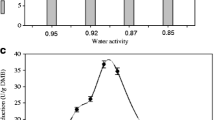
Abstract
Sixty-four wheat samples from Spanish flour factories were screened for patulin and patulin-producing moulds. None of them was found to contain any patulin, whereas samples experimentally contaminated with this toxin proved it to be highly unstable. On the other hand, Penicillium griseofulvum was the only in vitro patulin-producing species found (19 samples). Mould growth in the samples was investigated by using yeast-sucrose medium (YES) and high-performance liquid chromatography (HPLC) to measure the amounts of toxin produced during 40 day's incubation at 20 and 28°C. The highest yield rate of patulin was obtained between the 20th and 30th day of incubation; such a rate, however, was very low throughout the vigorous growth phase, during the first 20 days of incubation. The more appropriate temperature for incubation and patulin production was 28 °C. We also investigated the influence of other incubation conditions in the yield and found stationary dark cultures to be more efficient that shaken or fermentation cultures in YES medium. The best patulin yield achieved was 11.9 mg in the culture broth and 6.3 mg in the mycelium from 100 ml of medium.
Similar content being viewed by others
References
Smith JE, Moss MO. Mycotoxins, formation, analysis and significance. Chichester: John Wiley & Sons, 1985: 46.
Cole RJ, Cox RH. Handbook of toxic fungal metabolites. London: Academic Press, 1981.
Steiman R, Seigle-Murandi F, Sage L, Krovobok S. Production of patulin by micromycetes. Mycopathologia 1989; 109: 129–34.
Stott WT, Bullermann LB. Patulin: a mycotoxin of potential concern in foods. J Milk Food Technol 1975; 38: 695–705.
Laue W, Hollstein E, Donath R, Zapff G. Studies into occurrence and importance of molds and mycotoxins in collected fodder. Monatsh Veterinaermed 1988; 43: 829–32.
Purchio A, Gambale W, Paula CR, Barbieri W, Sabino M, Meireles MCA, Iida S. Survey of mycotoxins (aflatoxins, patulin, ochratoxin and sterigmatocystin) and correlate fungi in dairy cattle feedstuffs. Rev Microbiol 1988; 19: 172–6.
Brackett RE, Marth EH. Stability of patulin at pH 6.0–8.0 and 25 °C. Z Lebensm Unters Forsch 1979; 169: 92–4.
Brackett RE, Marth EH. Ascorbic acid and ascorbate cause disappearance of patulin from buffer solution and apple juice. J Food Protect 1979; 42: 864–6.
Burroughs LF. Stability of patulin to sulphur dioxide and to yeast fermentation. J Assoc Off Anal Chem 1977; 60: 100–3.
Pohland AE, Allen R. Stability studies with patulin. J Assoc Off Anal Chem 1970; 53: 688–91.
Ciegler A, Beckwith AC, Jackson LK. Reaction of patulin with amino acids. [Abstract]. Ann Meet Amer Soc Microbiol 1975; 75: 199.
Ciegler A, Beckwith AC, Jackson LK. Teratogenicity of patulin and patulin adducts formed with cysteine. Appl Environ Microbiol 1976; 31: 664–7.
Lieu FY, Bullerman LB. Binding of patulin and penicillic acid to glutathione and cysteine and toxicity of the resulting adducts. Milchewissenschaft 1978; 33: 16–20.
Lindroth S, Wright A. Comparison of the toxicity of patulin adducts formed with cysteine in Salmonella test systems. Mutat Res 1978; 58: 211–15.
Harwig J, Scott P, Kennedy B, Chen Y. Disappearance of patulin from apple juice fermented by Saccharomyces spp. Can Inst Food Sci Technol J 1973; 6: 45–6.
Lipovska T, Goszcs H. Januszwski T, Kubacki SJ, Effects of alcoholic fermentation on patulin stability. Przemyls Fermentacyjny i Owocowo-Warzywny 1987; 31: 8–10.
Ough CS, Corison CA. Measurement of patulin in grapes and wines. J Food Sci 1980; 45: 476–8.
Harwig J, Blanchfield BJ, Jarvis G. Effect of water activity on disappearance of patulin and citrinin from grains. J Food Sci 1977; 42: 1225–8.
Northolt MD, Van Egmond HP, Paulsch WE. Patulin production by some fungal species in relation to water activity and temperature. J Food Protect 1978; 41: 885–90.
Roland JO, Benchat LR. Influence of temperature and water activity on growth and patulin production by Byssochlamys nivea in apple juice. Appl Environ Microbiol 1984; 47: 205–7.
Zegota H, Zegota A, Bachman S. Effect of irradiation on the patulin content and chemical composition of apple juice concentrate. Z Lebensm Unters Forsch 1988; 187: 235–8.
Zegota H, Zegota A, Bachman S. Effect of irradiation and storage on patulin disappearance and some chemical constituents of apple juice concentrate. Z Lebensm Unters Forsch 1988; 187: 321–4.
Zegota H, Pietka M. Elimination of the toxic effects of patulin of Bacillus subtilis and Bacillus megaterium by irradiation. Z Lebensm Unters Forsch 1989; 188: 348–50.
Jiménez M, Mateo R, Querol A, Mateo JJ, Hernandez E. Differentiation of Penicillium griseofulvum Dierckx isolates by enzyme assays and by patulin and griseofulvin analyses. Appl Environ Microbiol 1990; 56: 3718–22.
Pohland AE, Allen R. Analysis and chemical confirmation of patulin in grains. J Assoc Off Anal Chem 1970; 53:686–7.
Scott PM, Kennedy BPC. Improved method for the thin layer Chromatographic determination of patulin in apple juice. J Assoc Off Anal Chem 1973; 56: 813–6.
Jiménez M, Sanchis V, Santamarina P, Hernández E. Penicillium in pre-harvest corn from Valencia (Spain), I: Influence of different factors on the contamination. Mycopathologia 1985; 92: 53–7.
Pitt JI. The genus Penicillium and its teleomorphic states Eupenicillium and Talaromyces. London: Academic Press, 1979.
Raper KB, Fennell DI. The genus Aspergillus. Baltimore, MD: Williams and Wilkins, 1965.
Jiménez M, Sanchis V, Mateo R, Hernández E. Detection and quantification of patulin and griseofulvin by high pressure liquid chromatography in different strains of Penicillium griseofulvum Dierckx. Mycotox Res 1988; 4: 59–66.
Scott PM, Somers E. Stability of patulin and penicillic acid in fruit juices and flour. J Agric Food Chem 1968; 16: 483–5.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jiménez, M., Mateo, R., Mateo, J.J. et al. Effect of the incubation conditions on the production of patulin by Penicillium griseofulvum isolated from wheat. Mycopathologia 115, 163–168 (1991). https://doi.org/10.1007/BF00462220
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00462220