Abstract
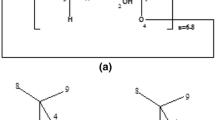
The crystal of theβCD-3,5-dimethylbenzoic acid inclusion complex is triclinicP1 witha=15.707(3),b=15.694(3),c=15.999(3) Å, α=101.502(5),β=101.557(5), γ=103.805(4)°,V=3624 Å3, andZ=1. Two crystallographically independentβCD molecules form a dimer by means of hydrogen bonds between secondary hydroxyl groups in which disordered guest molecules in six orientations are accommodated. In two of them the guest molecules are situated at the dimer interface, their carboxyl groups interacting with the carboxyl groups of two other guest orientations that are found on either side of the monomer cavity. In the two last orientations the guest molecules enter the monomer cavities in an opposite sense with the carboxyl groups protruding from the primary side and are stabilized by cyclodextrin hydroxyl groups. A dense water network is formed in the interdimer space consisted of 18.2 water molecules distributed over 24 sites.
Similar content being viewed by others
References
J. Szejtli:Cyclodextrin Technology, Kluwer Academic Publishers (1989).
D. Mentzafos, I.M. Mavridis, G. Le Bas, and G. Tsoucaris.Acta Crystallogr., Sect. B 47, 746 (1991).
I.M. Mavridis and E. Hadjoudis:Carbohydr. Res. 229, 1 (1992).
A. Rontoyianni, I.M. Mavridis, E. Hadjoudis, and A.J.M. Duisenberg:Carbohydr. Res. 252, 19 (1994).
G.M. Sheldrick: SHELX-76, Program for Crystal Structure Determination, University of Cambridge, England (1976).
T.A. Jones and M. Kjeldgaard: O version 5.9, Molecular Modelling Program, Uppsala, Sweden (1993).
G.M. Sheldrick: SHELXL-93, Program for Crystal Structure Refinement (1993).
ATOMS-PC V2.3, Atomic Structure Display Software, Shape Software (1993).
Author information
Authors and Affiliations
Additional information
Dedicated to Professor József Szejtli.
Supplementary Data relating to this article are deposited with the British Library as Supplementary Publication No. SUP 82175 (61 pages).
Rights and permissions
About this article
Cite this article
Rontoyianni, A., Mavridis, I.M. The crystal structure of the inclusion complex of cyclomaltoheptaose (β-cyclodextrin) with 3,5-dimethylbenzoic acid. J Incl Phenom Macrocycl Chem 18, 211–227 (1994). https://doi.org/10.1007/BF00708728
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00708728