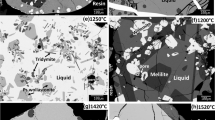
Abstract
The phase relationships over a wide range of temperature and compositions in the ZrO2-CeO2 system have been reinvestigated. From DTA results, thermal expansion measurements andK IC determinations it was established that additions of CeO2 to ZrO2 decreases the monoclinic to tetragonal ZrO2 transition temperature, from 990 ° C to 150 ∓ 50 ° C, and an invariant eutectoid point at approximately 15 mol% CeO2 exists. The extent of the different single- and two-phase fields were determined with precise lattice parameter measurements on quenched samples. Evidence for the existence of a binary compound Ce2Zr3O10 (ø-phase) was obtained by X-ray diffraction. The ø-phase was stable below approximately 800 ° C, above which it decomposes into tetragonal zirconia + fluorite ceria solid solutions. Taking into account the polymorphic tetragonal-cubic transition and the narrowness of the two-phase tetragonal zirconia + fluorite ceria field above 2000 ° C, the existence of a new invariant eutectoid point was assumed, in which the metastable fluorite zirconia solid solution decomposes into tetragonal zirconia + fluorite ceria solid solutions. From the results obtained, the phase diagram also incorporates a eutectic point located at approximately 2300 ° C and 24 mol % CeO2.
Similar content being viewed by others
References
K. Tsukuma,Amer. Ceram. Soc. Bull. 65 (1986) 1386.
K. Tsukuma andM. Shimada,J. Mater. Sci. 20 (1985) 1178.
J. G. Duh andH. T. Dai,J. Amer. Ceram. Soc. 71 (1988) 813.
S. F. Palguev andZ. S. Volchenkova,Russ. J. Phys. Chem. 34 (1960) 211.
F. F. Lange, in “Research of Microstructurally Developed Toughening Mechanisms in Ceramics”, Rockwell International Technical Report no. 12, ONR-N00014-77-C-0441, March 1971, p. 19.
R. C. Garvie, in “High Temperature Oxides”, edited by A. M. Alper (Academic Press, New York, 1970) p. 117.
A. Rouanet, Thesis, Montpellier, France, (1970).
W. H. Zachariasen,Phys. Rev. 73 (1948) 1104.
L. Passerini,Gazz. Chim. Ital. 60 (1930) 762.
P. Duwez andF. Odell,J. Amer. Ceram. Soc. 33 (1950) 274.
G. M. Volten,ibid. 46 (1963) 418.
M. Guillon, J. Millet andS. Palous,Electrochim. Acta 13 (1968) 1425.
V. Longo andD. Minichelli,J. Amer. Ceram. Soc. 56 (1973) 600.
M. Yoshimura andH. K. Bowen,Amer. Ceram. Soc. Bull. 56 (1977) 301.
M. Yoshimura, E. Tani andS. Somiya,Solid State Ionics 314 (1981) 477.
E. Tani, M. Yoshimura andS. Somiya,J. Amer. Ceram. Soc. 66 (1983) 506.
C. Pascual andP. Duran,ibid. 66 (1983) 23.
F. F. Lange,J. Mater. Sci. 17 (1982) 255.
S. Meriani,Mater. Sci. Engng 71 (1985) 396.
C. T. Lynch, in “High Temperature Oxides”, edited by A. M. Alper (Academic Press, New York, 1970) p. 193.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Duran, P., Gonzalez, M., Moure, C. et al. A new tentative phase equilibrium diagram for the ZrO2-CeO2 system in air. J Mater Sci 25, 5001–5006 (1990). https://doi.org/10.1007/BF00580121
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00580121