Abstract
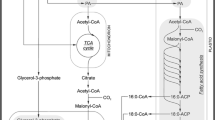
Embryos derived in vitro from isolated microspores of Brassica napus L. were compared with their zygotic counterparts. Parameters investigated included storage-protein accumulation and gene expression, fattyacid composition, storage-lipid biosynthesis, and the appearance of oil-body proteins. The microspore embryos accumulate storage-protein and show increases in levels of their transcripts during the torpedo stage. These embryos were sensitive to abscisic acid (ABA) with respect to accumulation of storage-protein mRNA and oil-body proteins. Post-transcriptional regulation of cruciferin accumulation is indicated by a disparity between ABA-enhanced transcript accumulation and a less marked effect at the level of protein accumulation. To investigate storage-lipid profiles, two cultivars of Brassica napus, Reston and Topas, were used. The former accumulates major quantities of C20 (11.2%) and C22 (39.9%) fatty acids in its seeds, the latter predominantly C18 fatty acids. The higher-molecular-weight fatty acids (>C18) normally occur only in seeds and were used as biochemical markers for seed-specific metabolism in microspore embryos. Microspore embryos from Reston were found to accumulate C20 (10.6%) and C22 (31.2%) fatty acids after 35 d in culture at levels and proportions comparable to those found in seeds. Similarly, microspore embryos of Topas had a fatty-acid profile similar to that of mature Topas seed. Activities of enzymes involved in the accumulation of storage lipids (erucoyl-CoA synthetase [EC 6.2.1.3], erucoyl-CoA thioesterase [EC 3.1.2.2] and erucoyl-CoA acyltransferase [EC 2.3.1.15 or EC 2.3.1.20]) were detected in torpedostage microspore embryos. Their specific activities were higher than have been reported to date for analogous preparations from zygotic embryos of B. napus. The similarities in storage-lipid and protein composition of these embryos to their zygotic counterparts, along with their sensitivity to ABA, indicate that microspore embryos might be exploited to facilitate studies of biochemistry and gene regulation in oilseeds.
Similar content being viewed by others
Abbreviations
- ABA:
-
abscisic acid
- DW:
-
dry weight
- FW:
-
fresh weight
- kDA:
-
kilodalton
- TAG:
-
triacylglycerol
References
Andrews, J., Keegstra, K. (1983) Acyl-CoA synthetase is located in the outer membrane and acyl-CoA thioesterase in the inner membrane of pea chloroplast envelopes. Plant Physiol. 72, 735–740
Battey, J.F., Ohlrogge, J.B. (1989) A comparison of the metabolic fate of fatty acids of different chain lengths in developing oilseeds. Plant Physiol. 90, 835–840
Battey, J.F., Schmid, K.M., Ohlrogge, J.B. (1989) Genetic engineering for plant oils: potentials and limitations. Trends Biotechnol. 7, 122–126
Bloch, K., Vance, D. (1977) Control mechanisms in the synthesis of saturated fatty acids. Annu. Rev. Biochem. 46, 263–298
Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254
Choi, J.H., Liu, L.S., Borkird, C., Sung, Z.R. (1987) Cloning of genes developmentally regulated during plant embryogenesis. Proc. Natl. Acad. Sci. USA 84, 1906–1910
Corley, R.H.V., Barrett, J.H., Jones, L.H. (1977) Vegetative propagation of oil palm via tissue culture. In: International development and the oil palm. Proceedings of Malaysian International Agriculture Oil Conference 1976, pp. 1–7
Crouch, M.L. (1982) Non-zygotic embryos of Brassica napus L. Contain embryo-specific storage proteins. Planta 156, 520–524
Crouch, M.L., Sussex, I.M. (1981) Development and storage-protein synthesis in Brassica napus L. tembryos in vivo and in vitro. Planta 153, 64–74
Crouch, M.L., Tenbarge, K.M., Simon, A.E., Ferl, R. (1983) cDNA clones for Brassica napus seed storage proteins: evidence from nucleotide sequence analysis that both subunits of napin are cleaved from a precursor polypeptide. J. Mol. Appl. Genet. 2, 273–283
Crouch, M.L., Tenbarge, K., Simon, A., Finkelstein, R., Scofield, S., Solberg, L. (1985) Storage-protein mRNA levels can be regulated by abscisic acid in Brassica embryos. In: Molecular form and function of the plant genome, pp. 555–566, van Vloten-Doting, L., Groot, G., Hall, T.C., eds. Plenum Press, New York
Denhardt, D. (1966) A membrane filter technique for the selection of complementary DNA. Biochem. Biophys. Res. Commun. 23, 641–646
Fan, Z., Armstrong, K.C., Keller, W.A. (1988) Development of microspores in vivo and in vitro in Brassica napus L. Protoplasma 147, 191–199
Feinberg, A.P., Vogelstein, B. (1984) A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity. Anal. Biochem. 137, 266–267
Finkelstein, R.R., Crouch, M.L. (1984) Precociously germinating rapeseed embryos retain characteristics of embryogeny. Planta 162, 125–131
Finkelstein, R.R., Tenbarge, K.M., Shumway, J.E., Crouch, M.L. (1985) Role of ABA in maturation of rapeseed embryos. Plant Physiol. 78, 630–636
Fouroughi-Wehr, B., Mix, G., Gaul, H., Wilson, H.M. (1976) Plant production from cultured anthers of Hordeum vulgare. Z. Pflanzenzücht. 77, 198–204
Franz, G., Hatzopoulos, P., Jones, T.J., Krauss, M., Sung, Z.R. (1989) Molecular and genetic analysis of an embryonic gene, DC 8 from Daucus carota L. Mol. Gen. Genet. 218, 143–151
Hakman, I., Fowke, L.C. (1987) Somatic embryogenesis in Picea glauca (white spruce) and Picea mariana (black spruce). Can. J. Bot. 65, 656–659
Halperin, W. (1966) Alternative morphogenetic events in cell suspensions. Am. J. Bot. 53, 443–453
Hara, A., Radin, N.S. (1978) Lipid extraction of tissues with a low-toxicity solvent. Anal. Biochem. 90, 420–426
Hara, S., Falk, H., Kleinig, H. (1985) Starch and triacylglycerol metabolism related to somatic embryogenesis in Papaver orientale tissue cultures. Planta 164, 303–307
Harwood, J. (1989) Lipid metabolism in plants. Crit. Rev. Plant Sci. 8, 1–44
Hills, M.J., Murphy, D.J., Beevers, H. (1989) Inhibition of neutral lipase from castor bean lipid bodies by coenzyme A (CoA) and oleoyl-CoA. Plant Physiol. 89, 1006–1010
Joyard, J., Stumpf, P.K. (1981) Synthesis of longchain acyl-CoA in the chloroplast envelope membranes. Plant Physiol. 67, 250–256
Kao, K.N., Michayluk, M.R. (1981) Embryoid formation in alfalfa cell suspensions from different plants. In Vitro 17, 645–648
Lichter, R. (1982) Induction of haploid plants from isolated pollen of Brassica napus. Z. Pflanzenzücht. 105, 427–434
Lu, C.-Y., Thorpe, T.A. (1987) Somatic embryogenesis and plantlet regeneration in cultured immature embryos of Picea glauca. J. Plant Physiol. 128, 297–302
Maheshwari, P., Sachar, R.C. (1963) Polyembryony. In: Recent advances in the embryology of angiosperms, pp. 265–296, Maheshwari, P., ed. International Society of Plant Morphologists, University of Delhi, Delhi, India
Murphy, D.J., Mukherjee, K.D. (1987) Acyltransferases in subcellular fractions of developing seeds of rape (Brassica napus L.). Lipids 22, 293–298
Murphy, D.J., Mukherjee, K.D., Latzko, E. (1983) Lipid metabolism in microsomal fraction from photosynthetic tissue: Effects of catalase and hydrogen peroxide on oleate desaturation. Biochem. J. 213, 249–252
Murphy, D.J., Cummins, I., Kang, A.S. (1989) Synthesis of the major oil-body membrane proteins in developing rapeseed (Brassica napus) embryos. Biochem. J. 258, 285–293
Nitsch, J.-P. (1969) Experimental androgenesis in Nicotiana. Phytomorphology 19, 389–404
Rabechault, H., Ahee, J., Guenin, G. (1970) Colonies cellulaires et formes embryoides obtenu in vitro a partir de cultures d'embryon de palmier de huile (Elaissi guineensis). C.R. Acad. Sci. Paris 270D, 233–237
Reinert, J., (1958) Untersuchungen über die Morphogenese an Gewebekulturen. Ber. Dtsch. Bot. Ges. 71, 15–19
Roughan, P.G., Slack, C.R. (1982) Cellular organization of glycerolipid metabolism. Annu. Rev. Plant Physiol. 33, 97–132
Shoemaker, R.C., Christofferson, S.E., Galbraith, D.W. (1987) Storage-protein accumulation patterns in somatic embryos of cotton (Gossypium hirsutum). Plant Cell Rep. 6, 12–15
Siebel, J., Pauls, K.P. (1989) A comparison of anther and microspore culture as a breeding tool in Brassica napus. Theor. Appl. Genet. 78, 473–479
Simon, A.E., Tenbarge, K.M., Scofield, S.R., Finkelstein, R.R., Crouch, M.L. (1985) Nucleotide sequence of a cDNA clone of Brassica napus 12S storage-protein shows homology with legumin from Pisum sativum. Plant Mol Biol. 5, 191–201
Smith, J.A., Krauss, M.R., Borkid, C., Sung, Z.R. (1988) A nuclear protein associated with cell division in plants. Planta 174, 462–472
Steward, F.C., Mapes, M.O., Kent, A.E., Holsten, R.D. (1964) Growth and development of cultured plant cells. Science 143, 20–27
Stymne, S., Stobart, A.K. (1987) Triacylglycerol biosynthesis. In: The biochemistry of plants, A comprehensive treatise, vol. 9, pp. 175–214., Stumpf, P.K., ed. Academic Press, New York
Sun, C., Cao, Y.-Z., Huang, A.H.C. (1988) Acyl coenzyme A preference of the glycerol phosphate pathway in the microsomes from the maturing seeds of palm, maize, and rapeseed. Plant Physiol. 88, 56–60
Sunderland, N., Wicks, F.M. (1969) Cultivation of haploid plants from tobacco pollen. Nature 224, 1227–1229
Taylor, D., Weber, N., Hogge, L.R., Underbill, E.W. (1990) A simple enzymatic method for the preparation of radiolabeled erucoyl-CoA and other long chain fatty acyl CoAs and their characterization by mass spectrometry. Anal. Biochem. 184, 311–316
Turnham, E., Northcote, D.H. (1984) Incorporation of [1-14C]acetate into lipids during embryogenesis in oil palm tissue cultures. Phytochemistry 23, 35–39
Vance, V.B., Huang, A.H.C. (1987) The major protein from lipid bodies of maize. J. Biol. Chem. 262, 11275–11279
Vance, V.B., Huang, A.H.C. (1988) Expression of lipid body protein during maize seed development. J. Biol. Chem. 263, 1476–1481
Verwoerd, T.C., Dekker, B.M.M., Hoekema, A. (1989) A small scale procedure for the rapid isolation of plant RNAs. Nucleic Acids Res. 17, 2362
Weber, N., Mangold, H.K.. (1989) Lipids. In: Cell culture and somatic cell genetics of plants, vol. 5, pp. 509–533, Vasil, I.K., Constabel, F., eds. Academic Press, New York
Yang, H.Y., Zhou, C. (1982) In vitro induction of haploid plants from unpollinated ovaries and ovules. Theor. Appl. Genet. 63, 87–104
Author information
Authors and Affiliations
Additional information
National Research Council Canada No. 31224
The authors would like to thank Marjorie Norris (Calgary), Nancy Long and John Grainger (Ottawa) for excellent technical assiatance with the preparation and amintenance of cultures. Helpful discussions with Drs. Randall Weselake, Sam MacKenzie, Lois Edwards and Ed Yeung are greatly appreciated. This work was supported in part by the National Research Council of Canada contribution program and by an operating grant (No. A3490) from Natural Sciences and Engineering Research Council of Canada to M.M.M.
Rights and permissions
About this article
Cite this article
Taylor, D.C., Weber, N., Underhill, E.W. et al. Storage-protein regulation and lipid accumulation in microspore embryos of Brassica napus L.. Planta 181, 18–26 (1990). https://doi.org/10.1007/BF00202320
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00202320