Abstract
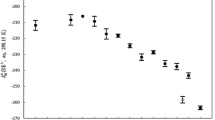
The molar entropy (or heat) of transport of aqueous rare earth chlorides at 0.001, 0.01 and 0.1N have been measured by the potentiometric method using the silver, silver chloride thermocell at a mean temperature of 25°C. Our results indicate that the entropy of transport of rare earth chlorides exhibits a two-series, step function type of dependence on ionic radii. Although this is not the usual S-shaped dependence on ionic radii observed in many thermodynamic and transport properties, the Soret data do seem to show that hydration of the heavier rare earth ions may be rather different from the lighter rare earth ions. The concentration dependence of the entropy of transport have also been investigated for LaCl3, SmCl3, and YbCl3. In all cases the experimental limiting slopes agree well with that predicted by theory based on the electrostatic model.
Similar content being viewed by others
References
A. Habenschuss and F. H. Spedding,J. Chem. Phys. 73, 442 (1980) and references therein. A summary of F. H. Spedding's work may be obtained from A. Habenschuss at Brookhaven National Laboratory, Upton, NY 11973.
F. H. Spedding, M. J. Pikal, and B. O. Ayers,J. Phys. Chem. 70, 2440 (1966).
A. Habenschuss and F. H. Spedding,J. Chem. Phys. 70, 2797, 3758 (1979) and73, 442 (1980).
J. N. Agar, ‘Thermal Diffusion and Related Effects in Solution of Electrolytes’ inThe Structure of Electrolyte Solutions, W. J. Hamer, ed., (Wiley, New York, 1959), Chap. 13.
H. Yow, B. Chakraborty, and J. Lin,Electrochimica Acta 22, 1013 (1977).
B. Chakraborty and J. Lin,J. Solution Chem. 3, 183 (1976).
H. J. V. Tyrrell,Chem. Comm. 456 (1967).
P. Y. Kahana and J. Lin,J. Chem. Phys. 74, 2993 (1980) and75, 5043 (1981).
J. N. Agar, ‘Thermal Diffusion and Related Effects in Solution of Electrolytes’ inThe Structure of Electrolyte Solutions, W. J. Hamer, ed., (Wiley, New York, 1959), Chap. 13.
J. A. Bierlein,J. Chem. Phys. 23, 101 (1955).
J. N. Agar,Trans. Faraday Soc. 56, 776 (1960).
J. Lin, J. A. Bierlein, and J. G. Becsey,J. Solution Chem. 3, 827 (1974).
J. N. Agar and J. C. R. Turner,Proc. Royal Soc., A 255, 307 (1960).
F. H. Spedding and I. S. Yaffee,J. Am. Chem. Soc. 74, 475 (1952).
F. H. Spedding and J. L. Dye,J. Am. Chem. Soc. 76, 879 (19540.
F. H. Spedding, R. A. Nelson, and J. A. Rard,J. Chem. Eng. 19, (4) 379 (1974).
P. N. Snowdon and J. C. R. Turner,Trans. Faraday Soc. 56, 1812 (1960).
E. Helfand and J. G. Kirkwood,J. Chem. Phys. 32, 857 (1960); E. Helfand, R. J. Bearman, and V. S. Vaidhyanathan,J. Math. Phys. 4 160 (1963); R. J. Bearman and V. S. Vaidhyanathan,J. Chem. Phys. 39, 3441 (1963).
J. N. Agar ‘Thermogalvanic Cells’ inAdvances in Electrochemistry and Electrochemical Engineering, Vol. 3, (Interscience, New York, 1963), Chap. 2.
J. N. Agar, private communication.
H. F. Halliwell and S. C. Nyburg,Trans. Faraday Soc. 59, 1126 (1963).
S. L. Bertha and G. R. Choppin,Inorg. Chem. 8, 613 (1969).
R. Pottel, ‘Dielectric Properties’ inWater: A Comprehensive Treatise, Volume 3, F. Franks, ed., (Plenum Press, New York, 1973), Chap. 8.
H. S. Frank and E. W. Evans,J. Chem. Phys. 13, 507 (1945).
C. D. Wood and W. A. Hawksworth,J. South African Chem. Inst. 24, 170 (1971).
J. Lin,J. Solution Chem. 8, 125 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yow, H., Lin, Jl. Thermal diffusion of lanthanide chlorides. J Solution Chem 12, 487–502 (1983). https://doi.org/10.1007/BF00651700
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00651700