Summary
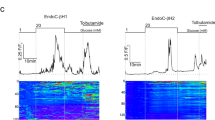
We have evaluated the potential of the clonal insulin-secretory cell line HIT-T15 as a model system for investigating stimulus-secretion coupling in pancreatic B cells. In contrast to other cell lines, HIT cell insulin secretion was consistently stimulated 2- to 3-fold by D-glucose. The maximally effective concentration of glucose was 10 mmol/1; between 2 and 10 mmol/l glucose the increase in insulin release was paralleled by an increased rate of glucose oxidation. The main characteristics of glucose-stimulated insulin release by HIT cells were essentially similar to those of normal islets. Thus, the response was (1) specific for metabolizable sugars (D-mannose and D-glyceraldehyde stimulated insulin release but L-glucose and D-galactose were ineffective); (2) markedly dependent on extracellular Ca2+ concentration; (3) potentiated by forskolin, glucagon, acetylcholine and 12-0-tetradecanoyl phorbol 13-acetate; (4) inhibited by adrenaline or somatostatin; (5) showed a biphasic pattern of release in perifusion experiments, with both phases being potentiated by forskolin. The secretory response of the HIT cells to amino acids was also similar to that of normal islets. Thus, L-leucine and its deamination product 2-ketoisocaproate were effective stimuli, whereas L-isoleucine and L-glutamine were ineffective. Insulin release from HIT cells could also be evoked by the sulphonylureas glibenclamide and tolbutamide and by an increase in concentration of extracellular K+ to 40 mmol/1. The content of cyclic AMP in HIT cells was increased modestly by glucose but not by an increase in extracellular K+. Forskolin elicited a 4-fold increase in cyclic AMP content. We conclude that HIT cells retain the essential features of the insulin secretory response of normal B cells and represent an important tool for further biochemical characterisation of the secretory system.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Gazdar AF, Chick WL, Oie HK, Sims HL, King DL, Weir GC, Lauris V (1980) Continuous, clonal insulin- and somatostatin-secreting cell lines established from a transplantable rat islet cell tumor. Proc Natl Acad Sci USA 77: 3519–3523
Santerre RF, Cook RA, Crisel RMD, Sharp JD, Schmidt RJ, Williams DC, Wilson CP (1981) Insulin synthesis in a clonal cell line of Simian virus 40-transformed hamster pancreatic beta cells. Proc Natl Acad Sci USA 78: 4339–4343
Chick WL, Warren S, Chute RN, Like AA, Lauris V, Kitchen KC (1977) A transplantable insulinoma in the rat. Proc Natl Acad Sci USA 74: 628–632
Praz GA, Halban PA, Wollheim CB, Blondel B, Strauss AJ, Renold AE (1983) Regulation of immunoreactive-insulin release from a rat cell line (RINm5F). Biochem J 210: 345–352
Halban PA, Praz GA, Wollheim CB (1983) Abnormal glucose metabolism accompanies failure of glucose to stimulate insulin release from a rat pancreatic cell line (RINm5F). Biochem J 212: 439–443
Hill RS, Boyd III AE (1985) Perifusion of a clonal cell line of Simian virus 40-transformed beta cells. Insulin secretory dynamics in response to glucose, 3-isobutylmethylxanthine and potassium. Diabetes 34:115–120
Boyd III AE, Hill RS, Oberwetter AM, Berg M (1986) Calcium dependency and free calcium concentrations during insulin secretion in a hamster beta cell line. J Clin Invest 77: 774–781
Swope SL, Schonbrunn A (1984) Bombesin stimulates insulin secretion by a pancreatic islet cell line. Proc Nail Acad Sci USA 81: 1822–1866
Christie MR, Ashcroft SJH (1985) Substrates for cyclic AMP-dependent protein kinase in islets of Langerhans. Studies with forskolin and catalytic subunit. Biochem J 227: 727–736
Ashcroft SJH, Crossley JR (1975) Effects of glucose, N-acetylglucosamine, glyceraldehyde and other sugars on insulin release in vivo. Diabetologia 11: 274–279
Ashcroft SJH, Hedeskov CJ, Randle PJ (1970) Glucose metabolism in mouse pancreatic islets. Biochem J 118: 143–154
Pipeleers D (1984) Islet cell interactions with pancreatic B-cells. Experientia 40: 1114–1126
Hellman B (1975) The significance of calcium for glucose stimulation of insulin release. Endocrinology 97: 392–398
Wollheim CB, Sharp GWA (1981) Regulation of insulin release by calcium. Physiol Rev 61: 914–973
Gylfe E, Hellman B, Sehlin J, Taljedal IB (1984) Interaction of sulphonylureas with the pancreatic B-cell. Experientia 40:1126–1134
Smith PH, Porte JRD, Robertson RP (1979) Neural regulation of the endocrine pancreas. In: Pierlussi J (ed) Endocrine pancreas and diabetes. Excerpta Medica, Amsterdam, pp 64–95
Best L, Dunlop M, Malaisse WJ (1984) Phospholipid metabolism in pancreatic islets. Experientia 40: 1075–1084
Harrison DE, Ashcroft SJH, Christie MR, Lord JM (1984) Protein phosphorylation in the pancreatic B-cell. Experientia 40: 1075–1084
Castagna M, Takai Y, Kaibuchi K, Sano K, Kikkawa U, Nishizuka Y (1982) Direct activation of calcium-activated, phospholipiddependent protein kinase by tumour-promoting phorbol esters. J Biol Chem 257: 7847–7851
Christie MR, Ashcroft SJH (1984) Cyclic AMP-dependent protein phosphorylation and insulin secretion in intact islets of Langerhans. Biochem J 218: 87–99
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ashcroft, S.J.H., Hammonds, P. & Harrison, D.E. Insulin secretory responses of a clonal cell line of simian virus 40-transformed B cells. Diabetologia 29, 727–733 (1986). https://doi.org/10.1007/BF00870283
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00870283