Summary
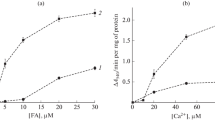
The possible property of fatty acid-binding proteins (FABPs) to transport fatty acid was investigated in various model systems with FABP preparations from liver and heart. An effect of FABP, however, was not detectable with a combination of oleic acid-loaded mitochondria and vesicles or liposomes due to the rapid spontaneous transfer. Therefore, the mitochondria were separated from the vesicles in an equilibrium dialysis cell. The spontaneous fatty acid transfer was much lower and addition of FABP resulted in an increase of fatty acid transport. Oleic acid was withdrawn from different types of monolayers by FABP with rates up to 10%/min. When two separate monolayers were used, FABP increased fatty acid transfer between these monolayers and an equilibrium was reached.
Similar content being viewed by others
Abbreviations
- FABP(s):
-
fatty acid-binding protein(s)
- PC:
-
phosphatidylcholine
- PS:
-
phosphatidylserine
- PE:
-
phosphatidylethanolamine
References
Glatz JF, Veerkamp JH: Intracellular fatty acid-binding proteins. Int J Biochem 17: 13–22, 1985
Sweetser DA, Heukenroth RO, Gordon JI: The metabolic significance of mammalian fatty acid-binding proteins: Abundant proteins in search of a function. Ann Rev Nutr 7: 337–359, 1987
Ockner RK, Manning JA, Kane JP: Fatty acid-binding protein: isolation from rat liver, characterization and immunological quantification. J Biol Chem 257: 7872–7878, 1982
Ketterer B, Tipping E, Hackney JA, Beale D: A low molecular weight protein from rat liver that resembles ligandin in its binding properties. Biochem J 155: 511–521, 1976
Burnett DA, Lysenko N, Manning JA, Ockner RK: Utilization of long-chain fatty acids by rat liver: studies of the role of FABP. Gastroenterology 77: 241–249, 1979
Daniels C, Noy N, Zakim D: Rates of hydration of fatty acids bound to unilamellar vesicles of phosphatidylcholine to albumin. Biochemistry 24: 3286–3291, 1985
Doody MC, Pownall HJ, Koa YJ, Smith LC: Mechanism and kinetics of transfer of a fluorescent fatty acid between single walled phosphatidylcholine vesicles. Biochemistry 19:108–116, 1980
Storch J, Kleinfeld A: Transfer of long-chain fluorescent free fatty acids between unilamellar vesicles. Biochemistry 25:1717–1726, 1986
Catala A, Avanzati B: Oleic acid transfer from microsomes to egg lecithin liposomes: participation of fatty acid-binding protein. Lipids 18: 803–807, 1983
McCormack M, Brecher P: Effect of liver fatty acid-binding protein on fatty acid movement between liposomes and rat liver microsomes. Biochem J 244: 717–723, 1987
Paulussen RA, van der Logt CP, Veerkamp JH: Characterization and binding properties of fatty acid-binding proteins from human, pig and rat heart. Arch Biochem Biophys 264: 533–545, 1988
Peeters RA, in't Groen MA, de Moel MP, van Moerkerk HT, Veerkamp JH: The binding affinity of fatty acid-binding proteins from human, pig and rat liver for different fluorescent fatty acids and other ligands. Int J Biochem, 1989
DiCorleto PE, Zilversmit DB: Protein catalyzed exchange of phosphatidylcholine between sonicated liposomes and multilamellar vesicles. Biochemistry 16: 2145–2150, 1977
Glatz JF, Paulussen RA, Veerkamp JH: Fatty acid-binding proteins from heart. Chem Phys Lipids 38: 115–129, 1985
Fournier NC, Mohammed R: Control of energy production in the heart: a new function for fatty acid-binding protein. Biochemistry 24: 2387–2396, 1985
Brecher P, Sauof R, Sugarman J, Eisenberg D, LaRosa K: Fatty acid transfer between multilamellar liposomes and fatty acid-binding proteins. J Biol Chem 259: 13395–13401, 1984
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Peeters, R.A., Veerkamp, J.H. Does fatty acid-binding protein play a role in fatty acid transport?. Mol Cell Biochem 88, 45–49 (1989). https://doi.org/10.1007/BF00223422
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00223422