Abstract
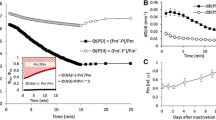
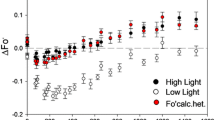
To determine the dependence of in vivo photosystem (PS) II function on photon exposure and to assign the relative importance of some photoprotective strategies of PSII against excess light, the maximal photochemical efficiency of PSII (Fv/Fm) and the content of functional PSII complexes (measured by repetitive flash yield of oxygen evolution) were determined in leaves of pea (Pisum satlvum L.) grown in moderate light. The modulation of PSII functionality in vivo was induced by varying either the duration (from 0 to 3 h) of light treatment (fixed at 1200 or 1800 μmol photons · m-2 · s-1) or irradiance (from 0 to 3000 μmol photons · m-2 · s-1) at a fixed duration (1 h) after infiltration of leaves with water (control), lincomycin (an inhibitor of chloroplast-encoded protein synthesis), nigericin (an uncoupler), or dithiothreitol (an inhibitor of the xanthophyll cycle) through the cut petioles of leaves of 22 to 24-day-old plants. We observed a reciprocity of irradiance and duration of illumination for PSII function, demonstrating that inactivation of functional PSII depends on the total number of photons absorbed, not on the rate of photon absorption. The Fv/Fm ratios from photoinhibitory light-treated leaves, with or without inhibitors, declined pseudo-linearly with photon exposure. The number of functional PSII complexes declined multiphasically with increasing photon exposure, in the following decreasing order of inhibitor effect: lincomycin > nigericin > DTT, indicating the central role of D1 protein turnover. While functional PSII and Fv/Fm ratio showed a linear relationship under high photon exposure conditions, in inhibitor-treated leaves the Fv/Fm ratio failed to reveal the loss of up to 25% of the total functional PSII under low photon exposure. The loss of this 25% of less-stable functional PSII was accompanied by a decrease of excitation-energy trapping capacity at the reaction centre of PSII (revealed by the fluorescence parameter, 1/Fo-1/Fm, where Fo and Fm stand for chlorophyll fluorescence when PSII reaction centres are open and closed, respectively), but not by a loss of excitation energy at the antenna (revealed by the fluorescence parameter, 1/Fm). We conclude that (i) PSII is an intrinsic photon counter under photoinhibitory conditions, (ii) PSII functionality is mainly regulated by D1 protein turnover, and to a lesser extent, by events mediated via the transthylakoid pH gradient, and (iii) peas exhibit PSII heterogeneity in terms of functional stability during photon exposure.
Similar content being viewed by others
Abbreviations
- D1 protein:
-
psbA gene product
- DTT:
-
dithiothreitol
- Fo:
-
chlorophyll fluorescence corresponding to open PSII reaction centres
- Fv, Fm:
-
variable and maximum fluorescence after dark incubation, respectively
- Fs, Fm′:
-
steady-state and maximum fluorescence during illumination, respectively
- P680:
-
reactioncentre chlorophyll and primary electron donor of PSII
- PS:
-
photosystem
References
Anderson, J.M. (1986) Photoregulation of the composition, function, and structure of thylakoid membranes. Annu. Rev. Plant Physiol. 37, 93–136
Anderson, J.M., Osmond, C.B. (1987) Shade-sun responses: compromises between acclimation and photoinhibition. In: Topics in photosynthesis. Photoinhibition, vol. 9, pp. 1–38, Kyle, D.M., Osmond, C.B., Arntzen, C.J., eds. Elsevier, Amsterdam
Aro, E.-M., McCaffery, S., Anderson, J.M. (1993) Photoinhibition and D1 protein degradation in peas acclimated to different growth irradiances. Plant Physiol. 103, 835–843
Aro, E.-M., McCaffery, S., Anderson, J.M. (1994) Recovery from photoinhibition in pea (Pisum sativum L.) acclimated to varying growth irradiances. Plant Physiol. 104, 1033–1041
Bell, C.J., Rose, D.A. (1981) Light measurement and the terminology of flow. Plant Cell Environ. 4, 89–96
Barber, J. (1995) Molecular basis of the vulnerability of photosystem II to damage by light. Aust. J. Plant Physiol. 22, 201–208
Bilger, W., Björkman, O. (1994) Relationships among violaxanthin deepoxidation, thylakoid membrane conformation, and nonphotochemical chlorophyll fluorescence quenching in leaves of cotton (Gossypium hirsutum L.). Planta 193, 238–246
Bilger, W., Björkman, O., Thayer, S.S. (1989) Light-induced spectral absorbance changes in relation to photosynthesis and the expoxidation state of xanthophyll cycle components in cotton leaves. Plant Physiol. 91, 542–551
Björkman, O. (1987) Low-temperature chlorophyll flluorescence in leaves and its relationship to photon yield of photosynthesis in photoinhibition, pp. 123–144, Kyle, D.J., Osmond, C.B., Arntzen, C.J., eds. Elsevier, Amsterdam
Björkman, O., Demmig, B. (1987) Photon yield of O2 evolution and chlorophyll fluorescence characteristics at 77 K among vascular plants of diverse origins. Planta 170, 489–504
Björkman, O., Boardman, N.K., Anderson, J.M., Thorne, S.W., Goodschild, D.J., Pyliotis, N.A. (1972) Effect of light intensity during growth of Atriplex patula on the capacity of photosynthetic reactions, chloroplast composition and structure. Carnegie Inst. Washington Yearb. 71, 115–135
Boekema, E. J., Hankamer, B., Bald, D., Kruip, J. Nield J., Boonstra, A.F., Barber, J., Rogner, M. (1995) Supramolecular structure of the photosystem II complex from green plants and cyanobacteria. Proc. Natl. Acad. Sci. USA 92, 175–179
Chow, W.S. (1994) Photoprotection and photoinhibitory damage. Adv. Mol. Cell Biol. 10, 151–196
Chow, W.S., Hope, A.B, Anderson, J.M. (1991) Further studies on quantifying photosystem II in vivo by flash-induced oxygen yield from leaf discs. Aust. J. Plant Physiol. 18, 397–410
Demmig, B., Björkman, O. (1987) Comparison of the effect of excessive light on chlorophyll fluorescence (77 K) and photon yield of O2 evolution in leaves of higher plants. Planta 171, 171–184
Demmig, B., Winter, K., Krüger, A., Czygan, F.-C. (1987) Photoinhibition and zeaxanthin formation in intact leaves. A possible role of the xanthophyll cycle in the dissipation of excess light. Plant Physiol. 84, 218–224
Demmig-Adams, B., Adams III, W.W. (1992) Photoprotection and other responses of plants to high light stress. Annu. Rev. Plant Physiol. Plant Mol. Biol. 43, 599–626
Eckert, H.-J., Geiken, B., Bernading, J., Napiwotzki, A., Eichler, H.-J., Renger, G. (1991) Two sites of photoinhibition of the electron transfer in oxygen evolving and Tris-treated PSII membrane fragments from spinach. Photosynth. Res. 27, 97–108
Franklin, L.A., Lavavasseur, G., Osmond, C.B., Henley, W.J., Ramus, J. (1992) Two components of onset and recovery during photoinhibition of Ulva rotundata. Planta 186, 399–408
Genty, B., Briantais, J.-M-, Baker, N.R. (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta 990, 87–92
Gilmore, A.M., Yamamoto, H.Y. (1992) Dark induction of zeaxanthin-dependent nonphotochemical fluorescence quenching mediated by ATP. Proc. Natl. Acad. Sci. USA 89, 1899–1903
Havaux, M., Strasser, R.J., Greppin, H. (1991) A theoretical and experimental analysis of the qP and qN coefficients of chlorophyll fluorescence quenching and their relation to photochemical and nonphotochemical events. Photosynth. Res. 27, 41–55
Hormann, H., Neubauer, C., Schreiber, U. (1994) On the relationship between chlorophyll fluorescence quenching and the quantum yield of electron transport in isolated thylakoids. Photosynth. Res. 40, 93–106
Jones, L.W., Kok, B. (1966) Photoinhibition of chloroplast reactions. Plant Physiol. 41, 1037–1043
Krause, G.H., Weiss, E. (1991) Chlorophyll fluorescence and photosynthesis: the basics. Annu. Rev. Plant Physiol. Plant Mol. Biol. 42, 313–349
Mattoo, A.K., Hoffman-Falk, H., Marer, J.B., Edelman, M. (1984) Regulation of protein metabolism: coupling of photosynthetic electron transport to in vivo degradation of the rapidly metabolized 32-kilodalton protein of the chloroplast membranes. Proc. Natl. Acad. Sci. USA 81, 1380–1384
Melis, A. (1991) Dynamics of photosynthetic membrane composition and function. Biochim. Biophys. Acta 1058, 87–106
Mullet, J.E. (1988) Chloroplast development and gene expression. Annu. Rev. Plant Physiol. Plant Mol. Biol. 39, 475–502
Ögren, E. (1991) Prediction of photosynthesis in willow leaves under field conditions. Planta 175, 229–236
Öquist, G., Chow, W.S. (1992) On the relationship between the quantum yield of photosystem II electron transport, as determined by chlorophyll fluorescence, and the quantum yield of CO2-dependent O2 evolution. Photosynth. Res. 33, 51–62
Öquist, G., Chow, W.S., Anderson J.M. (1992) Photoinhibition of photosynthesis represents a mechanism for the long-term regulation of photosystem II. Planta 186, 450–460
Osmond, C.B. (1994) What is photoinhibition: some insights from comparisons of shade and sun plants, pp. 1–24, Baker, N.R., Bowyer, J.R., eds. Bios Scientific Publishers, Oxford
Porra, R.J., Thompson, W.A., Kriedemann, P.E. (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophyll a and b with four different solvents: verification of the concentration of chlorophyll by atomic absorption spectroscopy. Biochim. Biophys. Acta 975, 384–394
Powles, S.B. (1984) Photoinhibition of photosynthesis induced by visible light. Annu. Rev. Plant Physiol. 35, 15–44
Russell, A.W., Critchley, C., Robinson, S. A., Franklin, L.A., Seaton, G.G.R., Chow, W.S., Anderson, J.M. Osmond, C.B. (1995) Photosystem II regulation and dynamics of the chloroplast D1 protein in Arabidopsis leaves during photosynthesis and photoinhibition. Plant Physiol. 107, 943–952
Santini, C., Tidu, V., Tognon, G., Magaldi, A.G., Bassi, R. (1994) Three-dimensional structure of the higher-plant photosystem II reaction centre and evidence for its dimeric organization in vivo. Eur. J. Biochem. 221, 307–315
Schreiber, U., Hormann, H., Neubauer, C., Klughammer, C. (1995) Assessment of PSII photochemical quantum yield by chlorophyll fluorescence quenching analysis. Aust. J. Plant Physiol. 22, 209–220
Seaton, G.G.R., Walker, D. A. (1990) Chlorophyll fluorescence as a measure of photosynthetic carbon assimilation. Proc. R. Soc. B. 242, 29–35
Seibert, M. (1995) Reflections on the nature and function of the photosystem II reaction centre. Aust. J. Plant Physiol. 22, 161–166
Sonoike, K., Terashima, I. (1994) Mechanism of photosystem-I photoinhibition in leaves of Cucumis sativus L. Planta 194, 287–293
Sundby, C., McCaffery, S., Anderson, J.M. (1993) Turnover of the photosystem II D1 protein in higher plants under photoinhibitory and nonphotoinhibitory irradiance. J. Biol. Chem. 268, 25476–25482
Trissl, H.-W., Lavergne, J. (1995) Fluorescence induction from photosystem II: analytical equations for the yields of photochemistry and fluorescence derived from analysis of a model including exciton-radical pair equilibrium and restricted energy transfer between photosynthetic units. Aust. J. Plant Physiol. 22, 183–193
Tyystjärvi, E., Ali-Yrkkö, K., Kettunen, R., Aro, E.-M. (1992) Slow degradation of the D1 protein is related to the susceptibility of low-light-grown pumpkin plants to photoinhibition. Plant Physiol. 100, 1310–1317
Walters, R.G., Horton, P. (1993) Theoretical assessment of alternative mechanisms for non-photochemical quenching of PSII fluorescence in barley leaves. Photosynth. Res. 36, 119–139
Author information
Authors and Affiliations
Additional information
Financial support of this work by Department of Employment, Education and Training/Australian Research Council International Research Fellowships Program (Korea) is gratefully acknowledged.
Rights and permissions
About this article
Cite this article
Park, YI., Chow, W.S. & Anderson, J.M. Light inactivation of functional photosystem II in leaves of peas grown in moderate light depends on photon exposure. Planta 196, 401–411 (1995). https://doi.org/10.1007/BF00203636
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00203636