Abstract
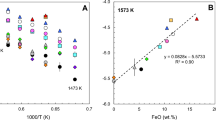
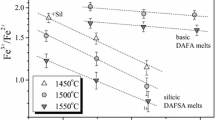
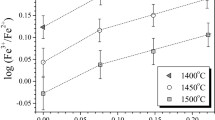
Ferric-ferrous ratios have been measured in 22 experiments on three natural compositions equilibrated at known temperature (950°–1100° C) and oxygen fugacity, and at water-saturated conditions over a pressure range from 0.05 to 0.2 GPa. There does not appear to be any reaction between the melt and the capsule material that affects the redox state of the iron in the melt. An empirical expression for the anhydrous behavior of the redox state of iron in each of these compositions has also been determined at 1 bar as a function of temperature and oxygen fugacity. A direct comparison of the hydrous ferric-ferrous values with the calculated anhydrous values shows that the dissolution of water in a per-alkaline rhyolite, andesite, and an augite minette has no effect on the redox state of the iron in these melts. This result parallels the effect of water on sulfide speciation in basaltic melts, and confirms published results on experimental hydrous basalts.
Similar content being viewed by others
References
Baker L, Rutherford MJ (1992) The effect of water saturation on the ferric-ferrous ratio in silicic melts (abstract). EOS, Trans Am Geophys Union 73:351
Burnham CW, Davis NF (1974) The role of H2O in silicate melts: II. Thermodynamic and phase relations in the system NaAl-Si3O8-H2O to 10 kilobars, 700 deg to 1100 deg C. Am J Sci 274:902–940
Candela PA (1986) The evolution of aqueous vapor from silicate melts: effect on oxygen fugacity. Geochim Cosmochim Acta 50:1205–1211
Carmichael ISE (1991) The redox states of basic and silicic magmas: a reflection of their source regions? Contrib Mineral Petrol 106:129–141
Carroll MR, Rutherford MJ (1985) Sulfide and sulfate saturation in hydrous silicate melts. J Geophys Res 90:C601-C612
Dingwell DB (1991) Redox viscometry of some Fe-bearing silicate melts. Am Mineral 76:1560–1562
Dingwell DB, Virgo D (1987) The effect of oxidation state on the viscosity of melts in the system Na2O-FeO-Fe2O3-SiO2. Geochim Cosmochim Acta 51:195–205
Hamilton DL, Burnham CW, Osborn EF (1964) The solubility of water and effects of oxygen fugacity and water content on crystallization in mafic magmas. J Petrol 5:21–39
Holloway JR (1987) Igneous fluids In: Engster HP, Carmichael ISE (eds) Thermodynamic modeling of geological materials: minerals, fluids and melts (Reviews in Mineralogy vol. 17). Mineralogical Society of America, Washington DC, pp 211–234
Holloway JR, Burnham CW (1972) Melting relations of basalt with equilibrium water pressure less than total pressure. J Petrol 13:1–29
Holloway JR, Dixon JE, Pawley AR (1992) An internally heated, rapid-quench, high pressure vessel. Am Mineral 77:643–646
Johnson JW, Oelkers EH, Helgeson HC (1992) SUPCRT92: a software package for calculating the standard molal thermodynamic properties of minerals, gases, aqueous species, and reactions from 1 to 5000 bar and 0 to 10 000°C. Computers and Geosciences 18:899–947
Kilinc A, Carmichael ISE, Rivers ML, Sack RO (1983) The ferricferrous ratio of natural silicate liquids equilibrated in air. Contrib Mineral Petrol 83:136–140
Kress VC, Carmichael ISE (1991) The compressibility of silicate liquids containing Fe2O3 and the effect of composition, temperature, oxygen fugacity and pressure on their redox states. Contrib Mineral Petrol 108:82–92
Lange RA, Carmichael ISE (1989) Ferric-ferrous equilibria in Na2O-FeO-Fe2O3-SiO2 melts: effects of analytical techniques on derived partial molar volumes. Geochim Cosmochim Acta 53:2195–2204
Mattioli GS, Baker MB, Rutter MJ, Stolper EM (1989) Upper mantle oxygen fugacity and its relationship to metasomatism. J Geol 97:521–536
Mueller RF (1971) Oxidative capacity of magmatic components. Am Sci 270:236–243
Naney MT, Swanson SE (1983) Iron redox kinetics in silicic liquids. Geol Soc Am Abstr Prog 15:650
Naney MT, Swanson SE (1984) Iron redox kinetics in silicic melts. EOS Trans Am Geophys Union 65:1150
Nicholls J, Carmichael ISE (1969) Peralkaline acid liquids: a petrological study. Contrib Mineral Petrol 20:268–294
Osborn EF (1959) Role of oxygen pressure in the crystallization and differentiation of basaltic magma. Am Sci 257:609–647
O'Neill HStC, Pownceby MI (1993) Thermodynamic data from redox reactions at high temperatures. I. An experimental and theoretical assessment of the electrochemical method using stabilized zirconia electrolytes, with revised values for the Fe-“FeO”, Co-CoO, Ni-NiO and Cu-Cu2O oxygen buffers, and new data for the W-WO2 buffer. Contrib Mineral Petrol 114:296–314
Pownceby MI, O'Neill HStC (1994) Thermodynamic data from redox reactions at high temperatures. III. Activity-composition relations in Ni-Pd alloys from EMF measurements at 850–1250K, and calibration of the NiO+Ni-Pd assemblage as a redox sensor. Contrib Mineral Petrol 116:327–339
Sack RO, Carmichael ISE, Rivers M, Ghiorso MS (1980) Ferricferrous equilibria in natural silicate liquids at 1 bar. Contrib Mineral Petrol 75:369–376
Sato M (1978) Oxygen fugacity of basaltic magmas and the role of gas-forming elements. Geophys Res Lett 5:447–449
Sisson TW, Grove TL (1993) Experimental investigations of the role of H2O in calc-alkaline differentiation and subduction zone magmatism. Contrib Mineral Petrol 113:143–166
Taylor JR, Wall VJ, Pownceby MI (1992) The calibration and application of accurate redox sensors. Am Mineral 77:284–295
Tormey DR, Grove TL, Bryan WB (1987) Experimental petrology of normal MORB near the Kane Fracture Zone: 22°–25° N, mid-Atlantic ridge. Contrib Mineral Petrol 96:121–139
Tuttle OF, Bowen NL (1958) Origin of granite in the light of experimental studies in the system NaAlSi3O8-KAlSi3O8-SiO2-H2O. Geol Soc Am Mem 74
Wallace PJ, Carmichael ISE (1994) S speciation in submarine basaltic glasses as determined by measurements of S K alpha x-ray wavelength shifts. Am Mineral 79:161–167
Wilson AD (1960) The micro-determination of ferrous iron in silicate minerals by a volumetric and a colorimetric method. Analyst 85:823–827
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moore, G., Righter, K. & Carmichael, I.S.E. The effect of dissolved water on the oxidation state of iron in natural silicate liquids. Contrib Mineral Petrol 120, 170–179 (1995). https://doi.org/10.1007/BF00287114
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00287114