Abstract
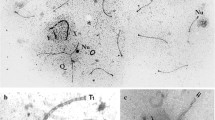
In chiasmatic meiosis of mosquitoes, ascomycetes and lilies the synaptinemal complex (SC) disassociates from the bivalent before metaphase I. Conversely, in the achiasmatic meiosis of Bolbe nigra, the SC remains associated with the bivalent during first metaphase. Light microscopy reveals mid-bodies between disjoining half-bivalents during early first anaphase in Bolbe. Optically controlled serial sections for electron microscopy show that the mid-bodies seen in light micrographs and synaptinemal complexes seen in electron micrographs are the same structure. Electron micrographs indicate that the SC breaks transversely at a point corresponding to the chromosomal kinetochore during anaphase I as the chromatin and the SC begin to separate. During telophase I, SC remnants are at the poles with the chromosomes or between poles. Presently, the evidence is inadequate to state whether the SC serves alternately or simultaneously as a biological contrivance for conjunction and crossing-over or singly as a device for one of these phenomena.
Similar content being viewed by others
References
Cooper, K. W.: The nuclear cytology of the grass mite, Pediculopsis graminum (Reut.), with special reference to karyomerokinesis. Chromosoma (Berl.) 1, 4–103 (1939).
—: Meiotic conjunctive elements not involving chiasmata. Proc. nat. Acad. Sci. (Wash.) 52, 1248–1255 (1964).
Fogg, L. C.: A study of chromatin diminution in Ascaris and Ephestia. J. Morph. 50, 413–452 (1930).
Franchi, L. L., and A. M. Mandl: The ultrastructure of oogonia and oocytes in the foetal and neonatal rat. Proc. roy. Soc. B 157, 99–114 (1963).
Fry, H. J.: Studies of the mitotic figure. VI. Mid-bodies and their significance for the central body problem. Biol. Bull. 73, 565–590 (1937).
Gassner, G.: Synaptinemal complexes: Recent findings. J. Cell Biol. 35, 166A-167A (1967).
Hughes-Schrader, S.: Meiosis without chiasmata in diploid and tetraploid spermatocytes of the mantid Callimantis antillarum Saussure. J. Morph. 73, 111–141 (1943).
Kawaguchi, E.: Zytologische Untersuchungen am Seidenspinner und seinen Verwandten, I. Gametogenese von Bombyx mori L. und B. mandarina M. und ihrer Bastarde. Z. Zellforsch. 7, 519–552 (1928).
Klingstedt, H.: Diagametie beim Weibchen der Trichoptere Limnophilus decipiens Kol. Acta zool. fenn. 10, 1–69 (1931).
Lu, B. C.: Meiosis in Coprinas lazopus: A comparative study with light electron microscopy. J. Cell Sci. 2, 529–536 (1967).
Luft, J. H.: Improvements in epoxy resin embedding methods. J. biophys. biochem. Cytol. 9, 409–415 (1961).
Meyer, G.: A possible correlation between the submicroscopic structure of meiotic chromosomes and crossing-over. In: Electron microscopy (M. Titlbach, ed.) B, p. 461–462. Proc. Third Eur. Reg. Conf. Electron Microscopy, Prague, 1964.
Moens, P. B.: The structure and function of the synaptinemal complex in Lilium longiflorum sporocytes. Chromosoma (Berl.) 23, 418–451 (1968).
Moses, M. J., and J. R. Coleman: Structural patterns and the functional organization of chromosomes. In: The role of chromosomes in development (M. Locke, ed.), p. 11–49. 23rd Symp. Soc. Stud. Growth and Develop., 1964.
Peacock, W. J.: Replication, recombination and chiasmata in Goniaea australiasiae (Orthoptera: Acrididae). Genetics (in press, 1968).
Roth, T. F.: Changes in the synaptinemal complex during meiotic prophase in mospuito oocytes. Protoplasma (Wien) 61, 346–386 (1966).
—, and M. Ito: DNA-dependent formation of the synaptinemal complex at meiotic prophase. J. Cell Biol. 35, 247–255 (1967).
Schin, K. S.: Meiotische Prophase und Spermatidenreifung bei Gryllus domesticus, mit besonderer Berücksichtigung der Chromosomenstruktur. Z. Zellforsch. 65, 481–513 (1965).
Seiler, J.: Das Verhalten der Geschlechtschromosomen bei Lepidopteren. Nebst einem Beitrag zur Kenntnis von Eireifung, Samenreifung und Befruchtung. Arch. Zellforsch. 13, 152–269 (1914).
—: Geschlechtschromosomenuntersuchungen an Psychiden. IV. Die Parthenogenese der Psychiden. Z. indukt. Abstamm.- u. Vererb.-L. 31, 1–99 (1923).
Sotelo, J. R., and O. Trujillo-Cenóz: Electron microscope study on spermatogenesis. Chromosome morphogenesis at the onset of meiosis (cyte I) and nucleus structure of early and late spermatids. Z. Zellforsch. 51, 243–277 (1960).
Ullerich, F.-H.: Achiasmatische Spermatogenese bei der Skorpionsfliege Panorpa (Mecoptera). Chromosoma (Berl.) 12, 215–232 (1961).
Venable, J. H., and R. Coggeshall: A simplified lead citrate stain for use in electron microscopy. J. Cell Biol. 25, 407–408 (1965).
Westergaard, M., and D. von Wettstein: The meiotic cycle in an ascomycete. The effects of ionizing radiations on meiotic systems. Panel Proceedings Series, Int. Atomic Energy Agency, Vienna 1967.
White, M. J. D.: The evolution of the sex chromosomes. I. The XO and X1Y2Y mechanism in praying mantids. J. Genet. 42, 143–172 (1941).
—: Sex chromosomes and meiotic mechanisms in some African and Australian mantids. Chromosoma (Berl.) 16, 521–547 (1965).
Author information
Authors and Affiliations
Additional information
Supported by a University of Melbourne Research Fellowship.
Rights and permissions
About this article
Cite this article
Gassner, G. Synaptinemal complexes in the achiasmatic spermatogenesis of Bolbe nigra Giglio-Tos (Mantoidea). Chromosoma 26, 22–34 (1969). https://doi.org/10.1007/BF00319497
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00319497