Abstract
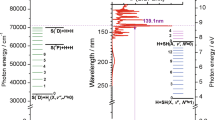
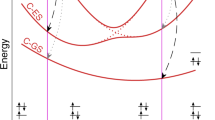
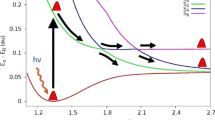
A unified picture of the photodissociation of theC 2 H radical has been developed using the results from the latest experimental and theoretical work. This picture shows that a variety of electronic states ofC 2 are formed during the photodissociation of theC 2 H radical even if photoexcitation accesses only one excited state. This is because the excited states have many avoided corssings and near intersections where two electronic states come very close to one another. At these avoided crossings and near intersections, the excited radical can hop from one electronic state to another and access new final electronic states of theC 2 radical. The complexity of the excited state surfaces also explains the bimodal rotational distributions that are observed in all of the electronic states studied. The excited states that dissociate through a direct path are limited by dynamics to produceC 2 fragments with a modest amount of rotational energy, whereas those that dissociate by a more complex path have a greater chance to access all of phase space and produce fragments with higher rotational excitation. Finally, the theoretical transition moments and potential energy curves have been used to provide a better estimate of the photochemical lifetimes in comets of the different excited states of theC 2 H radical. The photochemically active states are the 22∑+, 22II, 32II, and 32∑+, with photodissociation rate constants of 1.0×10−6, 4.0×10−6, 0.7×10−6, and 1.3×10−6s−1, respectively. These rate constants lead to a total photochemical lifetime of 1.4×105 s.
Similar content being viewed by others
References
Jackson, W.M., Bao, Y. and Urdahl, R.S.: 1991,J. Geophys. Res. 96, 17,569–17,572.
Duflot, D., Robbe, J.-M. and Flament, J.P.: 1994,J. Chem. Phys. 100, 1236–1246.
Jackson, W.M.: 1976,J. Photochem. 5, 107–118.
O'Dell, C.R., Robinson, R.R., Krishna Swamy, K.S., McMarthy, P.J. and Spinard, Hyron: 1988,Astrophys. J. 334, 476–488.
Crifo, J.F.: 1991, in: R.L. Newburn, Jr., M. Neugebauer and J. Rahe (eds.),Comets in the Post-Halley Era, Kluwer, Dordrecht, 2, 937–989.
Huebner, W.F., Keady, J.J. and Lyon, S.P.: 1992,Solar Photo Rates for Planetary Atmospheres and Atmospheric Pollutants, Kluwer, Dordrecht, p. 61 & p. 178.
Lambert, D.L., Sheffer, Y., Danks, A.C., Arpigny, C. and Magain, P.: 1990,Astrophys. J. 353, 640–653.
Jackson, W.M., Halpern, J.B. and Lin, C.S.: 1978,Chem. Phys. Letters 55, 254–258.
McDonald, J.R., Baronavski, A.P. and Donelly, V.M.: 1978,Chem. Phys. 33, 161–170.
Bao, Y., Urdahl, R.S. and Jackson, W.M.: 1991,J. Chem. Phys. 94, 808–809.
Urdahl, R.S., Bao, Y. and Jackson, W.M.: 1988,Chem. Phys. Letters 152, 485–490.
Goodwin, P.M. and Cool, T.A.: 1989,J. Mol. Spectrsc. 133, 230–232.
Wodtke, A.M. and Lee, Y.T.: 1985,J. Phys. Chem. 89, 4744–4751.
Balko, B.A., Zhang, J. and Lee, Y.T.: 1991,J. Chem. Phys. 94, 7958–7966.
Mohammad, F., Bao, Y., Urdahl, R.S., Zahedi, M. and Jackson, W. M.: 1994, in: W.M. Jackson and B.J. Evans (eds.),Henry C. McBay: A Chemical Festschrift, MIT Press, Cambridge, Mass., 101–122.
Fletcher, T.R. and Leone, S.R.: 1988,J. Chem. Phys. 89, 6600–6611.
Satyapal, S. and Bersohn, R.: 1991,J. Phys. Chem. 95, 8004–8006.
Seki, K. and Okabe, H.: 1993,J. Phys. Chem. 97, 5284–5290.
Haijima, A., Fujii, M. and Ito, M.: 1990,J. Chem. Phys. 92, 959–968.
Pechukas, P., Light, J.C. and Rankin, C.: 1966,J. Chem. Phys. 44, 794.
rousselot, P., Goidet-Devel, B., Clairemidi, J. and Moreels, G.: 1994, in: I. Nenner (ed.),Molecules and Grains in Space, AIP Conference Proceedings 312, AIP Press, New York, 255.
Bao, Y.: 1992,The photodissociation dynamics of C 2H2 and C2 H: Detection of the C 2 fragment using laser induced fluorescence, Ph.D. Dissertation, University of California, Davis.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jackson, W.M., Blunt, V., Lin, H. et al. Non-adiabatic interactions in excitedC 2 H molecules and their relationship toC 2 formation in comets. Astrophys Space Sci 236, 29–47 (1996). https://doi.org/10.1007/BF00644319
Issue Date:
DOI: https://doi.org/10.1007/BF00644319