Summary
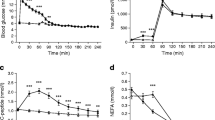
Hepatic glucose production and metabolic clearance rate of glucose were measured using (3-3H) glucose at steady state, basally and during two sequential 2 h insulin (25 and 40mU · kg–1 · h–1)/glucose(2 and 3mg · kg–1 · min–1) infusion periods. Eight diabetic subjects were studied before and after 1 week of twice daily insulin therapy; six control subjects matched for age, weight and degree of obesity were also studied. In the diabetic patients, pre-treatment hepatic glucose production was 20.0 ± 2.2, 9.9 ± 2.9, and 1.4 ± 0.8 μmol · kg–1 · min–1 respectively (± SEM) for each of the three periods, and fell significantly with treatment to 12.8 ± 1.7,4.0 ± 1.5 and 1.9 ± 1.0 μmol · kg–1 · min–1. Hepatic glucose production in normal subjects was 13.2 ± 0.6, 2.2 ± 0.8 and < 1 μmol · kg–1 · min–1. The pre-treatment metabolic clearance rate in all diabetic studies with insulin levels ⩾ 30 mU/l was 1.10 ± 0.14 ml · kg–1 · min–1 and remained virtually unchanged following insulin therapy; this was significantly lower than in the control subjects (6.83 ± 1.02, p < 0.001). Basal non-esterified fatty acid levels were higher (p < 0.02) in the pre-treated diabetic patients compared to post-treated diabetic patients and control subjects. Non-esterified fatty acids in each group fell to similar levels during the insulin infusions, but the rate of fall was slower in the pre-treated diabetic patients. Insulin receptor binding to erythrocytes was normal in the diabetic subjects and unchanged by treatment. Therefore, following insulin treatment of uncontrolled Type 2 (non-insulin-dependent) diabetes, the initially increased basal hepatic glucose production, and decreased hepatic sensitivity, return towards normal. However, the glucose clearance remains low, despite good diabetic control, and appears to be a major factor in the continuing glucose intolerance. As insulin receptor binding is normal, the defect of glucose clearance in Type 2 diabetes appears compatible with a post-receptor defect of glucose metabolism.
Article PDF
Similar content being viewed by others
References
Reaven G, Olefsky J (1978) The role of insulin resistance in the pathogenesis of diabetes mellitus. Adv Metab Disord 9: 313–331
Olefsky J (1981) Insulin resistance and insulin action. An in vitro and in vivo perspective. Diabetes 30: 148–162
Alford F, Martin F, Pearson M (1971) The significance and interpretation of mildly abnormal oral glucose tolerance. Diabetologia 7: 173–180
Kalant N, Csorba T, Heller N (1963) Effect of insulin on glucose production and utilization in diabetes. Metabolism 12: 1100–1111
Myers J (1950) Net splanchnic glucose production in normal man and in various disease states. J Clin Invest 29: 1421–1429
Bearn A, Billing B, Sherlock S (1951) Hepatic glucose output and hepatic insulin sensitivity in diabetes mellitus. Lancet 2: 698–701
DeFronzo R, Deibert D, Hendler R, Felig P, Soman V (1979) Insulin sensivitivity and insulin binding to monocytes in maturity-onset diabetes. J Clin Invest 63: 939–946
Forbath N, Hetenyi Jr G (1966) Glucose dynamics in normal subjects and diabetic patients before and after a glucose load. Diabetes 15: 778–789
Bowen H, Moorhouse J (1973) Glucose turnover and disposal in maturity-onset diabetes. J Clin Invest 52: 3033–3045
Kimmerling G, Javorski W, Olefsky J, Reaven G (1976) Locating the site(s) of insulin resistance in patients with nonketotic diabetes mellitus. Diabetes 25: 673–678
Hall SE, Saunders J, Sonksen PH (1979) Glucose and free fatty acid turnover in normal subjects and in diabetic patients before and after insulin treatment Diabetologia 16: 297–306
Steele R, Wall J, de Bodo RC, Altszuler N (1956) Measurement of size and turnover rate of body glucose pool by the isotope dilution method. Am J Physiol 187: 15–24
Proietto J, Alford F, Dudley F (1980) The mechanism of carbohydrate intolerance of cirrhosis. J Clin Endocrinol Metab 51: 1030–1036
Alford F, Bloom S, Nabarro J (1977) Glucagon levels in normal and diabetic subjects: use of a specific immunoabsorbent for glucagon radioimmunoassay. Diabetologia 13: 1–6
Altszuler N, Bardai A, Bjerkes C, Gottlieb B, Steele R (1975) Glucose turnover values in the dog obtained with various species of labelled glucose. Am J Physiol 229: 1662–1667
Albano J, Ekins R, Maritz G, Turner R (1972) A sensitive precise radioimmunoassay of serum insulin relying on charcoal separation of bound and free moieties. Acta Endocrinologia 70: 487–509
Carruthers M, Young D (1973) Free fatty acid estimation by a semiautomated fluorimetric method. Clin Chim Acta 49: 341–343
Williamson D, Mallanby J, Krebs H (1962) Enzymic determination of D(-)-β-hydroxybutyric acid and acetoacetic acid in blood. Biochem J 82: 190–196
Gambhir K, Archer J, Carter L (1977) Insulin radioreceptor assay for human erythrocytes. Clin Chem 23: 1590–1595
Steele R (1959) Influences of glucose loading and injected insulin on hepatic glucose output. Ann NY Acad Sci 82: 420–430
Riggs DA (1963) The mathematical approach to physiological problems. Williams & Wilkins, Baltimore, pp 913–929
Proietto J, Harewood M, Aitken P, Nankervis A, Caruso G, Alford F (1982) Validation of a practical in vivo insulin dose-response curve in man. Metabolism 31: 354–361
Best J, Judzewitsch R, Pfeifer M, Beard J, Halter J, Porte D, Jr (1982) The effect of chronic sulfonylurea therapy on hepatic glucose production in non-insulin dependent diabetes. Diabetes (in press)
Brown P, Tompkins C, Juul S, Sonksen P (1978) Mechanism of action of insulin in diabetic patients: a dose related effect on glucose production and utilization. Br Med J 1: 1239–1242
Sacca L, Hendler R, Sherwin R (1978) Hyperglycaemia inhibits glucose production in man, independent of changes in glucoregulatory hormones. J Clin Endocrinol Metab 47: 1160–1163
Liljenquist J, Mueller G, Cherrington A, Perry J, Rabinowitz D (1979) Hyperglycaemia per se (insulin and glucagon withdrawn) can inhibit hepatic glucose production in man. J Clin Endocrinol Metab 48: 171–175
Nankervis A, Proietto J, Ng K, Alford F, Larkins R (1981) The metabolic effects of chronic hyperglucagonaemia. Clin Endocrinol 15: 325–333
Soskin S, Levine R (1937) A relationship between the blood sugar level and the rate of sugar utilization, affecting the theories of diabetes. Am J Physiol 120: 761–770
Reaven G, Silvers A, Farquhar J (1970) Study of the relationship between plasma insulin concentration and efficiency of glucose uptake in normal and mildly diabetic subjects. Diabetes 19: 571–578
Davis B, Bernstein R, Kolterman O, Olefsky J, Reaven G (1978) Defect in glucose removal in nonketotic diabetic patients with fasting hyperglycaemia. Diabetes 28: 32–35
Olefsky J, Farquhar J, Reaven G (1973) Relationship between fasting plasma insulin level and resistance to insulin-mediated glucose uptake in normal and diabetic subjects. Diabetes 22: 507–513
Randle P, Garland P, Hales C, Newsholm E, Denton R, Pogson C (1966) Interactions of metabolism and the physiological role of insulin. Recent Prog Horm Res 22: 1–48
Olefsky J, Kolterman O (1981) Mechanisms of insulin resistance in obesity and non-insulin-dependent (Type 2) diabetes. Am J Med 70: 151–168
Beck-Nielson H, Pedersen O, Sorensen N (1980) Effects of dietary changes on cellular insulin binding and in vivo insulin sensitivity. Metabolism 29: 482–487
Reaven G, Miller R (1968) Study of the relationship between glucose and insulin responses to an oral glucose load in man. Diabetes 17: 560–567
Zimmet P, Whitehouse S, Alford F, Chisholm D (1978) The relationship of insulin response to a glucose stimulus over a wide range of glucose tolerance. Diabetologia 15: 23–27
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nankervis, A., Proietto, J., Aitken, P. et al. Differential effects of insulin therapy on hepatic and peripheral insulin sensitivity in Type 2 (non-insulin-dependent) diabetes. Diabetologia 23, 320–325 (1982). https://doi.org/10.1007/BF00253737
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00253737