Abstract
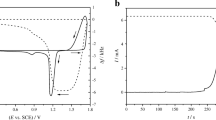
A study has been made of the anodic behavior of lead oxide deposits containing 92, 80 and 60%α-PbO2. The non-porous deposits were prepared by electrodeposition from a plumbite bath containing different proportions of 30% formaldehyde solution, acting as a reductant in the cases of 80 and 60%α-PbO2. Measurements of the steady-state oxygen evolution reaction under constant surface conditions of the electrodes, were carried out galvanostatically within the current-density range 30–190μAcm−2. The oxygen overpotential values were found to increase with increasing the amount of superficially formedβ-PbO2 during anodization in sulphuric acid solution. In the proposed mechanism for the o.e.r, the rate determining step is an electron-transfer reaction. Tafel slopes having values of 220±10 mV dec−1 were obtained. Possible interpretation of these higher slopes is given which is based on the dual barrier model. Potential-time decay curves reveal a retarded oxygen diffusion process.
Similar content being viewed by others
References
P. Rüetschi, R. T. Angstadt and B. D. Cahan,J. Electrochem. Soc. 106 (1959) 547.
U. B. Thomas,ibid. 94 (1968) 42.
R.T. Angstadt, C. J. Venuto and P. Rüetschi,ibid. 109 (1962) 177.
D. A. Frey and H. E. Weaver,ibid. 107 (1960) 930.
A. Kittel, Dissertation Technische Hochschule, Prague, Czechoslovakia (1944).
J. P. Hoare, ‘The Electrochemistry of Oxygen’, John Wiley and Sons, Inc. (1968), p. 243.
K. Topf,Z. Anal. Chem. 26 (1944) 296.
H. Beck and W. F. Wynne-Jones,Trans. Faraday Soc. 50 (1954) 136.
W. J. Lorenz and A. A. El Miligy,J. Electrochem. Soc. 106 (1973) 1698.
J. J. MacDonald and B. E. Conway,Proc. Roy. Soc. (London) A269 (1962) 419.
H. Gohr and E. Lange,Z. Elektrochem. 63 (1958) 673.
R. E. Meyer,J. Electrochem. Soc. 107 (1960) 847.
L. Young, Anodic Films, Academic Press Inc. New York, N.Y. (1961).
B. E. Conway and A. K. Vijh,J. Phys. Chem. 71 (1967) 3637.
A. K. Vijh and B. E. Conway,Z. Anal. Chem. 230 (1967) 81.
J. P. Farr and N. A. Hampson,Chemical Review,72 (1972) p. 702.
I. G. Kiseleva and B. N. Kabanov,Doklady Akad. Nauk SSSR 108 (1956) 864.
M. Steinberg and N. H. Nachttrieb,J. Amer. Chem. Soc. 72 (1950) 3558.
I. G. Kiseleva, B. N. Kabanov and D. Leikis,Dokl. Akad. Nauk SSSR 49 (1954) 805.
G. A. Kokarev, N. G. Bakhchsaraits'yan, V. V. Panteleeva,Tr. Mosk, Khim. Tkhnol. Inst. 54 (1967) 161.
D. I. Leikis and E. K. Ventrem,Proc. Acad. Sci. USSR, Phys. Chem. (Sect. Engl. Transl.)112 (1957) 97.
B. N. Kabanov, I. G. Kiseleva and D. I. Leikis,Dokl. Akad. Nauk SSSR 99 (1954) 805.
J. Burbank, N.R.L. Report 6859, Naval Research Laboratory, Washington, D.C.
E. Bears,Marburger Sitzungsberichte 63 (1928) 214.
A. L. Pitman and G. W. Work, N.R.L. Report 5031, Naval Research Laboratory, Washington, D.C.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Issa, I.M., Abdelaal, M.S. & El Miligy, A.A. Anodic behaviour of α-PbO2 substrates containing different percentages of lower lead oxides. J Appl Electrochem 5, 271–277 (1975). https://doi.org/10.1007/BF00608790
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00608790