Summary
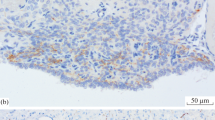
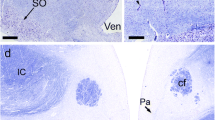
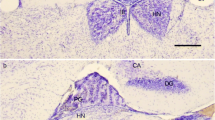
The innervation of the pineal organ was studied in 26 avian species under particular consideration of comparative aspects. A population of nerve cells and their pinealofugal (afferent) fiber systems were stained by means of the acetylcholinesterase method, while catecholamine-containing pinealopetal (efferent) fibers were demonstrated with the use of the glyoxylic acid method. Afferent axons were mainly found in the postero-proximal portion of the organ, and the patterns of their distribution were classified into three groups according to the characteristic densities of the reaction product. The number of acetylcholinesterase-positive neurons in the avian pineal organs examined in this study varied extremely from species to species, ranging from 0 to 362.
Catecholamine-containing nerve fibers penetrating the antero-lateral walls of the pineal follicles accompanied blood vessels and were arranged more densely in the distal portion of the organ, in contrast to the distribution of the acetylcholinesterase-positive nerve fibers. Three-dimensional reconstruction of the distributional patterns of both types of neural projections was performed for the pineal organ of every avian species examined. In avian species possessing a relatively conspicuous afferent projection, such as Passeriformes, Nycticorax, and Milvus, terminals of catecholamine-containing nerve fibers were observed exclusively in the interfollicular and perivascular tissues. In Galliformes, which display only few pineal afferents, catecholamine-containing fibers terminate not only in the interfollicular space, but also in the neuroepithelial parenchyma.
The regional differences in the innervation in the avian pineal organ suggest that the pinealocytes ranging from more sensory-like to more secretory-like elements are arranged in a mosaic-like pattern.
Similar content being viewed by others
References
Axelrod J, Wurtman RJ, Winget CM (1964) Melatonin synthesis in the hen pineal gland and its control by light. Nature 201:1134
Axelsson S, Björklund A, Falck B, Lindvall O, Svensson LA (1973) Glyoxylic acid condensation: A new fluorescence method for the histochemical demonstration of biogenic monoamines. Acta Physiol Scand 87:57–62
Binkley SA, Riebman JB, Reilly KB (1978) The pineal gland: A biological clock in vitro. Science 202:1198–1201
Björklund A, Lindvall O, Svensson LA (1972) Mechanisms of fluorophore formation in the histochemical glyoxylic acid method for monoamines. Histochemie 32:113–131
Boya J, Calvo J (1978) Post-hatching evolution of the pineal gland of the chicken. Acta Anat 101:1–9
Calvo J, Boya J (1979a) Ultrastructural study of the embryonic development of the pineal gland of the chicken (Gallus gallus). Acta Anat 103:39–73
Calvo J, Boya J (1979b) Evolution and nature of the dense bodies in the chicken pinealocytes. Acta Anat 104:61–71
Collin JP (1969) Contribution à l'étude de l'organe pineal. De l'épiphyse sensorielle à la glande pinéale: Modalités de transformation et implications fonctionnelles. Ann Stat Biol de Besseen Chandesse Suppl 1:1–359
Collin JP (1979) Recent advances in pineal cytochemistry. Evidence of the production of indolamines and proteinaceous substances by rudimentary photoreceptor cells and pinealocytes of amniota. In: Ariëns Kappers J, Pévet P (eds) The pineal gland of vertebrates including man (Progr in Brain Research, vol 52) Elsevier, Amsterdam, pp 271–295
Collin JP, Oksche A (1981) Structural and functional relationships in the nonmammalian pineal gland. In: Reiter RJ (ed) The pineal gland (Anatomy and Biochemistry, vol 1) CRS Press Inc, USA, pp 27–67
Deguchi T (1979a) Circadian rhythm of serotonin N-acetyltransferase activity in organ culture of chicken pineal gland. Science 203:1245–1247
Deguchi T (1979b) A circadian oscillator in cultured cells of chicken pineal gland. Nature 282:94–96
Deguchi T (1981) Rhodopsin-like photosensitivity of isolated chicken pineal gland. Nature 290:702–704
El-Badawi A, Schenk EA (1967) Histochemical methods for separate, consecutive, and simultaneous demonstration of acetylcholinesterase and norepinephrine in cryostat sections. J Histochem Cytochem 15:580–588
Eränkö O, Eränkö L (1971) Loss of histochemically demonstrable catecholamines and acetyl-cholinesterase from sympathetic nerve fibers of the pineal body of the rat after chemical sympathectomy with 6-hydroxydopamine. Histochem J 3:357–363
Eränkö O, Rechardt L, Eränkö L, Cunningham A (1970) Light and electron microscopic histochemical observations on cholinesterase-containing sympathetic nerve fibers in the pineal body of the rat. Histochem J 2:479–489
Falck B, Hillarp NÅ, Thieme G, Torp A (1962) Fluorescence of catecholamines and related compounds condensed with formaldehyde. J Histochem Cytochem 10:348–354
Falcon J (1979) Unusual distribution of neurons in the pike pineal organ. In: Ariëns Kappers J, Pévet P (eds) The pineal gland of vertebrates including man (Progr in Brain Research, vol 52). Elsevier, Amsterdam, pp 89–91
Gaston S, Menaker M (1968) Pineal function: The biological clock in the sparrow? Science 160:1125–1127
Hedlund L (1970) Sympathetic innervation of the avian pineal body. Anat Rec 166:406
Hedlund L, Nalbandov AV (1969) Innervation of the avian pineal body. Am Zool 9:1090
Hetherington T (1981) Morphology of the pineal organ in the salamander, Ensatina eschscholtzi J Morphol 169:191–206
Kappers JA (1960) The development topographical relations and innervation of the epiphysis cersebri in the albino rat. Z Zellforsch 52:163–215
Kappers JA (1965) Survey of the innervation of the epiphysis cerebri and the accessory pineal organs of vertebrates. In: Ariëns Kappers J, Schadé JP (eds) Structure and function of the epiphysis cerebri (Progr in Brain Research, vol 10). Elsevier, Amsterdam, pp 87–153
Karnovsky MJ, Roots L (1964) A “direct coloring” thiocholine method for cholinesterase. J Histochem Cytochem 12:219–221
Korf HW (1974) Acetylcholinesterase-positive neurons in the pineal and parapineal organs of the rainbow trout, Salmo gairdneri (with special reference to the pineal tract). Cell Tissue Res 155:475–489
Korf HW, Liesner R, Meissl H, Kirk A (1981) Pineal complex of the clawed toad, Xenopus laevis Daud.: Structure and function. Cell Tissue Res 216:113–130
Korf HW, Zimmerman NH, Oksche A (1982) Intrinsic neurons and neural connections of the pineal organ of the house sparrow, Passer domesticus, as revealed by anterograde and retrograde transport of horseradish peroxidase. Cell Tissue Res 222:243–260
Lindvall O, Björklund A (1974) The glyoxylic acid fluorescence histochemical method: a detailed account of the methodology for the visualization of central catecholamine neurons. Histochemistry 39:97–127
Lindvall O, Björklund A, Hökfelt T, Ljungdahl Å (1973) Application of the glyoxylic acid method to vibratome sections for the improved visualization of central catecholamine neurons. Histochemie 35:31–38
Matsuura T, Herwig HJ (1981) Histochemical and ultrastructural study of the nervous elements in the pineal organ of the eel, Anguilla anguilla. Cell Tissue Res 216:545–555
Ohba S, Wake K, Ueck M (1979a) Histochemical and electron-microscopical findings in the pineal organ of Carassius gibelio (Langsd.). In: Ariëns Kappers J, Pévet P (eds) The pineal gland of vertebrates including man (Progr in Brain Research, vol 52). Elsevier, Amsterdam, pp 93–96
Ohba S, Wake K, Ohnishi R, Ueck M (1979b) Neue Befunde am pinealen Sinnesapparat von Funa, Carassius gibelio langsdorfi (Teleostei). Verb Anat Ges 73:953–959
Oksche A, Kirschstein H (1969) Elektronenmikroskopische Untersuchungen am Pinealorgan von Passer domesticus. Z Zellforsch 102:214–241
Oksche A, Vaupel-von Harnack M (1966) Elektronenmikroskopische Untersuchungen zur Frage der Sinneszellen im Pinealorgan der Vögel. Z Zellforsch 69:41–60
Oksche A, Morita Y, Vaupel-von Harnack M (1969) Zur Feinstruktur und Funktion des Pinealorgans der Taube (Columba livia). Z Zellforsch 102:1–30
Oksche A, Kirschstein H, Kobayashi H, Farner DS (1972) Electron microscopic and experimental studies of the pineal organ in the white-crowned sparrow, Zonotrichia leucophrys gambelii. Z Zellforsch 124:247–274
Omura Y (1980) Histochemical and ultrastructural studies on the nervous organization of the pineal organ of the ayu, Plecoglossus altivelis. Bull Jap Soc Sci Fish 46:1483–1488
Owman C, Rüdeberg C (1970) Light, fluorescence, and electron microscopic studies on the pineal organ of the pike, Esox lucius L., with special regard to 5-hydroxytryptamine. Z Zellforsch 107:522–550
Owman C, Rüdeberg C, Ueck M (1970) Fluoreszenzmikroskopischer Nachweis biogener Monoamine in der Epiphysis cerebri von Rana esculenta und Rana pipiens. Z Zellforsch 111:550–558
Quay WB (1966) Rhythmic and light-induced changes in levels of 5-hydroxyindoles in the pigeon (Columba livia). Gen Comp Endocrinol 6:371–377
Sato T, Wake K (1981) Organization of the sensory and sympathetic nerves in the avian pineal organs. Jikeikai Med J 28 (Suppl 1):7–12
Sato T, Wake K (1982) Nervous organization in the pineal organs of birds. In: Mikami S et al. (eds) Avian endocrinology: Environmental and ecological perspectives. Japan Sci Soc Press, Tokyo, Springer-Verlag, Berlin, 57–65
Ueck M (1969) Zur Ultrastruktur der Epiphysis cerebri der Vögel. (Verh Zool Ges) Zool Anz Suppl 33:509–518
Ueck M (1970) Weitere Untersuchungen zur Feinstruktur und Innervation des Pinealorgans von Passer domesticus L. Z Zellforsch 105:276–302
Ueck M (1973) Fluoreszenz- und elektronenmikroskopische Untersuchungen am Pinealorgan verschiedener Vogelarten. Z Zellforsch 137:37–62
Ueck M, Kobayashi H (1972) Vergleichende Untersuchungen über acetylcholinesterasehaltige Neurone im Pinealorgan der Vögel. Z Zellforsch 129:140–160
Ueck M, Wake K (1982) Histochemische und elektronenmikroskopische Befunde zum Pinealorgan des schwarzen Milans, Milvus migrans (Falconidae). Verh Anat Ges 76:457–460
Ueck M, Ohnishi R, Wake K (1977) Photoreceptor-like outer segments in the pineal organ of the lovebird, Uroloncha domestica (Aves: Passeriformes). A scanning electron microscopic study. Cell Tissue Res 182:139–143
Vigh B, Vigh-Teichmann I (1981) Light- and electron-microscopic demonstration of immunore-active opsin in the pinealocytes of various vertebrates. Cell Tissue Res 221:451–463
Vigh-Teichmann I, Korf HW, Oksche A, Vigh B (1982) Opsin-immunoreactive outer segments and acetylcholinesterase-positive neurons in the pineal complex of Phoxinus phoxinus (Teleostei, Cyprinidae). Cell Tissue Res 227:351–369
Wake K (1973) Acetylcholinesterase-containing nerve cells and their distribution in the pineal organ of the goldfish, Carassius auratus. Z Zellforsch 145:287–298
Wake K, Ueck M, Oksche A (1974) Acetylcholinesterase-containing nerve cells in the pineal complex and subcommissural area of the frogs, Rana ridibunda and Rana esculenta. Cell Tissue Res 154:423–442
Wight PAL, Mackenzie GM (1970) Dual innervation of the pineal of the fowl, Gallus domesticus. Nature (Lond) 228:474–475
Zimmerman NH, Menaker M (1975) Neural connections of sparrow pineal: Role in circadian control of activity. Science 190:477–479
Author information
Authors and Affiliations
Additional information
This work was supported by a grant (No. 56480080) from the Ministry of Education, Science and Culture of Japan
Scholar of the Alexander von Humboldt Foundation
Rights and permissions
About this article
Cite this article
Sato, T., Wake, K. Innervation of the avian pineal organ. Cell Tissue Res. 233, 237–264 (1983). https://doi.org/10.1007/BF00238294
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00238294