987) Voltage-dependent K channels in protoplasts of trap-lobe cells of Dionaea muscipula. J. Membr. Biol. 100, 73–81
Keifer, D.W., Franceschi, V.R., Lucas, W.J. (1982) Plasmalemma chloride transport in Chara corallina. Inhibition by 4,4′-diisothiocyano-2–2′disulfonic acid stilbene. Plant Physiol. 70, 1327–1334
Keller, B.U., Hedrich, R., Raschke (1989) Voltage-dependent anion channels in the plasma membrane of guard cells. Nature 341, 450–453
Ketchum, K.A., Poole, R.J. (1991) Cytosolic calcium regulates a potassium current in corn (Zea mays) protoplasts. J. Membr. Biol. 119, 277–288
Kingsbury, R.W, Epstein, E. (1986) Salt sensitivity in wheat. Plant Physiol. 80, 651–654
Kochian, L.V., Xin-Zhi, J., Lucas, W.J. (1985) Potassium transport in corn roots: IV. Characterisation of the linear component. Plant Physiol. 79, 771–776
Kourie, J.I., Findlay, G.P. (1990) Ionic currents across the plasmalella of Chara inflata cells. J. Exp. Bot. 41, 151–163
Lew, R.R. (1991) Substrate regulation of single potassium and chloride channels in Arabidopsis plasma membrane. Plant Physiol. 95, 642–647
Lin, W. (1980) Corn root protoplasts: isolation and general characterisation of ion transport. Plant Physiol. 66, 550–554
Lynch, J., Polito, V.S., Lauchli, A. (1989) Salinity stress increases cytoplasmic Ca activity in maize root protoplasts. Plant Physiol. 90, 1271–1274
Marten, L, Busch, H., Raschke, K., Hedrich, R. (1993) Modulation and block of the plasma membrane anion channel of guard cells by stilbene derivatives. Eur. Biophys. J. 21, 403–408
Marten, I., Zeiilinger, C., Redhead, C., Landry, D.W., Al-Awqati, Q., Hedrich, R. (1992) Identification and modulation of a voltagedependent anion channel in the plasma membrane of guard cells by high-affinity ligands. EMBO Journal 11, 3569–3575
Pantoja, O., Dainty, J., Blumwald, E. (1992) Cytoplasmic chloride regulates cation channels in the vacuolar membrane of plant cells. J. Membr. Biol. 125, 219–229
Schachtman, D.P., Tyerman, S.D., Terry, B.R. (1991) The K+/Na+ selectivity of a cation channel in the plasma membrane of root cells does not differ in salt-tolerant and salt-sensitive wheat species. Plant Physiol. 97, 598–605
Schauf, C.L., Wilson, K.J. (1987) Properties of single K+ and Cl− channels in Asclepias tuberosa protoplasts. Plant Physiol. 85, 413–418
Schroeder, J.I. (1989) Quantitative analysis of outward rectifying K+ channel currents in guard cell protoplasts from Vicia faba. J. Membr. Biol. 107, 229–235
Schroeder, J.I., Hagiwara, S. (1989) Cytosolic calcium regulates ion channels in the plasma membrane of Vicia faba guard cells. Nature 338, 427–430
Schroeder, J.I., Keller, B.U. (1992) Two types of anion channel currents in guard cells with distinct voltage regulation. Proc. Natl. Acad. Sci. 89, 5025–5029
Schroeder, J.I., Raschke, K., Neher, E. (1987) Voltage dependence of K+ channels in guard cell protoplasts. Proc. Natl. Acad. Sci. 84, 4108–4112
Siddiqi, M.Y., Glass, A.D.M., Ruth, T.J., Rufty, T.W. Jr. (1990) Studies of uptake of nitrate in barley I. Kinetics of 13NO −3 Influx. Plant Physiol. 93, 1426–1432
Terry, B.R., Tyerman, S.D., Findlay, G.P. (1991) Ion channels in the plasma membrane of Amaranthus protoplasts: one cation and one anion channel dominate the conductance. J. Membr. Biol. 121, 223–236
Terry, B.R., Findlay, G.P., Tyerman, S.D. (1992a) Direct effects of Ca2+-channel blockers on plasma membrane cation channels of Amaranthus tricolor protoplasts. J. Exp. Bot. 43, 1457–1473
Terry, B.R., Tyerman, S.D., Findlay, G.P. (1992b) Ion channels in the plasma membrane of plant cells. In: Swelling mechanics: From clays to whole organisms (NATO ASI Ser.), pp. 225–236, T.K. Karalis, ed. Elsevier, Amsterdam
Tester, M. (1988) Potassium channels in the plasmalemma of Chara corallina are multi-ion pores: voltage-dependent blockade by Cs+ and anomalous permeabilities. J. Membr. Biol. 105, 87–94
Tester, M. (1990) Plant ion channels: whole-cell and single channels studies. New Phytol. 114, 305–40
Tyerman, S.D. (1992) Anion channels in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 43, 351–373
Tyerman, S.D., Findlay, G.P., Patterson, G.J. (1986) Inward membrane current in Chara inflata: I. A voltage- and time-dependent Cl− component. J. Membr. Biol. 89, 139–152
Tyerman, S.D., Schachtman, D.P. (1992) The role of ion channels in plant nutrition and prospects for their genetic manipulation. Plant Soil 146, 137–144
Tyerman, S.D., Oats, P., Gibbs, J., Dracup, M., Greenway, H. (1989) Turgor-volume regulation and cellular water relations in Nicotiana tabacum roots grown in high salinities. Aust. J. Plant Physiol. 16, 517–31
Walker, N.A. (1981) The transport systems of charophyte and chlorophyte giant algae and their integration into modes of behaviour. In: Plant membrane transport: current conceptual issues, pp. 287–300, R.M. Spanswick, W.J. Lucas, J. Dainty eds. Elsevier, Amsterdam
Author information
Authors and Affiliations
Additional information
We thank the Australian Research Council for financial support, G.P. Findlay and A. Garrill for helpful discussions, and K. Morris and D. Mackenzie for expert technical assistance. M.S. was supported by an Australian Postgraduate Research Award.
Rights and permissions
About this article
Cite this article
Skerrett, M., Tyerman, S.D. A channel that allows inwardly directed fluxes of anions in protoplasts derived from wheat roots. Planta 192, 295–305 (1994). https://doi.org/10.1007/BF00198563
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00198563