Summary
The ultrastructure of the innervation of the anterior cerebral artery of the rat was studied in control animals and in animals after superior cervical ganglionectomy.
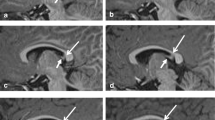
Fluorescence histochemistry shows a periarterial network of intensely fluorescent fibers which are divided into two groups, adventitial and periadventitial. The fluorescence begins to decrease 26 hours after, and completely disappears about 32 hours after, ganglionectomy.
Fine structural changes are first observed 18 hours after ganglionectomy, when the axoplasm of degenerating axons becomes electron dense. This density gradually increases up to about 32 hours. By 32 hours most axons with disintegrating axolemmas become inclusion bodies of the Schwann cells. At this stage, synaptic vesicles can still be distinguished as less dense areas, but the membrane structures of synaptic vesicles and mitochondria are difficult to recognize. The degenerating axons are gradually absorbed and by 38 hours dense, residual bodies are observed in the Schwann cells. Generally speaking, the degeneration occurs first in the adventitial fibers and then in the periadventitial fibers. The transient appearance of small, granular vesicles is noticed in axon terminals about 18 hours after denervation, although very few small, granular vesicles are seen in control tissue or at later stages of degeneration.
Similar content being viewed by others
References
Adinolfi, A. M.: Degenerative changes in the entopeduncular nucleus following lesions of the caudate nucleus: An electron microscopic study. Exp. Neurol. 25, 246–254 (1969).
Birks, R., Katz, B., Miledi, R.: Physiological and structural changes at the amphibian myoneural junction, in the course of nerve degeneration. J. Physiol. (Lond.) 150, 145–168 (1960).
Bondareff, W.: Submicroscopic morphology of granular vesicles in sympathetic nerves of rat pineal body. Z. Zellforsch. 67, 211–218 (1965).
Chorobski, S., Penfield, W.: Cerebral vasodilator nerves and their pathway from the medulla oblongata with observations of the pial and intracerebral vascular plexuses. Arch. Neurol. Psychiat. (Chic.) 28, 1257–1289 (1932).
Dahl, E., Nelson, E.: Electron microscopic observations on human intercranial arteries. II. Innervation. Arch. Neurol. (Chic.) 10, 158–164 (1964).
Duncan, D., Micheletti, G.: Notes on the fine structure of the pineal organ of cats. Tex. Rep. Biol. Med. 24, 576–587 (1966).
Duve, C., de: The lysosome concept. In: Ciba Foundation Symposium Lysosomes (A. V. S. de Reuck and M. P. Cameron, eds.), p. 1–35. London: J. & A. Churchill Ltd. 1963.
—, Pressman, B. C., Gianetto, R., Wattiaux, R., Appelmans, F.: Tissue fractionation studies. 6. Intracellular distribution patterns of enzymes in rat-liver tissue. Biochem. J. 60, 604–617 (1955).
Falck, B.: Observations on the possibilities of the cellular localization of monoamines by a fluorescence method. Acta physiol. scand. 56. Supp. 197, 1–25 (1962).
—, Mchedlishvili, G. I., Owman, Ch.: Histochemical demonstration of adrenergic nerves in cortex-pia of rabbit. Acta pharmacol. (Kbh.) 23, 133–142 (1965).
Farrell, K. E.: Fine structure of nerve fibres in smooth muscle of the vas deferens in normal and reserpinized rats. Nature (Lond.) 217, 279–281 (1968).
Gray, E. G., Guillery, R. W.: Synaptic morphology in the normal and degenerating nervous system. Int. Rev. Cytol. 19, 111–182 (1966).
Hagen, E., Wittkowski, W.: Licht- und elektronenmikroskopische Untersuchungen zur Innervation der Piagefäße. Z. Zellforsch. 95, 429–444 (1969).
Hámori, J., Láng, E., Simon, L.: Experimental degeneration of the preganglionic fibers in the superior cervical ganglion of the cat: An electron microscope study. Z. Zellforsch. 90, 37–52 (1968).
Hökfelt, T., Jonsson, G.: Studies on reaction and binding of monoamines after fixation and processing for electron microscopy with special reference to fixation with potassium permanganate. Histochemie 16, 45–67 (1968).
Hunt, C. C., Nelson, P. G.: Structural and functional changes in the frog sympathetic ganglion following cutting of the presynaptic nerve fibres. J. Physiol. (Lond.) 177, 1–20 (1965).
Iwayama, T., Furness, J. B., Burnstock, G.: Dual adrenergic and cholinergic innervation of the cerebral arteries of the rat: An ultrastructural study. Circulat. Res. 26, 635–646 (1970).
Luft, J. H.: Improvements in epoxy resin embedding methods. J. biophys. biochem. Cytol. 9, 409–414 (1961).
Machado, A. B. M.: Straight OSO4 versus glutaraldehyde-OSO4 in sequence as fixatives for the granular vesicles in sympathetic axons of the rat pineal body. Stain Technol. 42, 293–300 (1967).
Merrillees, N. C. R., Burnstock, G., Holman, M. E.: Correlation on fine structure and physiology of the innervation of smooth muscle in the guinea-pig vas deferens. J. Cell Biol. 19, 529–550 (1963).
Millonig, G.: A modified procedure for lead staining of thin sections. J. biophys. biochem. Cytol. 11, 736–739 (1961).
Nelson, E., Rennels, M.: Neuromuscular contacts in intracranial arteries of the cat. Science 167, 301–302 (1970).
Nielsen, K. C., Owman, C.: Adrenergic innervation of pial arteries related to the circle of Willis in the cat. Brain Res. 6, 773–776 (1967).
Nishida, S., Sears, M.: Fine structural innervation of the dilator muscle of the iris of albino guinea pig studied with permanganate fixation. Exp. Eye Res. 8, 292–296 (1969).
Orden, III, L. S., van, Bensch, K. G., Langer, S. Z., Trendelenberg, U.: Histochemical and fine structural aspects of the onset of denervation supersensitivity in the nictitating membrane of the spinal cat. J. Pharmacol. exp. Ther. 157, 274–283 (1967).
Pease, D. C., Molinari, S.: Electron microscopy of muscular arteries: pial vessels of the cat and monkey. J. Ultrastruct. Res. 3, 447–468 (1960).
Pellegrino de Iraldi, A., Zieher, L. M., de Robertis, E.: Ultrastructure and pharmacological studies of nerve endings in the pineal organ. In: Progress in brain research 10, structure and function of the epiphysis cerebri (J. A. Kappers, J. P. Schadé, eds.), p. 389–422. Amsterdam: Elsevier 1965.
Pluchino, S., Orden, III, L. S., van, Draskóczy, P. R., Langer, S. Z., Trendelenberg, U.: The effect of β-TM 10 on the pharmacological, biochemical and morphological changes induced by denervation of the nictitating membrane of the cat. J. Pharmacol. exp. Ther. 172, 77–90 (1970).
Richardson, K. C.: The fine structure of autonomic nerve endings in smooth muscle of the rat vas deferens. J. Anat. (Lond.) 96, 427–442 (1962).
—: The fine structure of the albino rabbit iris with special reference to the identification of adrenergic and cholinergic nerves and nerve endings in its intrinsic muscles. Amer. J. Anat. 114, 173–205 (1964).
—: Electron microscopic identification of autonomie nerve endings. Nature (Lond.) 210, 756 (1966).
Roth, C. D., Richardson, K. C.: Electron microscopical studies on axonal degeneration in the rat iris following ganglionectomy. Amer. J. Anat. 124, 341–360 (1969).
Saavedra, J. P., Vaccarezza, O. L., Reader, T. A., Pasqualini, E.: Synaptic transmission in the degenerating lateral geniculate nucleus: An ultrastructural and electrophysiological study. Exp. Neurol. 26, 607–620 (1970).
Samarasinghe, D. D.: The innervation of the cerebral arteries in the rat: An electron microscope study. J. Anat. (Lond.) 99, 815–828 (1965).
Sato, S.: An electron microscopic study on the innervation of the intracranial artery of the rat. Amer. J. Anat. 118, 873–890 (1966).
Straus, W.: Cytochemical observations on the relationship between lysosomes and phagosomes in kidney and liver by combined staining for acid phosphatase and intravenously injected horseradish peroxidase. J. Cell Biol. 20, 497–507 (1964a).
—: Occurrence of phagosomes and phago-lysosomes in different segments of the nephron in relation to the reabsorption, transport, digestion, and extrusion of intravenously injected horseradish peroxidase. J. Cell Biol. 21, 295–308 (1964b).
Szentágothai, J., Hámori, J., Tömböl, Th.: Degenration and electron microscope analysis of the synaptic glomeruli in the lateral geniculate body. Exp. Brain Res. 2, 283–301 (1966).
Taxi, J.: Contribution à l'étude des connexions des neurones moteurs du système nerveux autonome. Ann. Sci. Nat. Zool., XII sèr. 7, 413–671 (1965).
Tranzer, J. P., Thoenen, H.: Significance of “empty vesicles” in postganglionic sympathetic nerve terminals. Experientia (Basel) 23, 123–124 (1967).
Walberg, F.: An electron microscopic study of terminal degeneration in the inferior olive of the cat. J. comp. Neurol. 125, 205–222 (1965).
Watson, M. L.: Staining of tissue sections for electron microscopy with heavy metals. J. biophys. biochem. Cytol. 4, 475–478 (1958).
Webster, H. de F.: Transient, focal accumulation of axonal mitochondria during the early stages of Wallerian degeneration. J. Cell Biol. 12, 361–383 (1962).
Wolfe, D. E., Potter, L. T., Richardson, K. C., Axelrod, J.: Localizating tritiated norepinephrine in sympathetic axons by electron microscopic autoradiography. Science 138, 440–441 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Iwayama, T. Ultrastructural changes in the nerves innervating the cerebral artery after sympathectomy. Z. Zellforsch. 109, 465–480 (1970). https://doi.org/10.1007/BF00343962
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00343962