Summary
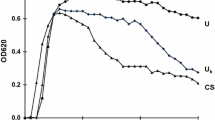
Ultrafiltration membranes of 10,000 d, 1,000 d and 500 d were used to remove urinary macromolecules from the urine of normal subjects and from the urine of stone forming patients. The filtrated urines were examined for their residual inhibitory potential for calcium-oxalate precipitation, by the discrimination method of Sarig et al. (D.I. test). The results of testing the filtrate were complementary to the information gained by analyses of retentates obtained in successive ultrafiltration. The method has an inherent advantage because the manipulation of solids retained on membranes may inadvertantly modify their inhibitory potential. At least two distinct groups of inhibitors were found in 20 normal urines. The first group has MW above 10,000 d while the second group of inhibitors has MW in the range of 500–1,000 d. The mean of the D.I. values increased dramatically from the normal range (<0.6) to the stone former range (>1.1) (p<0.001) after the 500 d filtration. Some of the normal urines, even after the 500 d filtration, still had a degree of inhibitory potential. This inhibitory potential may be related to the inorganic compounds which were found in the urines. The inhibitory activity of macromolecules with MW above 10,000 d and below 500 δ was negligible in 7 stone formers (SF) urines. The relative contribution of 500–1,000 d macromolecules is the highest both in SF and normal urines. Conclusions: 1) inhibitors in human urine are of wide range in MW; 2) stone formers and normals differ in the level of inhibitor activity at all MW ranges; especially in above 10,000 d and below inhibitors.
Similar content being viewed by others
References
Boyce WH, Swanson M (1955) Biocolloids of urine in health and in calculous disease. II. Electrophoretic and biochemical studies of mucoprotein insoluble in molar sodium chloride. J Clin Invest 34:1581
Pinto B, Bernshtam J, Paternian JL (1981) Mechanism of the heterogenous nucleation by a urinary mucoprotein. In: Smith LH et al (eds) Urolithiasis, clinical and basic research. Plenum Press, p 697
Hallson P, Rose GA (1979) Uromucoids and urinary stone formation. Lancet 12:1000
Garti N, Sarig S, Tibika F (1980) Retardation of calcium oxalate formation by polyacidic peptides. Invest Urol 18:149
Robertson EG, Scure DS, Bridge CM (1981) Factors influencing the crystallization of calcium oxalate in urine-critique. JCS 53:182
Nakagawa Y, Kaiser ET, Coe FL (1978) Isolation and characterization of calcium oxalate crystal growth inhibitors from human urine. Biochem Biophys Res Commun 84:1038
Gill WB, Karesh JW, Garsin L, Roma MJ (1977) Inhibitory effects of urinary macromolecules on the crystallization of calcium oxalate. Invest Urol 15:95
Fleisch H (1978) Inhibitors and promotors of stone formation. Kidney Int 13:361
Sallis JD, Lumley MF (1979) On the possible role of glycosaminoglycans as natural inhibitors of calcium oxalate stones. Invest Urol 16:296
Drach GW, Kraljevich Z, Randolph AD (1982) Effects of high molecular weight urinary macromolecules on crystallization of calcium oxalate dihydrate. J Urol 127:805
Garti N, Tibika F, Sarig S, Perlberg S (1981) The inhibitory effect of polymeric carboxylic amino acid and urine on calcium oxalate crystallization. In: Smith LH, Robertson WG, Finlayson B (eds) Urolithiasis, clinical and basic research. Plenum Press, New York London, p 567
Resnick MI, Boyce WH (1979) Low molecular weight urinary proteins and renal lithiasis. Invest Urol 16:270
Gardner GL, Doremus RH (1978) Crystal growth inhibitors in human urine. Effect on calcium oxalate kinetics. Invest Urol 15:478
Bowyer RC, Brockis JG, McCulloch RK (1979) Glycosaminoglicans as inhibitors and calcium oxalate crystal growth and aggregation. Clin Chim Acta 95:23
Bourrillon R (1970) Glycoproteins, glycopeptides and other carbohydrate complexes of normal human urine. In: Revillard MY, Baltimor BH (eds) Proteins in normal and pathological urine. University Park Press, p 20
McGeown MG (1957) The urinary amino acids in relation to calculus disease. J Urol 78:318
Azoury R, Garti N, Perlberg S, Sarig S (1982) May enzyme activity in urine play a role in kidney stone formation. Urol Res 10:185
Fleish H (1975) Mechanism of stone formation: role of promoters and inhibitors. In: Danielson BG (ed) Symposium on urolithiasis. The Wellcome Foundation, p 53
Sarig S, Garti N, Azoury R, Wax Y, Perlberg S (1982) A method for discrimination between calcium oxalate kidney stone formers and normals. J Urol 128:645
Morison DF (1967) Multivariate statistical methods. McGraw-Hill, New York
Beale EML, Little RJA (1975) Missing values in multivariate analysis. J R Stat Soc B 37:129
Dunnett CW (1980) Pairwise multiple comparison in the homogeneous variance, unequal sample size case. J Am Stat Assoc 75:789
Brown MB, Forsy AB (1974) The small sample behavior of some statistics which test the equality of several means. Technometrics 16:129
Dunnett CW (1980) Pairwise multiple comparisons in the unequal variance case. J Am Stat Assoc 75:796
Bichler KH, Kirchner CH, Ideler V (1976) Uromucoid excretion of normal individuals and stone formers. Br J Urol 47:733
Samuell CT (1979) Uromucoid excretion in normal subjects, calcium stone formers and in patients with chronic renal failure. Urol Res 7:5
Kitamura T, Zerwekh JE, Pak CYC (1982) Partial biochemical and physicochemical characterization of organic macromolecules in urine from patients with renal stones and control subjects. Kidney Int 21:379
Nicar MJ, Holt K, Pak CYC (1980) Ion binding by small molecular weight substances (1,000–10,000 daltons) in urine. Invest Urol 18:162
Meyer JL, Smith LH (1975a) Growth of calcium oxalate crystals. II. Inhibition by natural crystal growth inhibitors. Inves Urol 13:36
Gill WB, Karesh JW (1976) Demonstration of protective (inhibitory) effects of urinary macromolecules on the crystallization of calcium oxalate. In: Fleisch H, et al (eds) Urolithiasis research. Plenum Press, New York, p 227
Goldwasser B, Sarig S, Azoury R, Orda S, Boichis H, Wax Y, Many M (1983) Hyperuricosuria and calcium oxalate stone formation — is there a relation. Suppl to J Urol
Drach GW (1978) Urinary lithiasis. In: Harrison JH et al (eds) Campbell's Urology. Voll 1. Saunders Co, p 779
Meyer JL, Angino EE (1977) The role of trace metals in calcium urolithiasis. Invest Urol 14:347
Kitamura T, Pack CYC (1982) Tamm and Horsfoll glycoprotein does not promote spontaneous precipitation and crystal growth of calcium oxalate in vitro. J Urol 127:1024
Robertson WG, Peacock M, Nordin BEC (1973) Inhibitors of the growth and aggregation of calcium oxalate crystals in vitro. Clin Chim Acta 43:31
Ito H, Coe FL (1977) Acidic peptide and polyribonucleatide crystal growth inhibitors in human urine. Am J Physiol 233: F455
Drach GW, Sarig S, Randolph AD, Thoreson S (1982) The paradox of inhibition and enhancement of the formation of urinary stones. Urol Res 10:165
Lanzalaco AC, Sheehan ME, White DJ, Nancollas GH (1982) The mineralization inhibitory potential of urines: A constant composition approach. J Urol 128:845
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Azoury, R., Goldwasser, B., Wax, Y. et al. Evaluation of the relative inhibitory potential of fractionated urinary macromolecules. Urol. Res. 13, 199–205 (1985). https://doi.org/10.1007/BF00261824
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00261824