Abstract
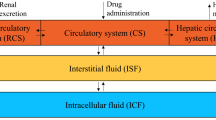
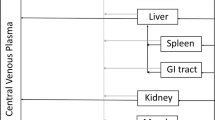
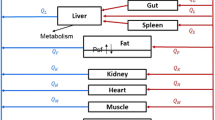
A lumped compartmental model has been derived to predict methotrexate concentration as a function of time for L1210 cells in BD2F 1 female mice at doses ranging from 3 mg/kg to 400 mg/kg. Using standard methods of parameter estimation as well as experimental determinations, an integrated approach was derived to account for the differences between the subcutaneous (s.c.) and intraperitoneal (i.p.) modes of injection. It was found that a single generalized forcing function can be used to fit plasma concentration after s.c. injection for all doses. Adequate fits (average error<20% while the standard deviation of experimental determinations was±22%) of L1210 cell data after s.c. injection were obtained. The best results were for a maximum facilitated influx constant Vmax of 0.424 Μg/min/ml, a Michaelis influx constant Km of 1,42 Μg/ml, and a first-order efflux constant α of 0.047 min−1.The model simulations were not sensitive to Vmax, Km,and αso long as the ratio Vmax/αwas approximately 9Μg/ml. The values of V max ,K m ,and α which were obtained from our analysis of the in vivodata can be explained on the basis of previously performed in vitroexperiments. The parameters obtained from modeling the s.c. data were then applied for i.p. injection data. The resulting fits were adequate (average error<20% while the standard deviation of experimental determinations was±22%). A single generalized forcing function for drug concentration in the peritoneal cavity after i.p. injection for all doses was derived. The application of these results enables the prediction of methotrexate concentration in neoplastic cells at other doses after either s.c. or i.p. injection.
Similar content being viewed by others
References
R. L. Dedrick, D. S. Zaharko, and R. J. Lutz. Transport and binding of methotrexatein vivo.J. Pharm. Sci. 62:882–890 (1973).
R. K. Jain. Dynamics of drug distribution in solid tumors. Ph.D dissertation, University of Delaware. University Microfilms, Ann Arbor, Mich., 1976.
I. D. Goldman. Uptake of drugs and resistance. InDrug Resistance and Selectivity: Biochemical and Cellular Basis, Academic Press, New York, 1973.
R. J. Lutz, R. L. Dedrick, J. A. Straw, M. M. Hart, P. Klubes, and D. S. Zaharko. The kinetics of methotrexate distribution in spontaneous canine lymphosarcoma.J. Pharmacokin. Biopharm. 3:77–97 (1975).
R. L. Dedrick, R. J. Lutz, K. H. Yang, W. P. Fung, and D. S. Zaharko. Pharmacokinetic model for the transport of methotrexate into Lewis lung tumor in mice. Reprint of paper presented at the 70th Annual AIChE Meeting, New York, 1977.
F. M. Sirotnak and R. C. Donsbach. Comparative studies on the transport of aiminopterin, methotrexate, and methasquin by the L1210 leukemia cell.Cancer Res. 32:2120–2126 (1972).
F. M. Sirotnak and R. C. Donsbach. Kinetic correlates of methotrexate transport and therapeutic responsiveness in murine tumors.Cancer Res. 36:1151–1158 (1976).
M. Dembo and F. M. Sirotnak. Antifolate transport in L1210 leukemia cells: Kinetic evidence for the non-identity of carriers for influx and efflux.Biochim. Biophys. Acta 448:505–516 (1976).
B. E. P. Box. Fitting empirical data.Ann. N.Y. Acad. Sci. 86:792–816 (1960).
F. M. Sirotnak and R. C. Donsbach. Differential cell permeability and the basis for selective activity of methotrexate during therapy of the L1210 leukemia.Cancer Res. 33:1290–1294 (1973).
F. M. Sirotnak and R. C. Donsbach. The intracellular concentration dependence of antifolate inhibition of DNA synthesis in L1210 leukemia cells.Cancer Res. 34:3332–3340 (1974).
F. M. Sirotnak and R. C. Donsbach. Further evidence for a basis of selective activity and relative responsiveness during antifolate therapy of murine tumors.Cancer Res. 35:1737–1744 (1975).
F. M. Sirotnak and R. C. Donsbach. A basis for the difference in toxicity of methotrexate, aminopterin, and methasquin in mice.Biochem. Pharmacol. 24:156–158 (1975).
D. J. Hutchison, D. J. Robinson, D. Martin, O. L. Ittelsohn, and J. Dillenberg. Effects of selected anticancer drugs on the survival time of mice with L1210 leukemia: Relative response of antimetabolite resistant strains.Cancer Res. 22:57–72 (1962).
S. Margolis, F. S. Philips, and S. S. Sternberg. The cytotoxicity of methotrexate in mouse small intestine in relation to inhibition of folic acid reductase and of DNA synthesis.Cancer Res. 31:2037–2046 (1971).
F. M. Sirotnak, R. C. Donsbach, D. M. Dorick, and D. M. Moccio. Tissue pharmacokinetics, inhibition of DNA synthesis, and tumor cell kill after high-dose methotrexate in murine tumor models.Cancer Res. 36:4672–4678 (1976).
W. C. Werkheiser. Specific binding of 4-amino folic analogues by folic acid reductase.J. Biol. Chem. 236:888–893 (1961).
F. S. Philips, F. M. Sirotnak, J. E. Sodergren, and D. J. Hutchison. Uptake of methotrexate, aminopterin, and methasquin and inhibition of dihydrofolate reductase and of DNA synthesis in mouse small intestine.Cancer Res. 33:153–158 (1973).
K. B. Bischoff, R. L. Dedrick, D. S. Zaharko, and J. A. Longstreth. Methotrexate pharmacokinetics.J. Pharm. Sci. 60:1128–1133 (1971).
F. M. Sirotnak and R. C. Donsbach. Stereochemical characteristics of the folate-antifolate transport mechanism in L1210 leukemia cells.Cancer Res. 34:371–377 (1974).
I. D. Goldman, N. S. Lichtenstein, and V. T. Oliverio. Carrier-mediated transport of the folic acid analogue, methotrexate, in L1210 leukemia cells.J. Biol. Chem. 243:5004–5017 (1968).
A. L. Babb, P. J. Johansen, M. J. Strand, H. Tenckhoff, and B. H. Scribner. Bi-directional permeability of the human peritoneum to middle molecules.Proc. Eur. Dialysis Transplant. Assoc. 10:247–261 (1973).
R. P. Popovich, J. W. Moncrief, J. F. Decherd, J. S. Na, and J. W. Sawyer. Physiological transport parameters in patients in peritoneal and hemodialysis.Proceedings of 10th Annual Contractors' Conference, Artificial Kidney-Chronic Uremia Program, National Institute of Arthritis, Metabolism, and Digestive Diseases, Bethesda, Md., January 1977, pp. 95–98.
R. L. Dedrick, C. E. Myers, R. M. Bungay, and V. T. De Vita, Jr. Pharmacokinetic rationale for peritoneal drug administration in treatment of ovarian cancer.Cancer Treat. Rep. 62:1–11 (1978).
Knud Schmidt-Nielsen. Energy metabolism, body size, and problems of scaling.Fed. Proc. 29:1524 (1970).
K. B. Bischoff, R. L. Dedrick, and D. S. Zaharko. Preliminary model for methotrexate pharmacokinetics.J. Pharm. Sci. 59:149–154 (1970).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weissbrod, J.M., Jain, R.K. & Sirotnak, F.M. Pharmacokinetics of methotrexate in leukemia cells: Effect of dose and mode of injection. Journal of Pharmacokinetics and Biopharmaceutics 6, 487–503 (1978). https://doi.org/10.1007/BF01062105
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01062105