Abstract
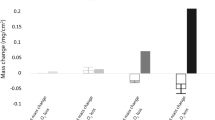
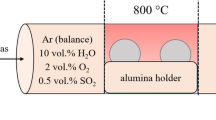
The corrosion of Co-Nb alloys containing up to 30 wt.% Nb in H2-H2S-H2O gas mixtures was studied over the temperature range of 600–800°C. The gas composition falls in the stability region of cobalt sulfide and Nb2O5 in the phase diagrams of the Co-O-S and Nb-O-S systems at all temperatures studied. Duplex scales, consisting of an outer layer of cobalt sulfide and a complex, heterophasic inner layer, were formed at all temperatures studied. In addition to cobalt sulfide and CoNb3S6, a small amount of NbO2 was found in the inner layer. The reason for the formation of NbO2 over that of Nb2O5 in the scale is that the outer sulfide scale lowers the oxygen activity within the scale into the NbO2-stability region. Two-stage kinetics were observed for all alloys, including an initial irregular stage usually followed by a steady-state parabolic stage. The steady-state parabolic rate constants decreased with increasing amounts of Nb, except for Co-20Nb corroded at 700°C. Nearly identical kinetics were observed for Co-20Nb corroded at 600°C and 700°C. The presence of NbO2 particles leads only to a limited decrease of the available cross-section area for the outward-diffusing metal ions. The activation energies for all alloys are similar and are in agreement with those obtained in a study of the sulfidation of the same alloys. The primary corrosion mechanism involves an outward Co transport.
Similar content being viewed by others
References
E. J. Kubel, Jr.,Adv. Mater. Proc. 135(3), 37 (1989).
K. Natesan,Corrosion 41(11), 646 (1985).
T. T. Huang, L. E. Toth, and E. Pfender, inProceedings of the International Conference on Behavior of High Temperature Alloys in Aggressive Environments, I. Kirmanet al., eds. (Metal Society, London, 1980), p. 717.
K. N. Strafford and P. K. Datta,Mater. Sci. Tech. 5, 765 (1989).
M. F. Chen and D. L. Douglass,Oxid. Met. 31, 237 (1989).
B. Gleeson, D. L. Douglass, and F. Gesmundo,Oxid. Met. 31, 209 (1989).
R. V. Carter, D. L. Douglass, and F. Gesmundo,Oxid. Met. 31, 341 (1989).
M. F. Chen, D. L. Douglass, and F. Gesmundo,Oxid. Met. 32, 185 (1989).
G. Wang, R. V. Carter, D. L. Douglass,Oxid. Met. 32, 273 (1989).
B. Gleeson, D. L. Douglass, and F. Gesmundo,Oxid. Met. 33, 425 (1990).
P. Singh and N. Birks,Oxid. Met. 12, 23 (1978).
F. Gesmundo and C. De Asmundis, inProceedings of the International Conference on Behavior of High Temperature Alloys in Aggressive Environments, I. Kirmanet al., ed. (Metal Society, London, 1980), p. 435.
K. Holthe and P. Kofstad,Corros. Sci. 20, 919 (1980).
N. S. Jacobson and W. L. Worrell,J. Electrochem. Soc. 131, 1182 (1984).
Metals Handbook, Metallography, Structures and Phases, Vol.8, 8th Ed. (ASM, Metals Park, Ohio, 1973).
D. Hodouin,Met. Trans. B 6B, 223 (1975).
S. R. Shatynski,Oxid. Met. 11, 307 (1977).
H. R. Larson and J. F. Elliott,Trans. Met. Soc. AIME 239, 1713 (1967).
G. J. Yurek and M. H. LaBranche, inProceedings of the Conference on Corrosion-Erosion-Wear of Materials in Emerging Fossil Energy Systems, A. V. Levy, ed. (NACE, Houston, 1982). p. 933.
T. Flatley and N. Birks,J. Iron Steel Inst. 209, 523 (1971).
F. Gesmundo, D. J. Young, and S. K. Roy,High Temp. Mater. Proc. 8, 149 (1989).
J. Stringer and D. P. Whittle,Rev. Int. Htes Temp. Refract. 14, 6 (1977).
J. M. van den Berg and P. Cossee,Inorg. Chim. Acta 2, 143 (1968).
P. Kofstad,High-Temperature Oxidation of Metals (Wiley and Sons, New York, 1966).
H. Rau,J. Phys. Chem. 37, 931 (1976).
S. Mrowec, S. Rusiecki, and A. Wojtowicz,Bull. Pol. Acad. Sci. Chem. 34, 411 (1986).
A. Davin,Cobalt. 30, 19 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shing, C.C., Douglass, D.L. & Gesmundo, F. The high-temperature corrosion behavior of Co-Nb alloys in mixed-gas atmospheres. Oxid Met 37, 167–187 (1992). https://doi.org/10.1007/BF00665188
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00665188