Abstract
The partitioning of Cr and Al between coexisting spinel and clinopyroxene and the dependence of spinel-cpxgarnet equilibria on Cr/Al ratio have been investigated by a combination of phase equilibrium experiments, high temperature solution calorimetry and thermodynamic calculations.
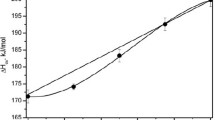
The exchange equilibrium: \(\begin{gathered} {\raise0.7ex\hbox{$1$} \!\mathord{\left/ {\vphantom {1 {2 }}}\right.\kern-\nulldelimiterspace}\!\lower0.7ex\hbox{${2 }$}}MgAl_2 O_4 + NaCrSi_2 O_6 = {\raise0.7ex\hbox{$1$} \!\mathord{\left/ {\vphantom {1 {2 }}}\right.\kern-\nulldelimiterspace}\!\lower0.7ex\hbox{${2 }$}}MgCr_2 O_4 + NaAlSi_2 O_6 \hfill \\ spinel clinopyroxene spinel clinopyroxene \hfill \\ \end{gathered} \) has a measured enthalpy change for pure phases of −2,100±500 cal at 970 K and 1 atm. Experimental reversals of Cr-Al partitioning between the spinel and clinopyroxene phases yield the following partitioning relationship: \(X\) where X ji refers to atomic fraction of i in the octahedral sites of phase j. The compositional dependence of partitioning implies that Al-Cr mixing in spinel is nonideal with, on the symmetrical model, a W SpCr-Al of 2,700±500 cal/gm. atom. In contrast, aluminum-chromium mixing in clinopyroxene is close to ideal.
The measured stability field of knorringite (Mg3Cr2Si2O12) and mixing properties of garnet have been used in conjunction with our experimental data to calculate the influence of Cr/Al ratio on the important reaction: orthopyroxene+clinopyroxene+spinel=olivine+garnet
The stability field of spinel lherzolite increases by about 2.8 Kb for every increase of 0.1 in Cr/(Cr+Al) ratio up to Cr/(Cr+Al) of 0.7. The calculated stabilization is in very good agreement with the experimental results of O'Neill (1981). The partitioning relationships are such that, at the low ratios of Cr/Al (∼0.07) of primitive lherzolite, clinopyroxene buffers spinel composition and sharpens the spinelgarnet reaction interval from 10 Kb (little or no clinopyroxene) down to about 2 Kb in pyroxene-rich “pyrolite.”
Similar content being viewed by others
References
Adams GE, Bishop FC (1982) Experimental investigation of Ca-Mg exchange between olivine, orthopyroxene, and clinopyroxene: potential for geobarometry. Earth Planet Sci Lett 57:241–250
Boyd FR (1973) A pyroxene geothermometer. Geochim Cosmochim Acta 37:2533–2546
Burnham CW (1962) Lattice constant refinement. Carnegie Inst Washington Yearb 61:132–135
Carswell DA, Clarke DB, Mitchell RH (1979) The petrology and geochemistry of ultramafic nodules from pipe 200, northern Lesotho. In: Boyd FR, Meyer HOA (eds) The Mantle Sample. Am Geophys Union, Washington, pp 127–144
Coleman RG (1961) Jadeite deposits of the Clear Creek area, New Idria District, San Benito County, California. J Petrol 2:209–247
Chistensen NI (1982) Seismic velocities. In: RS Carmichael (ed) Handbook of physical properties of rocks. C.R.C. Press, Boca Raton, Florida, 1–228 pp
Clark JR, Appleman DE, Papike JJ (1969) Crystal-chemical characterization of clinopyroxene based on eight new structure refinements. Mineral Soc Am, Spec Pap 2:31–50
Engi M (1983) Equilibria involving Al-Cr spinel: Mg-Fe exchange with olivine. Experiments, thermodynamic analysis and consequences for geothermometry. Amer J Sci 283-A:29–71
Fyfe WS, Turner FJ, Verhoogen J (1958) Metamorphic reactions and metamorphic facies. Geol Soc Am Mem 72: Waverley Press, Baltimore, 259 pp
Gasparik T (1984) Two-pyroxene thermobarometry with new experimental data in the system CaO-MgO-Al2O3-SiO2. Contrib Mineral Petrol 87:87–97
Gasparik T Newton RC (1984) The reversed alumina contents of orthopyroxene in equilibrium with spinel and forsterite in the system MgO-Al2O3-SiO2. Contrib Mineral Petrol 85:186–196
Green DH, Ringwood AE (1967) The stability fields of aluminous pyroxene periodotite and garnet peridotite and their relevance in upper mantle structure. Earth Planet Sci Lett 3:151–160
Hervig RL, Smith JV (1982) Temperature-dependent distribution of Cr between olivine and pyroxenes in lherzolite xenoliths. Contrib Mineral Petrol 81:184–189
Irifune T, Ohtani E, Kumazawa M (1982) Stability field of knorringite Mg3Cr2Si3O12 at high pressure and its implication to the occurrence of Cr-rich pyrope in the upper mantle. Phys Earth Planet Int 27:263–272
Jenkins DM, Newton RC (1979) Experimental determination of the spinel peridotite to garnet peridotite inversion at 900° C and 1,000° C in the CaO-MgO-Al2O3-SiO2 system, and at 900° C with natural garnet and olivine. Contrib Mineral Petrol 68:407–419
LADLE study group (1983) A lithospheric seismic refraction profile in the western North Atlantic Ocean. Geophys JR Astr Soc 75:23–61
Lindsley DH (1980) Pyroxene thermometry. Am Mineral 68:477–493
Mattioli GS, Bishop FC (1984) Experimental determination of the chromium-aluminum mixing parameter in garnet. Geochim Cosmochim Acta 48:1367–1371
Nixon PH, Boyd FR (1973) Petrogenesis of the granular and sheared ultrabasic nodule suite in kimberlites. In: Nixon PH (ed) Lesotho Kimberlites, Lesotho National Development Corp, pp 48–56
O'Hara MJ, Richardson SW, Wilson G (1971) Garnet-peridotite stability and occurrence in crust and mantle. Contrib Mineral Petrol 32:48–67
Oka Y, Steinke P, Chatterjee ND (1984) Thermodynamic mixing properties of Mg(Al, Cr)2O4 spinel crystalline solution at high temperatures and pressures. Contrib Mineral Petrol 87:196–204
O'Neill HStC (1981) The transition between spinel lherzolite and garnet lherzolite, and its use as a geobarometer. Contrib Mineral Petrol 77:185–194
O'Neill HStC, Navrotsky A (1984) Cation distributions and thermodynamic properties of binary spinel solid solutions. Am Mineral 69:733–753
Popp RK, Gilbert MC (1972) Stability of acmite-jadeite pyroxenes at low pressure. Am Mineral 57:1210–1231
Ringwood AE (1975) Composition and Petrology of the Earth's Mantle. McGraw-Hill, New York
Robie RA, Hemingway BS, Fisher JR (1978) Thermodynamic properties of minerals and related substances at 298.15 K and 1 bar (105Pa) pressure and at higher temperatures. US Geol Surv Bull 1452:456
Roeder PL, Campbell IH, Jamieson HE (1979) A reevaluation of the olivine-spinel geothermometer. Contrib Mineral Petrol 68:325–334
Sachtleben TH, Seck HA (1981) Chemical control of Al-solubility in orthopyroxene and its implications on pyroxene geothermometry. Contrib Mineral Petrol 78:157–165
Sack RO (1982) Spinels as petrogenetic indicators: activity-composition relations at low pressure. Contrib Mineral Petrol 71:169–186
Shimamura H, Asada T (1976) Apparent velocity measurements on oceanic lithosphere. Phys Earth Planet Intern 13:15–22
Vickerman JC (1971) Magnetic properties of chromium ions in oxide matrixes. Part 2. MgCr2O4-MgAl2O4 solid solutions. Trans Faraday Soc 67:665–673
Warshaw I, Keith ML (1954) Solid solution and chromium oxide loss in part of the system MgO-Al2O3-Cr2O3-SiO2. J Am Ceram Soc 37:161–168
Wood BJ, Banno S (1973) Garnet-orthopyroxene and orthopyroxene-clinopyroxene relationships in simple and complex systems. Contrib Mineral Petrol 42:109–124
Wood BJ, Holland TJB, Newton RC, Kleppa OJ (1980) Thermochemistry of jadeite-diopside pyroxenes. Geochim Cosmochim Acta 44:933–941
Wood BJ, Holloway JR (1982) Theoretical prediction of phase relationships in planetary mantles. J Geophys Res 87:A19-A30
Wood BJ, Holloway JR (1984) A thermodynamic model for subsolidus equilibria in the system CaO-MgO-Al2O3-SiO2. Geochim Cosmochim Acta 48:159–176
Wood BJ, Kleppa OJ (1980) Thermochemistry of forsterite-fayalite olivine solutions. Geochim Cosmochim Acta 45:569–581
Wood BJ, Kleppa OJ (1984) Chromium-aluminum mixing in garnet: a thermochemical study. Geochim Cosmochim Acta 48:1373–1375
Wood BJ, Nicholls J (1978) The thermodynamic properties of reciprocal solid solutions. Contrib Mineral Petrol 66:389–400
Wood BJ, Yuen DA (1983) The role of lithospheric phase transitions on seafloor flattening at old ages. Earth Planet Sci Lett 66:303–314
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Webb, S.A.C., Wood, B.J. Spinel-pyroxene-garnet relationships and their dependence on Cr/Al ratio. Contrib Mineral and Petrol 92, 471–480 (1986). https://doi.org/10.1007/BF00374429
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374429