Summary
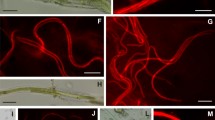
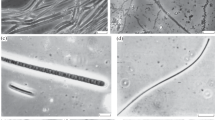
Chloroflexis, a gliding, filamentous, photosynthetic bacterium, is present in the stratified algal-bacterial mats which occur in the 50°–70°C temperature range of alkaline hot spring effluents. The organism is in association with the alga in the upper, algal layer, and also forms thick, orange mats beneath the algal layer. Natural populations of Chloroflexis from these mats demonstrated light-stimulated uptake of some 14C-labelled organic compounds. Photosynthetic 14CO2 fixation by natural samples of Chloroflexis was investigated with respect to temperature, light intensity and mat depth. Bacterial photosynthesis was determined in samples in which algae were present by use of the inhibitor 3-(3,4-dichlorophenyl)-1,1-dimethylurea (DCMU). Bacterial photosynthesis was maximal at depths down to about 3 mm and then decreased rapidly to very low levels at greater depths. The greatest amounts of bacteriochlorophyll pigments were also concentrated in the top 3–4 mm of the mat. The optimum light intensity for bacterial photosynthesis (about 400 ft-c) was considerably lower than the normal summer light intensity at the surface of the mat (5000-8000 ft-c).
The temperature optima for photosynthesis by the bacterial component of natural mat samples from several sites of different temperatures in a hot spring thermal gradient were determined. Temperature optima approximated the environmental temperatures, indicative of the occurrence of strains of Chloroflexis adapted to different temperatures. Although bacterial standing crop was greatest in the temperature range 50°–55°C, maximum photosynthetic efficiency was observed at about 45°C. Sulfide was stimulatory to photosynthetic 14CO2 fixation by naturally occurring populations of Chloroflexis under field conditions. These data are consistent with the hypothesis that Chloroflexis may utilize sulfide as an electron donor for photosynthetic CO2 reduction. However, it is also likely that Chloroflexis grows photoheterotrophically in these mats, obtaining organic compounds from algal excretory products.
Similar content being viewed by others
References
Bauld, J.: Algal-bacterial interactions in alkaline hot spring effluents. Ph. D. dissertation, University of Wisconsin (1973)
Bauld, J., Brock, T. D.: Algal excretion and bacterial assimilation in mats of alkaline hot spring effluents. J. Phycol. (to be submitted for publication)
Bishop, N. I.: The influence of the herbicide, DCMU, on the oxygen-evolving system of photosynthesis. Biochim. biophys. Acta (Amst.) 27, 205–206 (1958)
Bott, T. L., Brock, T. D.: Bacterial growth rates above 90°C in Yellowstone hot springs. Science 164, 1411–1412 (1969)
Brock, M. L., Wiegert, R. G., Brock, T. D.: Feeding by Paracoenia and Ephydra (Dipteria: Ephydridae) on the microorganisms of hot springs. Ecology 50, 192–200 (1969)
Brock, T. D.: Relationship between standing crop and primary productivity along a hot spring thermal gradient. Ecology 48, 566–571 (1967a)
Brock, T. D.: Microorganisms adapted to high temperatures. Nature (Lond.) 214, 882–885 (1967b)
Brock, T. D.: Taxonomic confusion concerning certain filamentous blue-green algae. J. Phycol. 4, 178–179 (1968)
Brock, T. D.: Vertical zonation in hot spring algal mats. Phycologia 8, 201–205 (1969)
Brock, T. D.: High temperature systems. Ann. Rev. Ecol. Syst. 1, 192–220 (1970)
Brock, T. D., Brock, M. L.: Temperature optima for algal development in Yellowstone and Iceland hot springs. Nature (Lond.) 209, 733–734 (1966)
Brock, T. D., Brock, M. L.: The measurement of chlorophyll, primary productivity, photophosphorylation, and macromolecules in benthic algal mats. Limnol. Oceanogr. 12, 600–605 (1967)
Brock, T. D., Brock, M. L.: Relationship between environmental temperature and optimum temperature of bacteria along a hot spring thermal gradient. J. appl. Bact. 31, 54–58 (1968)
Brock, T. D., Brock, M. L.: Effect of light intensity on photosynthesis by thermal algae adapted to natural and reduced sunlight. Limnol. Oceanogr. 14, 334–341 (1969)
Brock, T. D., Brock, M. L.: Temperature optimum of non-sulphur bacteria from a spring at 90°C. Nature (Lond.) 233, 494–495 (1971)
Castenholz, R. W.: Thermophilic blue-green algae and the thermal environment. Bact. Rev. 33, 476–504 (1969)
Cohen-Bazire, G., Sistrom, W. R.: The procaryotic photosynthetic apparatus, pp. 313–341. In: L. P. Vernon and G. R. Seely, Eds.: The chlorophylls. New York: Academic Press 1966
Copeland, J. J.: Yellowstone thermal Myxophyceae. Ann. N.Y. Acad. Sci. 36, 1–229 (1936)
Goodwin, T. W.: The nature and distribution of carotenoids in some blue-green algae. J. gen. Microbiol. 17, 467–473 (1957)
Halfen, L. N., Pierson, B. K., Francis, G. W.: Carotenoids of a gliding organism containing bacteriochlorophylls. Arch. Mikrobiol. 82, 240–246 (1972)
Lascelles, J.: The formation of ribulose 1:5-diphosphate carboxylase by growing cultures of Athiorhodaceae. J. gen. Microbiol. 23, 499–510 (1960)
Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J.: Protein measurement with the Folin phenol reagent. J. biol. Chem. 193, 265–275 (1951)
Meeks, J. C., Castenholz, R. W.: Growth and photosynthesis in an extreme thermophile Synechococcus lividus (Cyanophyta). Arch. Mikrobiol. 78, 25–41 (1971)
Oelze, J., Weaver, P.: The adjustment of photosynthetically grown cells of Rhodospirillum rubrum to aerobic light conditions. Arch. Mikrobiol. 79, 108–121 (1971)
Peary, J. A., Castenholz, R. W.: Temperature strains of a thermophilic blue-green alga. Nature (Lond.) 202, 720–721 (1964)
Pfennig, N.: Photosynthetic bacteria. Ann. Rev. Microbiol. 21, 285–324 (1967)
Pierson, B. K., Castenholz, R. W.: Bacteriochlorophylls in gliding filamentous prokaryotes from hot springs. Nature New Biol. 233, 25–27 (1971)
Schmidt, K., Schiburr, R.: Die Carotinoide der grünen Schwefelbakterien: Carotinoidzusammensetzung in 18 Stämmen. Arch. Mikrobiol. 74, 350–355 (1970)
Sheridan, R. P., Castenholz, R. W.: Production of hydrogen sulphide by a thermophilic blue-green alga. Nature (Lond.) 217, 1063–1064 (1968)
Sistrom, W. R., Clayton, R. K.: Studies on a mutant of Rhodopseudomonas spheroides unable to grow photosynthetically. Biochim. biophys. Acta (Amst.) 88, 61–73 (1964)
Smith, J. H. C., Benitez, A.: Chlorophylls; analysis in plant materials, pp. 142–196. In: K. Peach and M. V. Tracey, Eds.: Moderne Methoden der Pflanzenanalyse, Vol. IV. Berlin-Göttingen-Heidelberg: Springer 1955
Stanier, R. Y., Smith, J. H. C.: The chlorophylls of green bacteria. Biochim. biophys. Acta (Amst.) 41, 478–484 (1960)
Uffen, R. L., Wolfe, R. S.: Anaerobic growth of purple nonsulfur bacteria under dark conditions. J. Bact. 104, 462–472 (1970).
Walter, M. R., Bauld, J., Brock, T. D.: Siliceous algal and bacterial stromatolites in hot spring and geyser effluents of Yellowstone National Park. Science 178, 402–405 (1972)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bauld, J., Brock, T.D. Ecological studies of Chloroflexis, a gliding photosynthetic bacterium. Archiv. Mikrobiol. 92, 267–284 (1973). https://doi.org/10.1007/BF00409281
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00409281