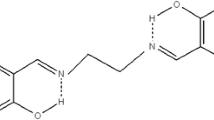
Conclusions
A kinetic study has been made of the hydrolysis of a number of o-aminomethylphenols and their iodomethylates at various levels of pH of the medium. The reaction proceeds through the zwitterion and anionic forms by an elimination-addition mechanism, leading to the formation of o-hydroxybenzyl alcohol.
Similar content being viewed by others
Literature cited
P. D. Gardner, H. S. Rafsajani, and L. Rand, J. Am. Chem. Soc.,81, 3364 (1959).
S. V. Cavit, H. R. Sarrafizaden, and P. D. Gardner, J. Org. Chem.,27, 1121 (1962).
F. Troxler, Helv. Chim. Acta,51, 1214 (1968).
R. Andrisano, C. Delia Casa, and M. Tramontini, J. Chem. Soc. C, 1866 (1970).
B. E. Ivanov and L. A. Khismatullina, Izv. Akad. Nauk SSSR, Ser. Khim., 2650 (1968).
B. E. Ivanov, L. A. Valitova, and T. G. Vavilova, Izv. Akad. Nauk SSSR, Ser. Khim., 768 (1968).
S. Yukitani, K. Mamoru, and N. Kenji, J. Chem. Soc. Jpn., Chem. Ind. Chem., 1123 (1978).
J. H. Burchalter, J. Am. Chem. Soc.,68, 1894 (1946).
B. Reichert, Die Mannich Reaction, Springer-Verlag, Berlin (1959).
A. B. Teitel'baum, K. A. Derstuganova, N. A. Shishkina, L. A. Kudryavtseva, V. E. Bel'skii, and B. E. Ivanov, Izv. Akad. Nauk SSSR, Ser. Khim., 803 (1980).
C. McIntosh and O. L. Champan, J. Chem. Soc., Chem. Commun., 71 (1971).
A. B. Teitel'baum, A. M. Kurguzova, L. A. Kudryavtseva, V. E. Bel'skii, and B. E. Ivanov, Izv. Akad. Nauk SSSR, Ser. Khim., 531 (1981).
N. A. Shishkina, K. A. Derstuganova, L. A. Kudryavtseva, V. E. Bel'skii, and B. E. Ivanov, Izv. Akad. Nauk SSSR, Ser. Khim., 1259 (1976).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 1, pp. 81–85, January, 1982.
Rights and permissions
About this article
Cite this article
Teitel'baum, A.B., Kudryavtseva, L.A., Bel'skii, V.E. et al. Kinetics of hydrolysis of o-aminomethylphenols and their ammonium salts. Russ Chem Bull 31, 72–75 (1982). https://doi.org/10.1007/BF00954412
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00954412