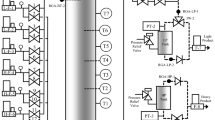
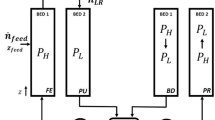
Abstract
A detailed model for the recuperative parametric pumping is presented. The model includes intraparticle mass transfer resistance, axial diffusion and non-linear equilibrium represented by Langmuir equation. The sensitivity studies shows that process performance strongly increases when cycle time increases and φ B /φ T ratio and particle size decreases. It also shows that bottom and top dead volumes do not influence much the process performance. Evolution of the histories of concentrations and temperatures, the bed performance from cycle to cycle and the bed dynamics at the cyclic steady state have been discussed.
The model revealed itself as useful to simulate the behavior of the recuperative parametric pumping process and was applied to predict optimal experimental results for the system phenol-water/Duolite ES-861 (Part II).
Similar content being viewed by others
Abbreviations
- a i (z*, ϑ):
-
coefficients of the trial solution
- a w :
-
specific area of wall, m−1
- b :
-
separation parameter
- C :
-
solute concentration in the fluid phase, kg/m3
- C E :
-
initial (feed) solute concentration, kg/m3
- c pf :
-
heat capacity of the fluid, kJ/(kg.K)
- C p :
-
solute concentration in the pores, kg/m3
- c s :
-
heat capacity of the adsorbent, kJ/(kg.K)
- d :
-
column diameter, m
- D ax :
-
axial dispersion coefficient, m2/s
- D m :
-
molecular diffusion coefficient, m2/s
- D P :
-
pore diffusivity, m2/s
- d p :
-
particle diameter, m
- f H :
-
humidity factor for the adsorbent (kg dry resin/kg wet resin)
- G f :
-
fluid mass flowrate, kg/(m2.s)
- h k :
-
length of subintervalk
- h we :
-
wall heat transfer coefficient, kJ/(m2.s.K)
- (−ΔH):
-
heat of adsorption, kJ/kgmol
- H i (g k ):
-
Hermite polynomials over the subintervalk
- j D :
-
Chilton Colburn factor
- K(T):
-
slope of linear isotherm
- K ae :
-
axial thermal conductivity, kJ/(m.s.K)
- K f :
-
film mass transfer coefficient, m/s
- K L :
-
parameter of the Langmuir isotherm, m3 solution /kg solute
- k 0 :
-
parameter of the modified Langmuir isotherm, m3 solution /kg solute
- L :
-
bed height, m
- m(T):
-
mass capacity parameter
- n :
-
number of cycles
- N D :
-
number of mass transfer units for pore diffusion
- N f :
-
number of film mass transfer units
- N hw :
-
number of wall heat transfer units
- Pe:
-
mass Peclet number
- Pe h :
-
thermal Peclet number
- q :
-
adsorbed solute concentration, kg solute /kg dry resin
- Q :
-
flowrate in the column, m3/s
- Q hc :
-
flowrate in the hot half cycle, m3/s
- Q cc :
-
flowrate in the cold half cycle, m3/s
- Q ∞ :
-
maximum adsorbent capacity, kg solute /kg dry resin
- (Qπ/w):
-
reservoir displacement volume, m3
- r :
-
radial coordinate, m
- R :
-
ideal gas constant, kJ/(kgmol.K)
- R 0 :
-
particle radius, m
- Re:
-
particle Reynolds number
- Sc:
-
Schmidt number
- Sh:
-
Sherwood number
- t :
-
time, s
- t c :
-
cycle time, s
- t cc :
-
cold half cycle time, s
- t hc :
-
hot half cycle time, s
- T :
-
temperature, K
- T a :
-
ambient temperature, K
- T cc :
-
cold temperature, K
- T hc :
-
hot temperature, K
- u :
-
superficial velocity, m/s
- u cn :
-
concentration wave velocity, m/s
- u i :
-
interstitial velocity, m/s
- u th :
-
thermal wave velocity, m/s
- u*:
-
normalized radial coordinate
- u * k :
-
inferior limit of subintervalk
- u +1/* k :
-
upper limit of subintervalk
- V U :
-
volume percolated in upward flow, m3
- X :
-
normalized fluid phase concentration
- X p :
-
normalized fluid concentration in the pores
- z :
-
spatial coordinate, m
- z*:
-
normalized axial coordinate
- 〈〉:
-
average value
- ξ h :
-
thermal capacity parameter
- ε :
-
bed porosity
- ε p :
-
intraparticle porosity
- φ B ,φ T :
-
fractions of theQπ/ω that are withdrawn as bottom and top products, respectively
- v :
-
kinematics viscosity, m2/s
- ρ ap :
-
apparent density of the adsorbent, kg/m3
- ρ f :
-
density of the fluid, kg/m3
- ρ s :
-
density of the adsorbent, kg/m3
- ρ h :
-
wet density of the adsorbent, kg/m3
- ϑ :
-
normalized time
- ω :
-
frequency of temperature change
- τ :
-
space time, s
- τ p :
-
tortuosity factor
- π/ω :
-
duration of half cycle
- E :
-
entrance
- BP:
-
bottom product
- cc:
-
cold half cycle
- hc:
-
hot half cycle
- 0:
-
initial
- TP:
-
top product
References
Apostolopoulos, G.P., “The Parametric Pumping as a Chemical Reactor,”Ind. Eng. Chem. Fundam.,14, 11–16 (1975).
Apostolopoulos, G.P., “The Parametric Pumping as a Chemical Reactor,”Ind. Eng. Chem. Fundam.,15, 14 (1976).
Chen, H.T. and F.B. Hill, “Characteristics of Batch, Semicontinuous and Continuous Equilibrium Parametric Pumps,”Separation Science,6, 411–434 (1971).
Chen, H.T., J.L. Rak, J.D. Stokes, and F.B. Hill, “Separation Via Semicontinuous Parametric Pumping,”AIChE J.,18, 356–361 (1972).
Chen, H.T., J. Reiss, J.J. Stokes, and F. Hill, “Separation Via Continuous Parametric Pumping,”AIChE J.,19, 589–595 (1973).
Chen, H.T., T.K. Hsieh, H.C. Lee, and F.B. Hill, “Separation of Proteins Via Semicontinuous pH Parametric Pumping,”AIChE J.,23(5), 695–701 (1977).
Chen, H.T., Y.W. Wong, and S. Wu, “Separation of Proteins Via Multicolumn pH Parametric Pumping,”AIChE J.,26(5), 839–849 (1980).
Chen, H.T., W.T. Yang, U. Pancharoen, and R. Parisi, “Continuous Fractionation of Protein Mistures by pH Parametric Pumping,”AIChE J.,25(2), 320–327 (1979).
Costa, C., A. Rodrigues, G. Grevillot, and D. Tondeur, “Purification of Phenolic Wastewater by Parametric Pumping: Non Mixed Dead Volume Equilibrium Model,”AIChE J.,28(1), 73–85 (1982).
Costa, C. and A. Rodrigues, “Design of Cyclic Fixed Bed Adsorption Processes. Part I: Phenol Adsorption on Polymeric Adsorbents,”AIChE J.,3(1), 1645–1654 (1985).
Ferreira, L.M., “Dynamics of Sorption Processes: Separation by Parametric Pumping and Recovery of Metals with Complexing Resins”, Ph.D. Thesis, University of Porto, Portugal (1994).
Finlayson, B.A.,Non-linear Analysis in Chemical Engineering, McGraw-Hill Int., New York, 1980.
Foo, S.C. and R.G. Rice, “On The Prediction of Ultimate Separation in Parametric Pumps,”AIChE J.,21, 1149–1158 (1975).
Foo, S.C. and R.G. Rice, “Steady State Predictions for Nonequilibrium Parametric Pumps,”AIChE J.,23, 120–125 (1977).
Gregory, R.A. and N.H. Sweed, “Parametric Pumping Behavior of Open Systems Part I: Analytical Solutions,”The Chem. Eng. J.,1, 207–216 (1970).
Grevillot, G. and D. Tondeur, “Equilibrium Staged Parametric Pumping I—Single Transfer Step per Half-Cycle and Total Reflux—the Analogy with Distillation,”AIChE J.,22, 1055–1063 (1976).
Grevillot, G. and D. Tondeur, “Equilibrium Staged Parametric Pumping II—Multiple Transfer Steps per Half-Cycle and Reservoir Staging,”AIChE J.,23, 840–851 (1977).
Grevillot, G., “Equilibrium Staged Parametric Pumping Part III. Open Systems at Steady-State-McCabe-Thiele Diagrams,”AIChE J.,26, 120–131 (1980).
Gupta, R. and N.H. Sweed, “Modeling of Nonequilibrium Effects in Parametric Pumping,”Ind. Eng. Chem. Fundam.,12, 335–341 (1973).
Hollein, H.C., H.-C. Ma, C.-R. Huang, and H.T. Chen, “Parametric Pumping with pH and Electric Field: Protein Separations,”Ind. Eng. Chem. Fundam.,12, 205–214 (1982).
Lavie, R. and M.J. Reilly, “Limit Cycles in Fixed Beds Operation in Alternating modes,”Chem. Engng. Sci.,27, 1835–1843 (1972).
Madsen, N.K. and R.F. Sincovee, “PDECOL: General Collocation Software for Partial Differential Equations,”ACM Trans. Math. Software,3, 326–351 (1979).
Pigford, R., B. Baker, and D. Blum, “An Equilibrium Theory of the Parametric Pump,”Ind. Eng. Chem. Fundam.,8, 144–149 (1969).
Rice, R.G., “Dispersion and Ultimate Separation,”Ind. Eng. Chem. Fundam.,12, 406–412 (1973).
Rice, R.G., “Progress in Parametric Pumping, Separation and Purification Methods,”5, 139–188 (1976).
Rice, R.G. and S.C. Foo, and G.G. Gough, “Limiting Separation in Parametric Pumps,”Ind. Eng. Chem. Fundam.,18, 117–123 (1979).
Rice, R.G. and S.C. Foo, “Continuous Desalination Using Cyclic Mass Transfer on Bifunctional Resins,”Ind. Eng. Chem. Fundam.,20, 150–155 (1981).
Sweed, N.H. and R.H. Wilhelm, “Parametric Pumping Separations via Direct Thermal Mode,”Ind. Eng. Chem. Fundam.,8, 221–231 (1969).
Sweed, N.H. and R.A. Gregory, “Parametric Pumping: Modeling Direct Thermal Separations of Sodium Chloride-Water in Open and Closed Systems,”AIChE J.,17, 171–176 (1971).
Wankat, P.C. “Continuous Recuperative Mode Parametric Pumping,”Ind. Eng. Chem. Fundam.,12, 372–380 (1973).
Wankat, P.C. “Liquid-Liquid Extraction Parametric Pumping,”Chem. Engng. Sci.,33, 723–733 (1978).
Wilhelm, R.H., A.W. Rice, and A.R. Bendelius, “Parametric Pumping: A Dynamic Principle for Separation Fluid Mixtures,”Ind. Eng. Chem. Fundam.,5, 141–144 (1966).
Wilhelm, R.H., A.W. Rice, R.W. Rolke, and N.H. Sweed, “A Dynamic Principle for Separation Fluid Mixtures,”Ind. Eng. Chem. Fundam.,7, 337–348 (1968).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ferreira, L.M., Rodrigues, A.E. Adsorptive separation by thermal parametric pumping part I: Modeling and simulation. Adsorption 1, 213–231 (1995). https://doi.org/10.1007/BF00704225
Issue Date:
DOI: https://doi.org/10.1007/BF00704225