Summary
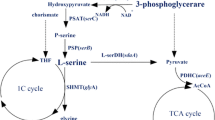
The effects of a pleiotropic mutation ssd are described. This mutation results in decreased efficiency in the use of glucose and fructose as carbon source, inability to use succinate or to grow anaerobically, an alteration in the activity of enzymes responsible for the synthesis and degradation of L-serine, increased resistance to certain antibiotics, and a deficiency in proline transport. This mutation resembles various previously described mutations throught to affect ‘energy coupling factor’ and is located in the same region of the chromosome. While the gene product affected by this mutation is still unidentified, it is clear that L-serine metabolism cannot be understood merely in terms of providing L-serine and its derivatives.
Similar content being viewed by others
References
Bragg PD (1979) ATPase reconstitution of an energy-linked reaction (reduced pyridine nucleotide transhydrogenation) in fractionated E. coli membranes with purified ATPase. In Fleischer S, Packer L (eds) Methods in Enzymology vol 55, Part F. Academic Press New York, pp 787–800
Cox GB, Gibson F (1974) Studies on electron transport and energy-linked reactions using mutants of Escherichia coli. Biochim Biophys Acta 346:1–25
Hasan SM, Tsuchiya T, Rosen BP (1978) Energy transduction in Escherichia coli: Physiological and Biochemical Effects of Mutation in the uncB Locus. J Bacteriol 133:108–113
Isenberg S, Newman EB (1974) Studies on L-serine deaminase in Escherichia coli K12. J Bacteriol 118:53–58
Lieberman MA, Hong J-S (1974) A mutant of Escherichia coli defective in the coupling of metabolic energy to active transport. Proc Natl Acad Sci USA 71:4395–4399
McKitrick JC, Pizer LI (1980) Regulation of Phosphoglycerate Dehydrogenase Levels and Effect on Serine Synthesis in Escherichia coli K12. J Bacteriol 141:235–245
Morris JF, Newman EB (1980) Map Location of the ssd mutation in Escherichia coli K-12. J Bacteriol (in the press)
Newman EB, Adley T, Fraser J, Potter R, Kapoor V (1976) The conversion of leucine to alpha-ketoisocaproic acid and its metabolic consequences for Escherichia coli K12. Can J Microbiol 22:922–928
Pardee AB, Prestidge LS (1975) Induced formation of serine and threonine deaminase by Escherichia coli. J Bacteriol 70:667–674
Plate CA (1976) Mutant of Escherichia coli Defective in Response to Colicin K and in Active Transport. J Bacteriol 125:467–474
Simoni RD, Postma PW (1975) Energetics of active bacterial transport. Biochemistry 44:523–544
Rider CC, Taylor CB (1974) Enolase isoenzymes in rat tissues: electrophoretic, chromatographic, immunologic and kinetic properties. Biochim Biophys Acta 365:285–300
Soffer RL, Savage M (1974) A mutant of Escherichia coli defective in leucyl, phenylalanyl-tRNA protein transferase. Proc Natl Acad Sci USA 71:1004–1007
Tam A, Herrington MB, Kapoor V, Newman EB (1978) A single mutation affects L-serine deaminase, L-leucyl, L-phenylalanyl-tRNA protein transferase, and proline oxidase activity in Escherichia coli K12. J Bacteriol 135:1154–1155
Thorbjarnardottor SH, Magnusdottor RA, Eggertsson G, Kagan SA, Andersson OS (1978) Mutations determining generalized resistance to aminoglycoside antibiotics in Escherichia coli. Mol Gen Genet 161:89–98
Warburg O, Christian W (1942) Isolierung und kristallisation des garungsferments enolase. Biochem 31:384–421
Winicov I, Pizer L (1964) The mechanism of end product inhibition of serine biosynthesis. J Biol Chem 249:1348–1355
Author information
Authors and Affiliations
Additional information
Communicated by G. O'Donovan
Rights and permissions
About this article
Cite this article
Newman, E.B., Morris, J.F., Walker, C. et al. A mutation affecting L-serine and energy metabolism in E. coli K12. Molec. Gen. Genet. 182, 143–147 (1981). https://doi.org/10.1007/BF00422781
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00422781