Abstract
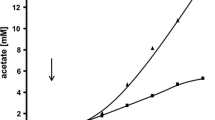
When Acetobacterium woodii was co-cultured in continuous or in stationary culture with Methanobacterium strain AZ, fructose instead of being converted to 3 mol of acetate was converted to 2 mol of acetate and 1 mol each of carbon dioxide and methane, showing that interspecies hydrogen transfer occurred. In continous culture the organisms formed a close physical association in clumps; the doubling time for each organism was 6h at 33°C. Methane mainly was derived from carbon positions 3 and 4 of the sugar, but other carbons also yielded methane; this was shown to be due to carbon dioxide-acetate exchange reactions by A. woodii in a manner similar to that carried out by Clostridium thermoaceticum. Four other methanogens, Methanobacterium M.o.H. and M.o.H. G, Methanobacterium formicicum, and Methanosarcina barkeri (not acetate-adapted) also produced similar results, when co-cultured with A. woodii.
Similar content being viewed by others
References
Andreesen, J. R., Schaupp, A., Neurauter, C., Brown, A., Ljungdahl, L. G.: Fermentation of glucose, fructose and xylose by Clostridium thermoaceticum: Effects of metals on growth yield, enzymes, and the synthesis of acetate from CO2. J. Bacteriol. 114, 743–751 (1973)
Balch, W. E., Schoberth, S., Tanner, R. S., Wolfe, R. S.: Acetobacterium a new genus of hydrogen-oxidizing, carbon dioxide-reducing, anaerobic bacteria. Int. J. Syst. Bacteriol. 27, 355–361 (1977)
Balch, W. E., Wolfe, R. S.: New approach to the cultivation of methanogenic bacteria: 2-mercaptoethanesulfonic acid (HS-CoM)-dependent growth of Methanobacterium ruminantium in a pressurized atmosphere. Appl. Environm. Microbiol. 32, 781–791 (1976)
Balch, W. E., Wolfe, R. S.: Specificity and biological distribution of coenzyme M (2-mercaptoethanesulfonic acid). J. Bacteriol. 137, 256–263 (1979)
Barker, H. A., Kamen, M. D.: Carbon dioxide utilization in the synthesis of acetate by Clostridium thermoaceticum. Proc. Natl. Acad. Sci. U.S.A. 31, 219–225 (1945)
Bray, G. A.: A simple efficient liquid scintillator for counting aqueous solutions in a liquid scintillation counter. Anal. Biochem. 1, 279–285 (1960)
Bryant, M. P.: Microbiology of anaerobic degradation and methanogenesis with special reference to sewage. In: Microbial energy conversion, (H. G. Schlegel, J. Barnea, eds.), pp. 107–117. Göttingen: Erich Goltze KG, 1976
Bryant, M. P., Campbell, L. L., Reddy, C. A., Crabill, M. R.: Growth of Desulfovibrio in lactate or ethanol media low in sulfate in association with H2-utilizing methanogenic bacteria. Appl. Environm. Microbiol. 33, 1162–1169 (1977)
Chen, M., Wolin, M. J.: Influence of CH4 production by Methanobacterium ruminantium on the fermentation of glucose and lactate by Selenomonas ruminantium. Appl. Environm. Microbiol. 34, 756–759 (1977)
Cooney, C. L., Ackerman, R. A.: Thermophilic anaerobic digestion of cellulosic waste. J. Appl. Microbiol. 2, 65–72 (1975)
Fontaine, F. E., Peterson, W. H., McCoy, E., Johnson, M. J., Ritter, G. J.: A new type of glucose fermentation by Clostridium thermoaceticum n. sp. J Bacteriol. 43, 701–715 (1942)
Ferry, J. G., Wolfe, R. S.: Anaerobic degradation of benzoate to methane by a microbial consortium. Arch. Microbiol. 107, 33–40 (1976)
Jeris, J. S., McCarty, P. L.: Biochemistry of methane fermentation using 14C tracers. J. Water Poll. Contr. Fed. 37, 178–192 (1965)
Kaspar, H. F., wuhrmann, K.: Kinetic parameters and relative turnovers of some important catabolic reactions in digesting sludge. Appl. Environm. Microbiol. 36, 1–7 (1978)
Latham, M. J., Wolin, M. J.: Fermentation of cellulose by Ruminococcus flavefaciens in the presence and absence of Methanobacterium ruminantium. Appl. Environm. Microbiol. 34, 297–301 (1977)
McCarty, P. L., Young, L. Y., Gossett, J. M., Stuckey, D. C., Healy, J. B., Jr.: Heat treatment for increasing methane yields from organic materials. In: Microbial energy conversion, (H. G. Schlegel and J. Barnea, eds.), pp. 179–199. Göttingen: Erich Goltze KG 1976
Miller, G. L.: Use of dinitrosalicylic acid reagent for determination of reducing sugars. Anal. Chem. 31, 426–428 (1959)
Rose, I. A., Grunberg-Manago, M., Korey, S. R., Ochoa, S.: Enzymatic phosphorylation of acetate. J. Biol. Chem. 211, 905–931 (1954)
Schulman, M., Ghambeer, R. K., Ljungdahl, L., Wood, H. G.: Total synthesis of acetate from CO2. VII. Evidence with Clostridium thermoaceticum that the carboxyl of acetate is derived from the carboxyl of pyruvate by transcarboxylation and not by fixation of CO2. J. Biol. Chem. 248, 6255–6261 (1973)
Smith, P. H., Mah, R. A.: Kinetics of acetate metabolism during sludge digestion. Appl. Microbiol. 14, 368–371 (1966)
Smith, M. R., Mah, R. A.: Growth and methanogenesis by Methanosarcina strain 227 on acetate and methanol. Appl. Environm. Microbiol. 36, 870–879 (1978)
Varel, V. H., Isaacson, H. R., Bryant, M. P.: Thermophilic methane production from cattle wastes. Appl. Environm. Microbiol. 33, 299–307 (1977)
Weimer, P. J., Zeikus, J. G.: Fermentation of cellulose and cellobiose by Clostridium thermocellum in the absence and presence of Methanobacterium thermoautotrophicum. Appl. Environm. Microbiol. 33, 289–297 (1977)
Winter, J., Wolfe, R. S.: Complete degradation of carbohydrates to CO2 and methane by syntrophic cultures of Acetobacterium woodii and Methanosarcina barkeri. Arch. Microbiol. 121, 97–102 (1979)
Wood, H. G.: A study of carbon dioxide fixation by mass determination of the types of 13C-acetate. J. Biol. Chem. 194, 905–931 (1952)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Winter, J.U., Wolfe, R.S. Methane formation from fructose by syntrophic associations of Acetobacterium woodii and different strains of methanogens. Arch. Microbiol. 124, 73–79 (1980). https://doi.org/10.1007/BF00407031
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00407031