Summary
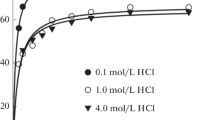
The hydrolysis of the palladium(II) ion in a sodium chloride medium was studied by the e.m.f. method at 25 °C. The data show that the extent of the palladium hydrolysis depends upon the concentration of both palladium and sodium chloride medium. Thus, at a definite pH, the extent of the hydrolysis increases with increasing concentration of palladium, but decreases with increasing concentration of sodium chloride. The stability constants of the complexes obtained, PdOH+ and Pd4(OH) 4+4 , also differ slightly depending upon the concentraton of sodium chloride. The observed medium effect is in agreement with the linear free energy relationship proposed for the metal ion hydrolysis.
Similar content being viewed by others
References
R. M. Izatt, D. Eatough, and J. J. Christensen,J. Chem. Soc. (A), 1301 (1967).
B. I. Nabivanets and L. V. Kalabina,Zh. Neorg. Khim., 15, 1595 (1970).
L. G. Sillén and A. E. Martell,Stability Constants of Metal-ion Complexes, Special Publ., The Chemical Society, London, 1964, No. 17; 1971, No. 25.
C. F. Baes, Jr. and R. E. Mesmer,The Hydrolysis of Cations, Wiley, Toronto, 1976.
N. B. Milić,J. Chem. Soc., Dalton Trans., 1445 (1981).
G. Gran,Analyst., 77, 661 (1952).
W. Forsling, S. Hietanen, L. G. Sillén,Acta Chem. Scand., 6, 901 (1952).
A. S. Brown,J. Amer. Chem. Soc., 56, 646 (1934).
N. B. Milić, Ž. D. Bugarčić, and M. V. Vasić,Bull. Soc. Chim. Beograd., 45, 349 (1980).
P. Brauner, L. G. Sillén, and R. Whiteker,Ark. Kemi, 31, 365 (1968).
D. W. Smith,J. Chem. Educ., 54, 541 (1977).
N. Ingri, W. Kakolowicz, L. G. Sillén, and B. Warnquist,Talanta, 14, 1261 (1967).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Milić, N.B., Bugarčić, Ž.D. Hydrolysis of the palladium(II) ion in a sodium chloride medium. Transition Met Chem 9, 173–176 (1984). https://doi.org/10.1007/BF00618610
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00618610