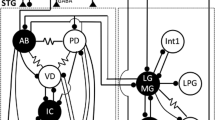
Summary
-
1.
Muscles of the posterior cardiac plate (pcp) and pyloric regions in the stomach of Squilla are innervated by motoneurons located in the stomatogastric ganglion (STG). The pattern of innervation of various muscles in these regions was determined using electrophysiological methods.
-
2.
The dilator muscles are singly or doubly innervated by the pyloric dilator neurons (PDs). The constrictor muscles are singly or doubly innervated by the pcp neuron (PCP) or the pyloric neurons (PYs). These muscles are sequentially activated by pcp-pyloric motor outputs produced by the PCP, PY, and PD. All muscles can generate an all-or-nothing spike.
-
3.
The constrictor muscles generate spikes followed by depolarizing afterpotentials which lead to a sustained depolarization with repetitive spikes. The PYs can entrain rhythmic spike discharges of these muscles.
-
4.
The spike of muscles remains unchanged by bath application of tetrodotoxin (10-7 M) to suppress neuronal impulse activities, but it is blocked by Mn2+ (10 mM).
-
5.
The constrictor muscle isolated from the STG displays an endogenous property of spontaneous membrane oscillation that produces a train of spikes. Brief depolarizing or hyperpolarizing stimuli can trigger or terminate an oscillatory potential, respectively, and reset the subsequent rhythm.
-
6.
The possible functions of myogenicity under the control of discharges of motoneurons in the pyloric constrictor neuromuscular system are discussed.
Similar content being viewed by others
Abbreviations
- CPG :
-
central pattern generator
- OG :
-
oesophageal ganglion
- EJP :
-
excitatory junction potential
- lvn :
-
lateral ventricular nerve
- pcp :
-
posterior cardiac plate
- PCP :
-
pcp neuron
- PD :
-
pyloric dilator neuron
- PY :
-
pyloric neuron
- STG :
-
stomatogastric ganglion
References
Atwood HL, Govind CK, Jahromi SS (1977) Excitatory synapses of blue crab gastric mill muscles. Cell Tissue Res 177:145–158
Atwood HL, Govind CK, Kwan I (1978) Nonhomogenous excitatory synapses of a crab stomach muscle. J Neurobiol 9:17–28
Calabrese RL, Maranto AR (1984) Neural control of the hearts in the leech, Hirudo medicinalis. III. Regulation of myogenicity and muscle tension by heart accessory neurons. J Comp Physiol A 154:393–406
Govind CK, Atwood HL, Maynard DM (1975) Innervation and neuromuscular physiology of intrinsic foregut muscles in the blue crab and spiny lobster. J Comp Physiol 96:185–204
Hagiwara S, Byerly L (1981) Calcium channel. Annu Rev Neurosci 4:69–125
Hartline DK, Maynard DM (1975) Motor patterns in the stomatogastric ganglion of the lobster Panulirus argus. J Exp Biol 62:405–420
Hooper SL, O'Neil MB, Wagner R, Ewer J, Golowasch J, Marder E (1986) The innervation of the pyloric region of the crab, Cancer borealis: homologous muscles in decapod species are differently innervated. J Comp Physiol A 159:227–240
Kunze JC (1981) The functional morphology of stomatopod Crustacea. Phil Trans R Soc Lond B 292:255–328
Lingle C (1981) The modulatory action of dopamine on crustacean foregut neuromuscular preparations. J Exp Biol 94:285–299
Maranto AR, Calabrese RL (1984) Neural control of the hearts in the leech, Hirudo medicinalis. II. Myogenic activity and its control by heart motor neurons. J Comp Physiol A 154:381–391
Maynard DM (1972) Simpler networks. Ann NY Acad Sci 193:59–72
Maynard DM, Dando MR (1974) The structure of the stomatogastric neuromuscular system in Callinectes sapidus, Homarus americanus, and Panulirus argus (Decapoda Crustacea). Phil Trans R Soc Lond B 268:161–220
Maynard DM, Selverston AI (1975) Organization of the stomatogastric ganglion of the spiny lobster. IV. The pyloric system. J Comp Physiol 100:161–182
Meiss DE, Norman RS (1977) Comparative study of the stomatogastric system of the decapod Crustacea. II. Musculature. J Morphol 152:55–76
Meyrand P, Moulins M (1986) Myogenic oscillatory activity in the pyloric rhythmic motor system of Crustacea. J Comp Physiol A 158:489–503
Meyrand P, Moulins M (1988a) Phylogenetic plasticity of crustacean stomatogastric circuits. I. Pyloric patterns and pyloric circuit of the shrimp Palaemon serratus. J Exp Biol 138:107–132
Meyrand P, Moulins M (1988b) Phylogenetic plasticity of crustacean stomatogastric circuits. II. Extrinsic inputs to the pyloric circuit of the shrimp Palaemon serratus. J Exp Biol 138:133–153
Noble D (1975) The initiation of the heartbeat. Clarendon, Oxford
Pinsker HM (1977) Aplysia bursting neurons as endogenous oscillators. I. Phase-response curves for pulsed inhibitory synaptic input. J Neurophysiol 40: 527–543
Reddy AR (1935) The structure, mechanism and development of the gastric armature in Stomatopoda with a discussion as to its evolution in Decapoda. Proc Ind Acad Sci BI: 650–675
Selverston AI, Moulins M (eds) (1987) The crustacean stomatogastric system. A model for the study of central nervous systems. Springer, Berlin Heidelberg New York
Selverston AI, Russell DF, Miller JP, King DG (1976) The stomatogastric nervous system: structure and function of a small neural network. Prog Neurobiol 7:215–289
Tazaki K (1988) The anatomy and physiology of the stomatogastric nervous system of Squilla. II. The cardiac system. Zool Sci 5:299–309
Tazaki K, Miyatani M, Ando F (1986) The anatomy and physiology of the stomatogastric nervous system of Squilla. I. The posterior cardiac plate and the pyloric systems. J Comp Physiol A 159:521–533
Watanabe A, Obara S, Akiyama T (1967) Pacemaker potentials for the periodic burst discharge in the heart ganglion of a stomatopod, Squilla oratoria. J Gen Physiol 50:839–862
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tazaki, K., Miyazaki, T. Neural control of the posterior cardiac plate and pyloric regions of the mantis shrimp Squilla oratoria: neurogenic and myogenic activities of muscles. J Comp Physiol A 168, 265–279 (1991). https://doi.org/10.1007/BF00218419
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00218419